Background:
The anemia highly associated with myelodysplastic syndrome (MDS) has been linked to numerous transcriptional factor regulation abnormalities, specifically SMAD 2/3 upregulation that forms an inhibitory feedback loop to downregulate erythrocyte stimulation (ESA), which can have therapeutic implications. There is evidence that zinc contributes to erythropoiesis through multifactorial mechanisms involving regulation and transportation of necessary micronutrients including copper and iron. Zinc is involved in cell signaling pathways for erythropoiesis including acting as a cofactor in the SMAD 2/3 complex, TGF-β signaling, and incorporation of zinc into GATA-1 (Zinc finger transcription factor). The interaction with zinc in signaling cascades such as the growth hormone and insulin-like growth factor-1 pathways also result in increased erythropoietin transcription and therefore increased erythropoiesis. In addition, zinc is regarded as an important erythroid differentiation factor which progress maturation of erythrocytes. Due to the multifactorial role of zinc in erythropoiesis, including acting as a cofactor in the SMAD 2/3 complex, it may represent an inexpensive, and well tolerated ESA or additional micronutrient to test and supplement in MDS.
This descriptive, retrospective study describes zinc deficient patients with MDS, who underwent zinc supplementation, and their changes in hemoglobin (HGB) over a 12-month period in an academic outpatient hematology center in California, United States of America.
Objectives:
Primary endpoints: determine if zinc supplementation in zinc deficient MDS patients will improve HGB by at least 1.0 g.dL by 1, 3, 6, and 12 month time points.
Methods:
Eight adult patients with MDS were identified to have zinc deficiency (plasma zinc concentration <65 ug/dL). Patients were not started on ESAs or their treatments were not changed for the management of their MDS. Patients were supplemented with Zinc Sulfate 50 mg daily and their HGB was followed over 12 months.
A retrospective analysis for medical history and lab data at time of zinc deficiency diagnosis and at 1, 3, 6 and 12 month following initiation of zinc supplementation was completed. ANOVA, Wilcoxon rank-sum, and unpaired Students and t tests were performed to determine significant differences where p <0.05 is considered statistically significant.
Results:
Prior to supplementation, average HGB among patients was 8.01 g/dL, WBC was 3.65 g/dL, MCV was 97.6 fl, PLTS were 140g/dL. Following 1 month of supplementation average HGB was 8.5 g/dL (p = 0.5, n = 7), at 3 months average HGB was 9.27 g/dL (p = 0.087, n = 7), at 6 months average HGB was 10.3 g/dL (p= 0.006, n = 7), and at 12 months average HGB was 10.28 g/dL (p = 0.04, n= 7).
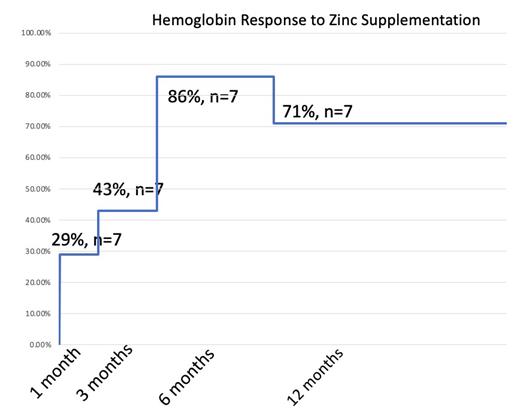
Primary endpoint of HGB improvement by at least 1.0 g/dL was reached in 29% of patients at 1 month, 43% of patients at 3 months, 86% of patients at 6 months and 71% of patients at 12 months. Resolution of anemia was reached in 1 patient (12.5%) of patients within 12 months.
Conclusions:
Zinc supplementation improved anemia in MDS patients with incremental improvement in HGB over a 1 year time frame. There was statistically significant improvement from baseline HGB at the 3, 6, and 12 month time markers. Further study is warranted to determine the specific mechanism of zinc deficiency in MDS and its mechanism as an erythrocyte stimulating agent as well as further study in non-zinc deficient patients, various zinc supplementation doses, monitoring, and toxicity.
Disclosures
Akhtari:Incyte: Speakers Bureau; SecuraBio: Speakers Bureau; Incyte: Speakers Bureau; PharmaEssentia: Speakers Bureau; CTI: Speakers Bureau; J&J: Speakers Bureau; Abbvie: Honoraria; Karyopharm: Speakers Bureau; JazzPharma: Speakers Bureau; Sobi: Honoraria; BMS: Membership on an entity's Board of Directors or advisory committees, Speakers Bureau.