Background:
Zinc is a necessary micronutrient used as a catalyst, structural element, and regulatory ion in many metabolic processes including erythropoiesis. Chronic anemia has an estimated prevalence of 5.6% within the US according to NHANES. Iron deficiency is considered the most common nutritional deficiency leading to anemia, but other nutritional deficiencies have been implicated in anemia including deficiencies of Vitamin A, B12, B6, C, D, and E, folate, Riboflavin, Copper, and Zinc. While zinc fortification has been studied in developing countries and has been associated with a decrease in the overall prevalence of anemia within the pediatric and maternal populations, there have been no studies in adults with chronic anemic in the developed world. The mechanism of Zinc in erythropoiesis is likely multifactorial with zinc acting as a catalyst in alpha-aminolevulinic acid dehydratase enzyme in iron metabolism and heme synthesis. Zinc's incorporation in the structure of the growth factor independent 1B (Gfi-1B) Zinc finger protein and GATA-1 Zinc finger transcription factor, which functions as a regulator in erythroid cell growth by modulating gene expression specific to erythroid series including the SMAD pathways and TGF-β signaling. Zinc's interaction in signaling cascades such as the growth hormone and insulin-like growth factor-1 pathways also result in increased erythropoietin transcription and therefore increased erythropoiesis. In addition, zinc is regarded as an important erythroid differentiation factor.
This descriptive, retrospective study describes zinc deficient patients with chronic anemia, who underwent zinc supplementation, and their changes in hemoglobin (HGB) over a 12-month period in an academic outpatient hematology center in California, United States of America.
Objectives:
Primary endpoints: determine the rate of anemia resolution over 12 months following supplementation of zinc in patients with zinc deficiency and chronic anemia.
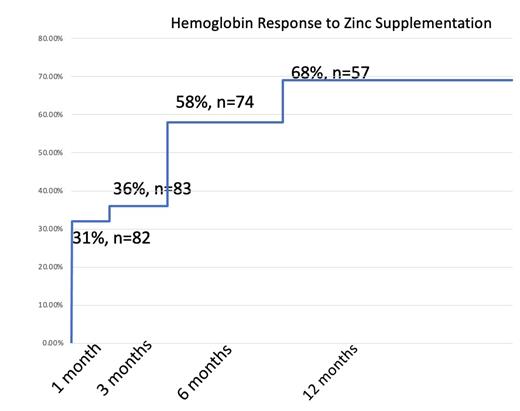
Secondary endpoints: determine if zinc supplementation in zinc deficient patients with chronic anemia will improve HGB by at least 1.0 g.dL by 1, 3, 6, and 12 month time points.
Methods:
Ninety Three patients with chronic anemia (HGB <12 for women, HGB <13 for men for >3 months) over the age of 18 were identified to have zinc deficiency (plasma zinc concentration <65 ug/dL) in an academic outpatient hematology center in California. Patients did not start new treatment for their anemia, including initiating new treatments for malignancy 3 months preceding, and within the data collection period. Patients were supplemented with Zinc Sulfate 50 mg daily and their HGB was followed over 12 months.
A retrospective analysis was completed for prior medical history, lab data, and HGB was tabulated at the time markers of 1, 3, 6, and 12 months following initiation of zinc supplementation. ANOVA, Wilcoxon rank-sum, and unpaired Students and t tests were performed to determine significant differences where p <0.05 is considered statistically significant.
Results
Prior to supplementation, average HGB among patients was 9.83 g/dL, WBC was 6.8 g/dL, MCV was 89 fl, PLTS were 211 g/dL. Following 1 month of supplementation average HGB was 10.46 g/dL (p = 0.015, n = 82), at 3 months average HGB was 10.75 g/dL (p = 0.0002, n = 83), at 6 months average HGB was 11.34 g/dL (p= 0.000000004, n = 74), and at 12 months average HGB was 11.48 g/dL (p = 0.00000002, n= 57).
Primary endpoint of anemia resolution was found in 39 of 93 patients (42%). Secondary endpoint of HGB improvement by at least 1.0 g/dL was reached in 31% of patients at 1 month, 36% of patients at 3 months, 58% of patients at 6 months and 68% of patients at 12 months.
Conclusions:
Zinc supplementation significantly improved HGB levels in patients with zinc deficiency and chronic anemia, with resolution of anemia in 42% of patients. There was statistically significant improvement from baseline HGB at the1, 3, 6, and 12 month time markers. Further study is warranted to determine the specific mechanism of zinc deficiency in anemia, as well as clinical studies determining zinc supplementation doses, monitoring, and toxicity.
Disclosures
Akhtari:Incyte: Speakers Bureau; SecuraBio: Speakers Bureau; Abbvie: Honoraria; J&J: Speakers Bureau; PharmaEssentia: Speakers Bureau; Karyopharm: Speakers Bureau; Sobi: Honoraria; JazzPharma: Speakers Bureau; CTI: Speakers Bureau; Incyte: Speakers Bureau; BMS: Membership on an entity's Board of Directors or advisory committees, Speakers Bureau.