Introduction
Delayed hemolysis transfusion reaction (DHTR) is a serious complication of sickle cell disease (SCD), accounting for 6% of all-cause mortality (Habibi Am J Hematol 2016). Although transfusion risk management has improved, the diagnosis of DHTR remains difficult and has long been underestimated. In fact, the main manifestation of DHTR in SCD patients is vaso-occlusive crisis (VOC), often associated with acute chest syndrome (ACS) and multiorgan failure, which makes transfusion attribution somewhat difficult. DHTR treatment, although not yet well codified, is based on immunoglobulins, corticoids or eculizumab in severe forms, depending on the team (Pirenne Blood 2018). Our team has proposed the use of a nomogram to guide diagnosis based on HbA decline (calculated by multiplying HbA and MCH) (Mekontso Dessap Am J Hematol 2016). However, this requires rapid quantification of hemoglobin fractions (often by HPLC) and a post-transfusion reference value, which is often lacking. Given the precise measurement of intravascular markers of hemolysis by spectroscopy that we have developed, we wondered if this could provide a simpler and faster diagnostic tool for DHTR. We performed a cross-sectional cohort study to test this hypothesis.
Methods
We performed intravascular hemolysis assays, including measurement of plasma hemoglobin, heme, methemoglobin, carboxyhemoglobin, hemopexin, and bilirubin, in a single run on thawed samples from patients in different cohorts. In short, the method combines a biochemical and mathematical multistep analysis (BET 18P3417). Cohorts of patients experiencing a DHTR were compared with patients in steady state and patients at the time of acute chest syndrome (ACS) in the ICU. Patients gave informed consent in accordance with French law (SCD transfu, Erythropedie, FCDREP and TASC cohorts). Two experts independently reviewed all DHTR cases to retrospectively confirm the diagnosis.
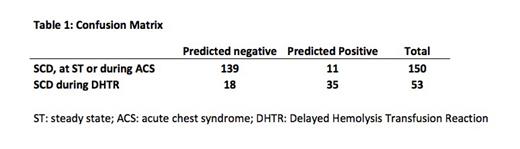
Logistic regression with k-fold cross-validation (3-fold) was applied to a dataset containing hemolytic biomarkers to predict the occurrence of DHTR using python3 and the sklearn library. The performance of the model was evaluated using various metrics such as accuracy and area under the ROC curve (AUC). Specificity, sensitivity, positive predictive value (PPV) and negative predictive value (NPV) were also calculated. The confusion matrix table shows the predictive performance of the model for the classification of DHTR occurrence.
Results
Samples and clinical data of 53 SCD patients with DHTR, Y patients at steady state, and Z patients during acute chest syndrome were analyzed and included in this study. During DHTR plasma Hemoglobin (47.8 vs 12.8 µM, p<0.0001), Heme (27.9 vs 3.2 µM, p<0.0001), Heme/Hemoglobin ratio (1.02 vs 0.4 ; p<0.0001), MetHb (11.7 vs 2.6 µM, p=0.0003) were significantly higher in DHTR whereas Hemopexin level was lower (1.53 µM vs 2.7 µM, p=0.03)
A diagnostic score was obtained by combining plasma levels of hemoglobin, heme, HbCO, MetHb, bilirubin, hemopexin. The AUC was 0.86 with a mean identical cross-validation. The accuracy and
precision of the score were 0.85 and 0.76, respectively. The specificity was 0.93, the sensitivity was 0.66, the positive predictive value (PPV) was 0.76 and the negative predictive value (NPV) was 0.88.
Conclusions
This score may help clinicians diagnose DHTR early and then provide appropriate treatment. However, a prospective study is needed to validate the score.
Disclosures
Bartolucci:Novartis: Consultancy, Membership on an entity's Board of Directors or advisory committees; Addmedica: Consultancy, Membership on an entity's Board of Directors or advisory committees; INNOVHEM: Current equity holder in private company; Bluebird: Consultancy; Roche: Consultancy; Emmaus: Consultancy; GBT: Consultancy; Jazz Pharma: Consultancy. Habibi:Add Medica: Honoraria; GBT: Consultancy; novartis: Consultancy. Perkins:INNOVHEM: Current Employment. Bencheikh:INNOVHEM: Current Employment. Cambot:INNOVHEM: Current equity holder in private company. Rapsode:INNOVHEM: Other: INtern.