Introduction:
Extramedullary AML (EM AML) is characterized by the presence of leukemic cells in organs or tissues outside of the blood or bone marrow and remains a therapeutic challenge with dismal response rates to standard chemotherapy and short overall survival. Gemtuzumab Ozogamicin (GO) is an anti-CD33 antibody drug conjugate currently approved for single-agent therapy or in combination regimens for CD33+ relapsed/refractory (R/R) or newly diagnosed (ND) AML, respectively. Although GO is an active drug in AML, the optimal employment of this agent for treatment of EM AML at initial diagnosis, or at relapse, remains to be defined.
Methods:
A retrospective chart review on outcomes of GO in adults with EM AML at our institute from 2000 to 2023 was performed. Variables collected included age, sex, type of myeloid malignancy, and European LeukemiaNet (ELN) risk, bone marrow, peripheral blood and radiographic involvement, Response Evaluation Criteria in Solid Tumors (RECIST), prior lines of treatments, including stem cell transplant, next-generation sequencing (NGS), cytogenetics, mutational profile, and overall survival (OS). We screened patients with a diagnosis of EM AML who received GO at any point during their EM AML therapy. Patients with only CNS involvement as the extramedullary site were excluded. The aims of this study were to characterize responses and potential clinical benefit of EM AML to GO based regimens in the upfront and R/R settings.
Results:
32 patients(pts) with EM AML received GO: 8 (25%) were ND and received GO-based induction regimen, while 22 (68.75%) received GO for R/R AML, and 2 (6.25%) patients received GO both at initial diagnosis and relapse. The median age of patient with ND EM AML was 66 years (y) (range: 28-84), and 60.5 (43-73) y in R/R EM AML. Twenty-four (75%) were men. One (12.5%) ND EM AML and 5 (10.1%) R/R EM AML had >1 extramedullary sites involved. The most frequent sites of EM-AML were skin (25%), lymph nodes (25%), soft tissues (25%), solid organs (25%), and visceral body cavity fluids (12.5%). Based on ELN 2022 risk group criteria, 17 patients were adverse, 8 with intermediate, and 3 with favorable risk. Next-Generation Sequencing (NGS) available for 21 patients identified mutations in TP53 (14.28%), FLT3 (19.04%), NPM1 (19.04%), KMT2A (19.04%), and ASXL1 (19.04%) as the most common mutations. Normal karyotype was noted in 6 (25%) R/R EM AML, while complex karyotype was present in 1 (12.5%) ND EM AML and 10 (41.67%) of R/R EM AML. While none of the ND pts had prior myeloid malignancy, R/R pts had prior AML (15), MDS (2), MDS-EB (1), CMML (1), therapy-related AML (1), myelofibrosis (1), and myeloid sarcoma (1). Concomitant bone marrow involvement at identification of EM AML was present in most ND cases (7/8, 88%) and R/R settings (16/23, 69.56%) (Table 1).
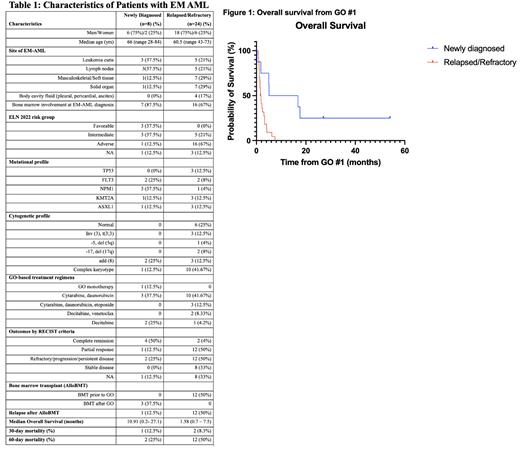
Combination 7+3 plus GO was most frequently administered for ND EM AML (3/8) and R/R EM AML (8/24, 33%). Eleven patients had measurable radiologic findings prior to and following GO. Median OS was 10.91 months (m) (range: 0.2 to 27.1) for ND EM AML and 1.58 m (range: 0.7 to 7.5) for R/R EM-AML (Figure 1). 3 of 8 newly diagnosed EM-AML underwent allogeneic bone marrow transplant (AlloBMT) after GO based induction, followed by relapsed EM AML in 1 patient. Despite 50 percent of R/R EM AML pts undergoing prior AlloBMT, none had a durable disease-free response and received GO after relapse.
Based on RECIST criteria, 7 of 11 (63.63%) pts achieved a response including 1 Complete Response (disappearance of all target lesions), 6 Partial Response (at least a 30% decrease in the sum of target lesions), and 2 Stable Disease (neither sufficient shrinkage nor increase). The 30-day mortality for ND and R/R were 12.5% and 8.33%, respectively. The 60-day mortality for ND and R/R were 25% and 50%, respectively.
Conclusion:
Our real-world single-institute experience in patients with EM AML suggests that GO combined with chemotherapy in the upfront setting results in promising outcomes with high overall response rates (CR 50%, PR 13%) by RECIST criteria, durable remissions, and prolonged overall survival in largely favorable and intermediate risk AML. However, despite high partial response rates (4% CR, 50% PR) following GO-based therapy in R/R EM AML, survival was less than 2 months likely due to the high proportion of patients in this cohort with poor risk disease. Further investigations and larger studies are warranted to establish the optimal use of GO in EM AML.
Disclosures
Griffiths:Takeda Oncology: Consultancy; Astex Pharmaceuticals: Research Funding; Celldex Therapeutics: Research Funding; Physicians Educational Resource: Honoraria; MDS International Foundation: Honoraria; American Society of Hematology: Honoraria; Taiho Oncology: Consultancy; Alexion Pharmaceuticals: Consultancy, Research Funding; Apellis Pharmaceuticals: Consultancy, Research Funding; Artis Ventures: Membership on an entity's Board of Directors or advisory committees; Genentech, Inc.: Consultancy, Research Funding; S. Karger Publishing: Honoraria; Picnic Health: Membership on an entity's Board of Directors or advisory committees; Bristol Myers Squibb: Consultancy, Research Funding; MediCom Worldwide, Inc.: Honoraria; Abbvie: Consultancy; Novartis: Consultancy, Research Funding; CTI Biopharma: Consultancy; AAMDSIF: Honoraria; Blueprint Medicines, Inc: Research Funding; NextCure, Inc: Research Funding; Partner Therapeutics: Consultancy; AstraZeneca Rare Disease: Consultancy, Research Funding; Medscape: Honoraria; Vera and Joseph Dresner Foundation: Membership on an entity's Board of Directors or advisory committees. Thompson:Bristol Myers Squibb: Research Funding; Novartis: Research Funding. Wang:Gilead: Consultancy; BMS: Consultancy; GlaxoSmithKline: Consultancy; Jazz: Consultancy; Kite: Consultancy, Speakers Bureau; Novartis: Consultancy, Speakers Bureau; Pfizer: Consultancy, Speakers Bureau; PharmaEssentia: Consultancy; Takeda: Consultancy; Dava oncology: Speakers Bureau; Kura Oncology: Speakers Bureau; Astellas: Consultancy, Speakers Bureau; Abbvie: Consultancy. Przespolewski:Jazz Pharmaceuticals: Research Funding.