BACKGROUND:
Hypereosinophilia (HE) (>1,5x109/L eosinophils in peripheral blood (pb)) is an occasional cause of consultation in hematology. The etiologies are varied, the diagnostic process is wide and may include other specialties. In addition, the absence of prospective studies and large cohorts makes the diagnosis and management of these patients very challenging.
OBJECTIVE:
To describe the diagnostic process of a series of patients with HE evaluated in our center between 2013-2023.
METHODS:
Clinical-biological characteristics and diagnostic evaluations performed were registered. Clonal HE and reactive HE were compared using Fisher exact test and Mann-Whitney U.
RESULTS:
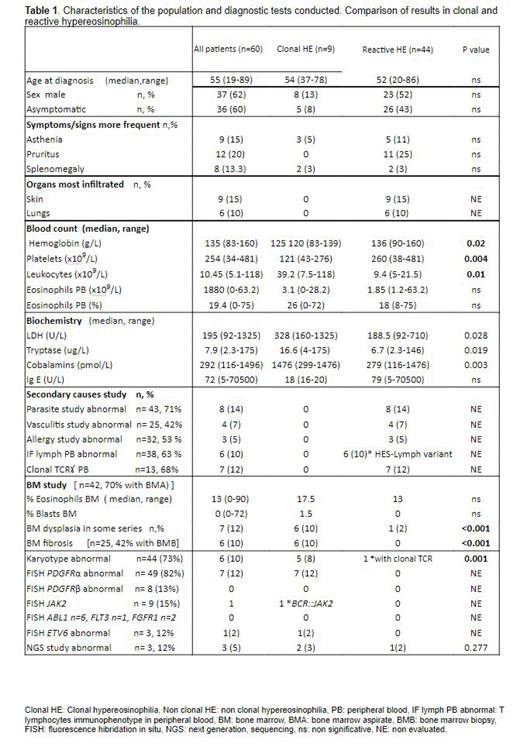
Sixty patients with HE (n=59) or tyrosine-kinase fusion genes eosinophilia related (n=1) were selected out of 109 patients with eosinophilia (>0.5x109/L and <1,5x109/L eosinophils in pb), detected in the last decade in our center. Characteristics of the study population and diagnostic tests conducted are summarized in Table 1. Interestingly, bone marrow data with cytogenetic study was made in up to 70% of patients. Despite extensive analysis of the PDGFRα rearrangement by FISH, other fusion genes were less evaluated and NGS studies were little used. Diagnostic studies for exclusion of secondary causes were carried out in more than 40% of patients ( Table 1).
Fifteen percent of HE attended were a primary clonal entity whereas 73% were secondary/reactive: 5% due to allergy, 7% to asthma, 2% to drugs 2%,13% parasites, 25% other, 2% lymphoma, 8% were hypereosinophilic syndrome (HES)-lymphocytic variants, 8% HES-overlap, 3% associated hematologic neoplasm. Twelve of the patients couldn't be classified, which evidences the diagnostic complexity. Among the variables used in differential diagnosis of HE, those significantly associated with clonal HE compared to reactive HE in our series were: BM dysplasia (85.7% vs 14.3%, p<0.001), BM fibrosis (100% vs 0, p<0.001), abnormal karyotype (83.3% vs 16.7%, p<0.001), LDH (328 vs 188.5 U/L, p=0.028), cobalamins (>1476 vs 279 pmol/L, p=0.003), tryptase (16.6 vs 6.7 ug/L, p=0.019) and leucocytes (39.2 vs 10.2 x109/L, p=0.014). Other analytical parameters were significantly lower in clonal vs. reactive HE: hemoglobin (125 vs. 136 g/L, p=0.020) and platelets (121 vs. 260x109/L, p=0.004). It was noteworthy that the number of eosinophils did not help always to distinguish clonal vs. non-clonal entities.
Patients diagnosed with a primary clonal HE were 9 (15%): 7 patients with rearrangement of PDGFRα, 1 patient with ETV6 and 1 with BCR::JAK2. The group of PDGFRα patients, all asymptomatic, showed a median diagnostic delay of 45 months (5.1-103) since the first hematological evaluation. Four out of 7 patients with PDGFRα showed hematological and cytogenetic response after a median of 3.37 months (0.95-78.2) from imatinib initiation. Two patients started imatinib recently (not evaluable) and one patient presented clonal evolution and transformation to the blastic phase after 10 years of disease. He has been treated with imatinib 400mg achieving hematological response. The overall survival of this subgroup was 80% CI 95% (62.8-97.2), with a median follow-up of 25.8 months.
One patient with ETV6 rearrangement presented as acute leukemia and died after few days from diagnosis and the patient with BCR::JAK2 fusion gen also presented with acute leukemia is in complete remission after being treated with intensive induction chemotherapy and allogeneic hematopoietic cell transplantation.
CONCLUSIONS:
HE requires a wide diagnostic evaluation with a multidisciplinary approach and the development of working groups in order to avoid these inconclusive diagnosis (12% in our series) and the missing diagnostic tests. Our study suggests the association between elevated LDH, cobalamins and tryptase and clonal cause of HE, as well as a higher rate of fibrosis, medullary dysplasia and altered karyotypes in these patients, indicating the need to include these evaluations in the diagnostic process. The group of PDGFRα patients had good prognosis with imatinib, but one patient presented clonal evolution and blastic transformation, showing the clinical challenge of these entities.
Disclosures
Sureda Balari:Kite: Consultancy, Speakers Bureau; Takeda: Consultancy, Honoraria, Speakers Bureau; MSD: Research Funding.