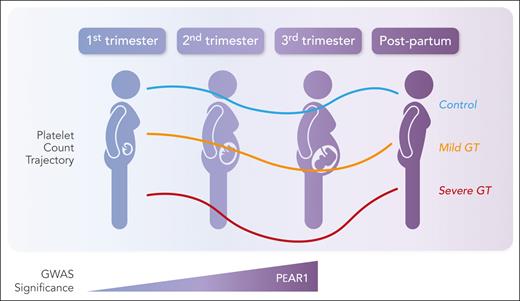
In this issue of Blood, Yang et al provide insight into the biological mechanisms underlying gestational thrombocytopenia (GT).1 Using sequencing data from noninvasive prenatal testing from 100 186 pregnancies in 2 independent cohorts, they conducted large-scale genome-wide association studies (GWASs) on platelet counts at 5 time points in pregnancy (first, second, and third trimesters, time of delivery, and postpartum). Their analysis defined 138 genome-wide loci with 2 candidate variants in platelet endothelial aggregation receptor 1 (PEAR1) and CBL associated with a significant change in platelet count during pregnancy; more important, PEAR1 was associated with a faster decline in platelets during pregnancy, especially in the second and third trimesters (see figure). A separate analysis of patients with GT and severe GT also linked PEAR1 variants to severity of thrombocytopenia. This study provides the first analysis of the genetic basis of GT linking PEAR1 directly to the decline in platelet count with a more pronounced thrombocytopenia in those pregnancies with this variant.
Expression of PEAR1 variants correlates with platelet counts during pregnancy. Professional illustration by Somersault18:24.
Expression of PEAR1 variants correlates with platelet counts during pregnancy. Professional illustration by Somersault18:24.
GT occurs in 5% to 10% of pregnant women during the peripartum period and accounts for most cases of thrombocytopenia in pregnancy.2 Starting in the second trimester, the platelet count begins to trend downward, plateauing before delivery. The underlying cause of this slow downward trend in platelet count has yet to be understood, having previously been attributed to hemodilution resulting from increased plasma volume during pregnancy and/or possibly also increased platelet clearance.3 A subset of patients will experience an even more rapid decline, with platelet counts of <100 × 109/L. To date, there are no biomarkers to predict which patients will develop GT and which are at risk of a more critical level of thrombocytopenia. Although most patients maintain an adequate platelet count for delivery, patients with severe GT may not respond to intravenous immune globulin or corticosteroids, complicating epidural anesthesia and delivery plans.
PEAR1 is not a newcomer to platelet biology, having previously been described as a transmembrane receptor involved in platelet activation and megakaryocytopoesis.4 With direct links to platelet functionality, PEAR1 is associated with increased platelet aggregation in response to known platelet agonists, including adenosine diphosphate, epinephrine, and collagen, with implications for cardiovascular disease.5 Although no studies have linked PEAR1 with thrombocytopenia, the authors of this study provide correlative data on mean platelet volume in pregnancy as a marker of increased platelet activation and turnover. What is not known, however, is if this decline in platelet count is a direct consequence of this platelet hyperactivation and aggregation, leading to thrombotic consumption.
Further correlation between the GWAS analysis and platelet activation was the identification of CBL loci with direct links to megakaryocytopoiesis and platelet production. CBL is an E3 ubiquitin ligase that acts as a negative regulator of megakaryocytopoeisis.6 In this article, changes in CBL were associated with a downregulation of expression, accounting for a reciprocal change in platelet count in the first trimester, with platelet counts remaining higher and not significantly associated with thrombocytopenia in later trimesters when the platelet count declined.
With a direct link to the pathogenesis of GT, the authors conducted GWASs on 11 138 GT cases and 906 cases of severe GT. In patients with known GT or severe GT, they identified 2 significant loci in PEAR1 and tubulin β1 class VI (TUBB1). TUBB1 has a known role in the platelet cytoskeleton, localizing to microtubules and the microtubule ring of proplatelets, and has been shown to be associated with thrombocytopenia.7 What underlies the development of thrombocytopenia in patients with this locus during pregnancy remains to be understood. As further validation of the role of PEAR1 in GT and severe GT, the locus was also identified in this subset analysis of patients with GT.
The implication of this work provides a biological mechanism to explain the changes in platelet count that occur in pregnancy. The identification of 2 loci with ties to platelet activation broadens our understanding of the molecular mechanisms driving GT and provides targets for further research to better understand how pregnancy impacts platelet production. Both PEAR1 and CBL are associated with platelet activation. Beyond hemodilution, another proposed mechanism for development of GT is placental blood flow dynamics with increased platelet activation and subsequent consumption within the vasculature.3 Given that both CBL and PEAR1 are associated with platelet activation and turnover, a potential hypothesis for why patients with this variant develop GT may be increased propensity for platelet activation and consumption in the placental vasculature, leading to the decline in platelet count. This could also explain why as the placenta grows in the second and third trimesters, the thrombocytopenia may become more pronounced. Dedicated studies of platelet activation in pregnancy and in women who develop GT will provide mechanistic evidence of baseline platelet activation as a potential regulator of thrombocytopenia.
A second potential mechanism revealed suggests that GT is a consequence of an underlying abnormality in megakaryocytopoiesis and platelet production. TUBB1 has previously been associated with thrombocytopenia, being mechanistically linked to abnormalities in the platelet cytoskeleton, leading to impaired proplatelet formation. What remains to be defined is how variants of TUBB1 disrupt megakaryocytopoiesis in pregnancy, leading to stunting of platelet production. Validation of cytoskeletal abnormalities in the platelets of patients with TUBB1-associated GT would provide mechanistic evidence of this association.
Given that an area of unmet need in GT remains the lack of biomarkers that can predict which patients are at risk of developing significant thrombocytopenia, this GWAS provides a foundation for laboratory diagnostics. Next steps involving identification of these variants directly in patients who develop GT will provide a necessary proof of concept. Being able to distinguish cases of mild GT from severe GT will enable clinicians to better care for patients at risk of hemostatic abnormalities, improving outcomes for both mother and child.
Conflict-of-interest disclosure: E.M.B. declares no competing financial interests.