Abstract
B-cell prolymphocytic leukemia (B-PLL) is an aggressive disorder of mature B cells with distinct clinical and pathologic features. To determine the incidence of abnormalities of p53, we analyzed 19 cases of B-PLL by DNA blot to assess loss of heterozygosity (LOH) at 17p13.3, by immunocytochemistry to assess p53 expression, and by direct DNA sequencing of polymerase chain reaction-amplified exons 5 to 9 of the p53 gene. LOH was detected in 10 of 19 (53%) cases, p53 expression was detected in 8 of 17 (47%), and p53 mutations were detected in 10 of 19 (53%) cases. The pattern of mutations was distinct from that observed in other B-cell malignancies. Six cases exhibited missense mutations; 4 were transversions and 2 were transitions. The G:C → A:T transition at cathepsin G dinucleotides commonly reported in p53 mutations in chronic lymphocytic leukemia (CLL) and other hematologic malignancies was observed in only 1 case of B-PLL. Three cases exhibited deletions (ranging from 3 to 35 bp in length) and one case exhibited a 2-bp insertion. In 1 case, a 27-bp deletion resulted in the expression of a p53 protein lacking 9 amino acids from the DNA binding region. All samples with p53 mutation showed loss of germline p53 sequences. However, 3 of 10 showed no LOH by Southern blot, indicating a localized deletion around the p53 locus at 17p13.1. Five of the 10 cases with p53 mutation exhibited detectable p53 expression, including 4 cases with p53 missense mutation and 1 case with deletion. Two of 7 cases with no detectable mutation of p53 nevertheless overexpressed p53. Therefore, there was no correlation between protein expression and p53 mutation in B-PLL. Our data indicate that the overall abnormalities of p53 occurred in 14 of 19 (75%) cases of B-PLL. The frequency of p53 mutation (53%) in B-PLL is the highest reported in B-cell malignancies and may be responsible for the frequent resistance to therapy of this disease. In addition, the pattern of p53 mutation was different from that observed in CLL and other hematologic malignancies and may indicate that a distinct pathogenic mechanism operates in B-PLL.
B-CELL PROLYMPHOCYTIC leukemia (B-PLL) is a distinct leukemia of mature B cells1,2 characterized by a high white blood cell count and splenomegaly without significant lymphadenopathy. The diagnosis is made by examination of peripheral blood film in which the predominant cells are prolymphocytes; a minimum of 55% of these cells must be found at presentation of the disease for this diagnosis to be established.3
The membrane phenotype of the prolymphocyte is quite distinct from that of chronic lymphocytic leukemia (CLL) but has similarities to that of other B-cell leukemias. They express strong SmIg that is IgM or IgM/IgD, pan-B–cell antigens, FMC7, and CD22 and they are usually negative for CD23. Several chromosome abnormalities have been described, including the t(11; 14) (q13; q32), although these are not consistent. Additional abnormalities include trisomy of chromosome 12 and deletions of chromosomes 1, 3, and 6q.4 However, little is known about the underlying molecular mechanisms of this rare disease that is frequently associated with resistance to therapy.5
The p53 gene, which is located on chromosome 17p13.1, functions as a specific transcriptional activator6 of genes controlling the G1 checkpoint of the cell cycle, ie, mdm2,7p21 (waf-1),8 and also appears to control the expression of certain genes involved in the control of programmed cell death or apoptosis, such as Bax and bcl-2.9 10
Mutations of the p53 gene are the most common genetic alteration in human cancer11,12 yet described. Most are missense mutations that change the amino acid composition of the protein. Such alterations can modify the conformation and stability of the p53 protein and may result in the alteration of p53's transcriptional activity as well as in an increase of the half life of the protein, allowing its detection by immunocytochemistry. In many cases, mutation of one allele is associated with deletion of the other allele, leading to inactivation of both normal p53 alleles. Most p53 mutations are clustered in four regions of the protein (residues 117-142, 171-181, 234-258, and 270-286) that are highly conserved among several different species.12
Mutations of the p53 gene have been documented in a wide variety of hematologic malignancies, including Burkitt's lymphomas (41%), adult T-cell leukemia (24%), chronic myeloid leukemia in blast crisis (20% to 30%), chronic lymphocytic leukemia (12%), multiple myeloma (10%), and high-grade non-Hodgkin's lymphomas (NHL; 30%), whereas low-grade NHL rarely have alterations of p53 unless they are transformed (reviewed in Imamura et al13 and Newcomb14 ).
We present here an analysis of 17p allele loss, p53 mutation, and p53 protein expression in 19 cases of B-PLL. Molecular analysis showed a high incidence of p53 mutations, loss of heterozygosity (LOH) at 17p, and p53 expression. Moreover, the pattern of p53 mutations was different from those reported in other lymphoid leukemias and may indicate a distinct pathogenic mechanism in this disease.
MATERIALS AND METHODS
Patients
Nineteen cases of B-PLL referred to our institution from 1990 to 1995 for diagnosis were selected for this study. Peripheral blood films for morphology analysis and immunologic markers were available in all of the cases studied. Diagnosis was made on the basis of morphologic appearances and detailed immunophenotypic analysis by flow cytometry using the criteria established by the French-American-British group.15 The main clinical and laboratory data are shown in Table 1.
Immunocytochemistry
Immunocytochemistry was performed on cytospin preparations of peripheral blood or spleen mononuclear cell suspensions using the monoclonal antibodies (MoAbs) PAb1801 (Oncogene Science Inc, Cambridge, MA) and DO-1 (Santa Cruz Biotechnology Inc, Santa Cruz, CA) that recognize the amino acids 1-45 and 37-72, respectively, at the N-terminal epitope of mutant and wild-type p53 protein; an indirect immunoperoxidase sandwich technique was also used.16 Briefly, after fixation for 10 minutes in acetone, cytospins were incubated for 30 minutes with PAb1801 or DO-1 in phosphate-buffered saline (PBS), followed by a peroxidase-conjugated goat antimouse antibody for 30 minutes. Peroxidase antiperoxidase complexes were added to increase the staining intensity. The peroxidase reaction was performed by incubating for 10 minutes in a developing solution containing diaminobenzidine tetrahydrochloride 0.6 mg/mL (Dako, Glostrup, Denmark) in PBS with 0.1% of hydrogen peroxide. Cells were counterstained with Haematoxylin Gill (H.D. Supplies, Ayelsbury, Bucks, UK). The proportion of cells positively immunostained was evaluated by light microscopy under high power (magnification × 1,000) and at least 500 cells were scored from each sample. Positive control for p53 expression was the CEM T-cell line, which carries a p53 mutation and expresses abundant p53. Peripheral blood from a healthy donor was used as a negative control.
Western Blot Analysis
Cells were lysed in sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) sample buffer (Sigma, St Louis, MO) and heated at 95°C for 5 minutes. Proteins were resolved on a SDS-polyacrylamide (13%) gel and transferred to nitrocellulose using a Transphor apparatus (Hoefer Pharmacia Biotech, San Francisco, CA) according to the instructions of the manufacturer. Filters were incubated in blotting solution (PBS, 0.05% Tween 20, 3% bovine serum albumin fraction V) for at least 1 hour at room temperature and then incubated with primary antibodies PAb1801, DO-1, or the antibody PAb 421 (Santa Cruz Biotechnology Inc) that recognize a C-terminal epitope of human p53 protein17 for 1 hour at room temperature. Filters were washed three times in PBS, 0.05% Tween 20 and incubated with antimouse horseradish peroxidase conjugate for 1 hour at room temperature. Bound antibody was detected using the ECL Western blotting system (Amersham, Amersham, Bucks, UK) as described by the manufacturer.
LOH Analysis
Conventional DNA blotting was performed as described.18 Briefly, high molecular weight DNA was extracted from cryopreserved mononuclear fraction of peripheral blood by routine methods. Ten micrograms of DNA was digested to completion with Pst I restriction enzyme (Promega, Madison, WI). DNA fragments were electrophoresed in 0.7% agarose and transferred to positively charge nylon membranes (Hybond-N+; Amersham) by overnight capillary transfer in alkali. Filters were probed sequentially with p144-D619 (a D17S34 probe released as a 5.1-kb EcoRI insert) and pYNZ2220 (a D17S30 probe released as a 1.7-kb BamHI insert). Both probes were obtained from the American Tissue Collection Culture (Rockville, MD) and were labeled with α-32P dCTP by the method of oligolabeling to a specific activity of greater than 3 × 109 dpm/μg DNA.
Single-Strand Conformational Polymorphism (SSCP) Analysis of Exons 2-4 and 10-11
To screen for p53 mutations outside the highly conserved region of p53, SSCP was used to analyze exons 2, 3, 4, 10, and 11. Primers for SSCP were obtained from Clontech (Human Amplimer Panel; Clontech, Palo Alto, CA). SSCP reactions were performed in a volume of 10 μL and contained 10 mmol/L Tris-HCl, pH 8.3, 50 mmol/L KCl, 1.5 mmol/L MgCl2 , 0.01% gelatine, 200 ng of genomic DNA, 200 μmol/L of each deoxyribonucleoside triphosphate (dNTP), 2.5 pmol each primer, 0.1 μL of 10 μCi/μL [α-32P] dCTP, and 0.2 U of Taq DNA polymerase. Thirty-five cycles of amplification were performed of 95°C for 30 seconds, 66°C for 45 seconds, and 72°C for 90 seconds. Samples were diluted twofold in formamide dye loading buffer, heated to 95°C for 5 minutes, chilled on ice, and then resolved in 1× TBE on nondenaturing 6% polyacrylamide gels with or without 10% glycerol. Dried gels were exposed to x-ray film for 2 to 24 hours. For each exon, control DNA containing a known missense mutation was included in the analysis.
p53 Sequencing
Sequencing of exons 5 to 9 of p53 was performed by polymerase chain reaction (PCR) amplification followed by direct automated sequencing of double-stranded DNA using termination nucleotides tagged with fluorescent dyes.
PCR amplification.PCR was performed using 200 ng of DNA, 1.5 mg MgCl2, 200 μmol/L of each dNTPs, 1 μmol/L of each primer, 0.4 μL of Taq polymerase enzyme (Promega), and 5 μL of 10× Taq buffer (Promega) in a final volume of 50 μL. The cycling profile comprised 30 cycles of denaturation (92°C for 45 seconds), annealing (61°C for exons 5, 8, and 9 and 62°C for exons 6 and 7 for 45 seconds), and extension (72°C for 45 seconds). PCR amplification was performed in a Biometra (Goettingen, Germany) thermal cycler.
DNA nucleotide primers.The primers were obtained from Oswell (University of Edinburgh, Edinburgh, UK) and used for PCR and DNA sequencing. These primers were designed using the OLIGO 4.1 program to anneal to intronic sequences immediately flanking the respective exons. Their sequences were as follows: exon 5 sense strand, 5′ ACTTGTGCCCTGACTTTCAACT 3′; exon 5 antisense strand, 5′ CAATCAGTGAGGAATCAGAGGC 3′; exon 6 sense strand, 5′ TCAGATAGCGATGGTGAGCAG 3′; exon 6 antisense strand, 5′ GCCACTGACAACCACCCTTA 3′; exon 7 sense strand, 5′ GCGACAGAGCGAGATTCCA 3′; exon 7 antisense strand, 5′ GAAATCGGTAAGAGGTGGGC 3′; exon 8 sense strand, 5′ GGAGTAGATGGAGCCTGGTTT 3′; exon 8 antisense strand, 5′ GGTGATGAAAAGTGAATCTGAGGC 3′; exon 9 sense strand, 5′ GGAGACCAAGGGTGCAGTTAT 3′; exon 9 antisense strand, 5′ GTTAGTTAGCTACAACCAGGAGCC 3′.
Direct DNA sequencing of PCR products.PCR products were first purified using phenol/chloroform (1:1) extraction and precipitation with isopropanol (100%) and 7.5 mol/L ammonium acetate. The same primers used for generating the PCR products were also used for the sequencing reactions. Other reagents were supplied in the ReadyReaction Taq DyeDeoxy termination sequencing kit (Applied Biosystems [ABI], Foster City, CA). Both strands were sequenced for each exon. The ABI protocol was followed for the cycle sequencing of PCR products with slight modifications. One microliter of the purified PCR product (approximately 40 ng) was used and the primer concentration was 10 pmol/μL. Unincorporated nucleotides were removed from the extended products using two phenol/chloroform steps followed by an ethanol precipitation step. Samples were loaded on a 7% denaturating polyacrylamide gel, and the electrophoresis was performed on a Model 373A DNA Sequencer (ABI). All PCR products were sequenced in both orientations. Because of the possibility of Taq polymerase DNA incorporation errors, PCR products of the mutated cases were sequenced from two separate PCR reactions. Identical results were obtained in all cases.
RESULTS
To determine the frequency and the nature of the abnormalities of p53 in B-PLL, we undertoook DNA blotting to assess LOH, direct DNA sequencing of p53 exons 5 through 9, and immunocytochemistry. The results have been summarized in Table 2.
LOH Analysis
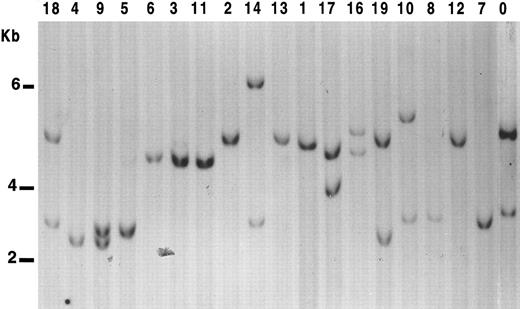
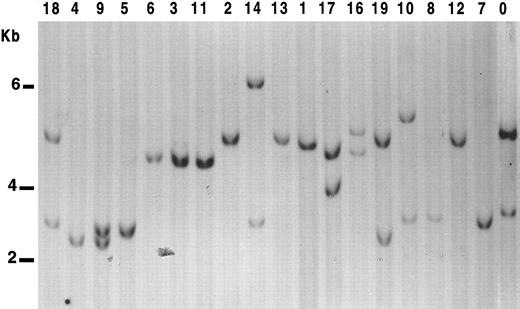
We tested allelic deletion at 17p13 using p144-D6 and pYNZ22, two highly informative polymorphic probes mapping near to p53 locus. Using both probes, a constitutive loss can be assessed within 95% confidence limits when both probes show a loss of one 17p allele, even when corresponding normal tissue is not available for comparison.11 Results are shown in Fig 1. In 10 of 19 (53%) cases, only one band was detected with both probes (Table 2).
LOH analysis at 17p13.3 in B-PLL cases. Genomic DNA from the samples studied were digested with Pst I, subjected to Southern blot, and probed with the clone p144-D6. Cases were scored positive for LOH when displaying only one allelic band with both probes. Nine of the B-PLL cases show only one band with p144-D6. However, in case no. 8, two bands were observed with pYNZ22 probe. Lane 0, control DNA from a normal sample.
LOH analysis at 17p13.3 in B-PLL cases. Genomic DNA from the samples studied were digested with Pst I, subjected to Southern blot, and probed with the clone p144-D6. Cases were scored positive for LOH when displaying only one allelic band with both probes. Nine of the B-PLL cases show only one band with p144-D6. However, in case no. 8, two bands were observed with pYNZ22 probe. Lane 0, control DNA from a normal sample.
Sequencing Analysis of p53 Gene
Nineteen cases of B-PLL were analyzed for the presence of mutations in the highly conserved region of p53 (exons 5 through 9) by automated cycle sequencing of PCR products. Given the relatively high rate of base misincorporation associated with PCR,11 all mutations found were confirmed by performing a second PCR reaction and resequencing.
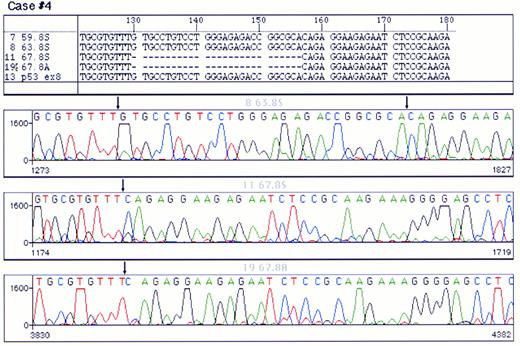
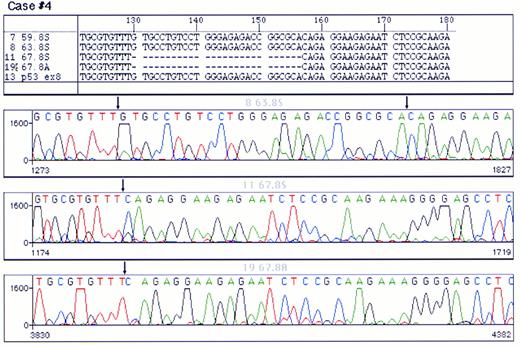
Mutations were detected in 10 cases (Table 2); 4 were localized in exon 8 and 2 in each of exons 5, 6, and 7, respectively. Examples of p53 mutations are shown in Fig 2. The following types of mutations were found: 6 cases of missense mutations, 3 cases of deletions (1 with a 5-bp deletion, 1 with a 27-bp deletion, and 1 with a 35-bp deletion) and 1 case with 2-bp insertion. Among the base substitution mutations, 4 were transversions (change of a pyrimidine to another pyrimidine or a purine to another purine) and 2 were transitions (change of a pyrimidine to a purine or vice versa). As shown in Table 2, the mutations in the cases with p53 deletions or insertions were localized in the central part of the molecule (3 in exon 8 and 1 in exon 7), whereas the cases with missense mutation were scattered in exons 5 through 8. Only in 1 case did the mutation fall in a hot spot nucleotide (Arg248). Analysis of the chromatograms at the site of the mutation showed that, in all samples with altered sequences, there was no additional signal corresponding to the normal sequence, indicating that, in B-PLL, all p53 mutations were accompanied by the loss of the other allele.
Analysis of a p53 mutation by PCR direct sequencing using fluorescent termination nucleotides. A chromatogram of control DNA is shown in (A). (B) and (C) correspond to chromatograms of the PCR product of exon 8 of p53 from a mutated case sequenced from both ends. A single base pair substitution (G → T) was detected in this case. Arrows point to the site of mutation. Note that the signal corresponding to the germline nuleotide could not be detected.
Analysis of a p53 mutation by PCR direct sequencing using fluorescent termination nucleotides. A chromatogram of control DNA is shown in (A). (B) and (C) correspond to chromatograms of the PCR product of exon 8 of p53 from a mutated case sequenced from both ends. A single base pair substitution (G → T) was detected in this case. Arrows point to the site of mutation. Note that the signal corresponding to the germline nuleotide could not be detected.
SSCP Analysis
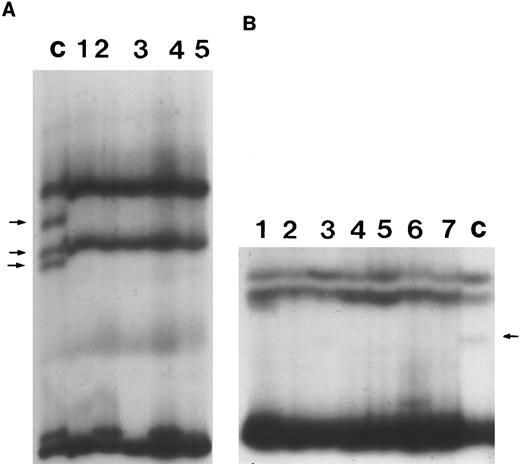
SSCP analysis was performed to screen for mutations in exons 2, 3, 4, 10, and 11 in those cases in which direct DNA sequencing in the highly conserved region of p53 gene did not show any mutations. No altered migration was observed in any of these cases (Fig 3).
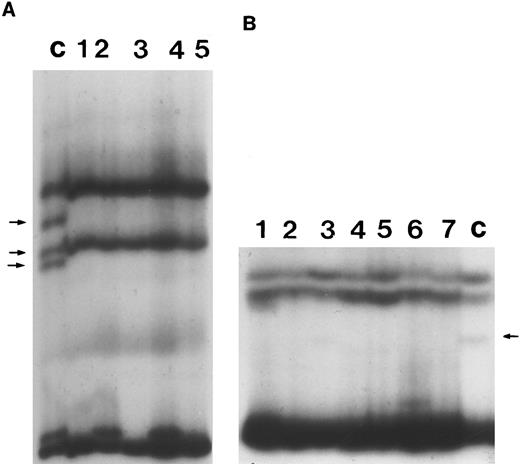
Representative SSCP analysis of genomic DNA from B-PLL showing absence of mutations in exons 4 and 10 of p53. SSCP was performed as described in the Materials and Methods. Abnormally migrating conformers in control DNAs are indicated with arrows. (A) SSCP of exon 4. Lane C, control DNA with known missense mutation at codon 102 of p53. Lanes 1 through 5, DNA from individual B-PLL cases. (B) SSCP of exon 10. Lane C, control DNA with a known missense mutation at codon 336 of p53. Lanes 1 through 7, DNA from individual B-PLL cases.
Representative SSCP analysis of genomic DNA from B-PLL showing absence of mutations in exons 4 and 10 of p53. SSCP was performed as described in the Materials and Methods. Abnormally migrating conformers in control DNAs are indicated with arrows. (A) SSCP of exon 4. Lane C, control DNA with known missense mutation at codon 102 of p53. Lanes 1 through 5, DNA from individual B-PLL cases. (B) SSCP of exon 10. Lane C, control DNA with a known missense mutation at codon 336 of p53. Lanes 1 through 7, DNA from individual B-PLL cases.
Expression of p53 Protein
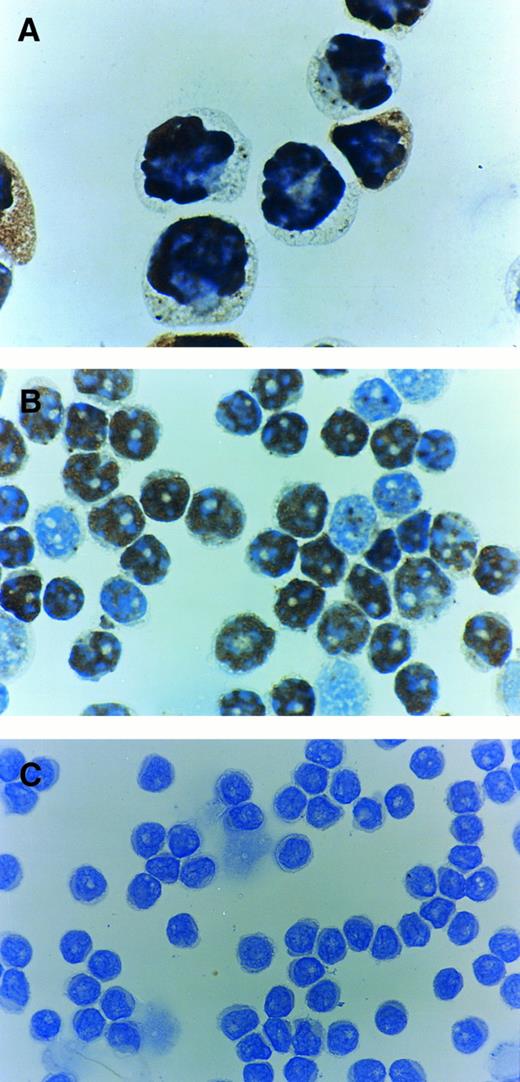
Immunostaining using PAb1801 MoAb directed against the N-terminal residues 1-45 was performed in 17 cases of B-PLL in which sufficient suitable material was available as well as in the control positive CEM cell line.21 The CEM cell line showed a strong nuclear immunostaining in 90% of cells and 8 B-PLL cases were stained positive for p53. The percentage of positive cells varied from 45% to 90% (Table 2). Representative examples of immunocytochemistry are shown in Fig 4. In 6 of 8 positive cases, we repeated the immunocytochemistry with another anti-p53 antibody (DO-1) and the same results were obtained (data not shown).
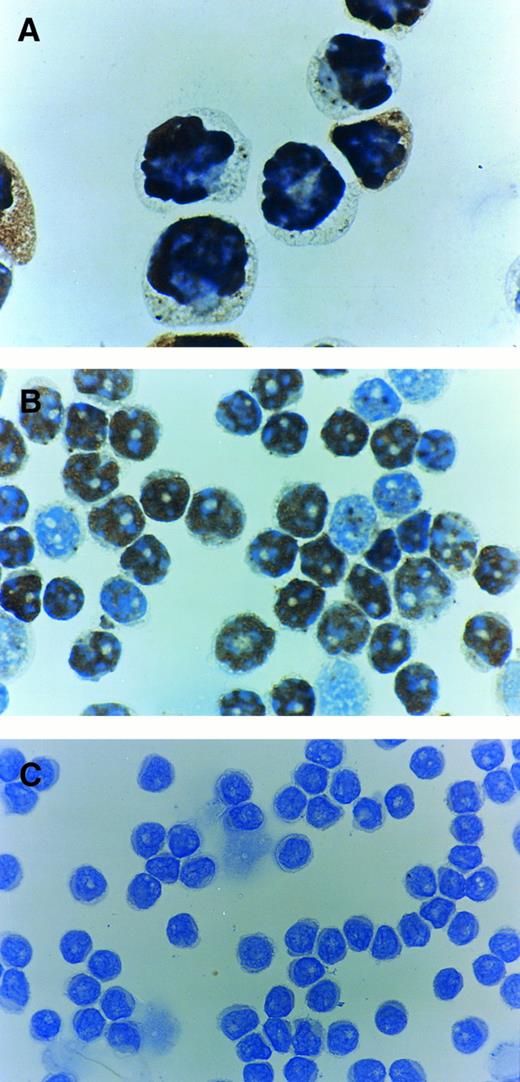
Immunocytochemistry of p53 protein using PAb1801 antibody. (A) Control positive CEM T-cell line showing most of the cells to be positive. (B) Case with a p53 mutation and with 80% of cells positive. (C) Case which is negative for p53 protein despite exhibiting a p53 mutation.
Immunocytochemistry of p53 protein using PAb1801 antibody. (A) Control positive CEM T-cell line showing most of the cells to be positive. (B) Case with a p53 mutation and with 80% of cells positive. (C) Case which is negative for p53 protein despite exhibiting a p53 mutation.
Relationship Between LOH, p53 Mutation, and Protein Expression
Among the 10 B-PLL cases with apparent LOH on 17p, 7 cases showed mutation in the residual allele. In the other 3 cases, no structural alteration of p53 gene was found. On the other hand, 3 cases with p53 mutation showed two bands on Southern blot (Table 2). However, sequencing analysis did not show residual wild-type signal. These data suggest the presence of a more localized deletion adjacent to the p53 gene in these 3 cases.
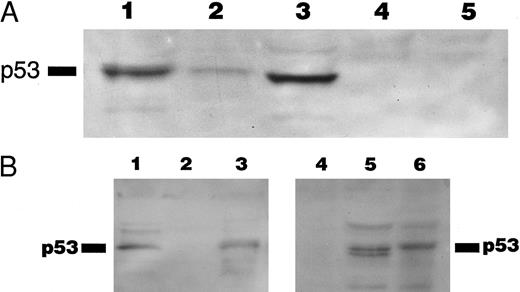
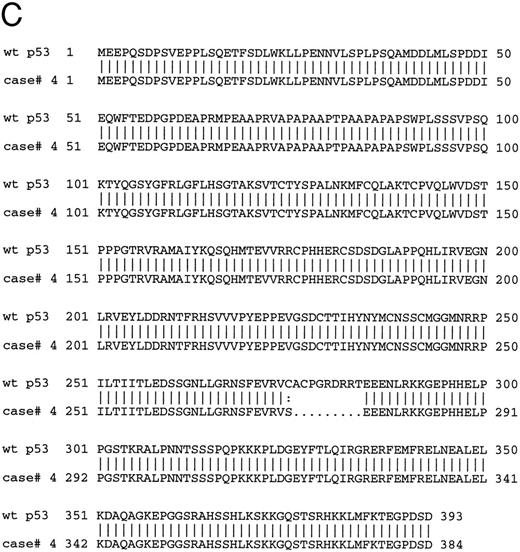
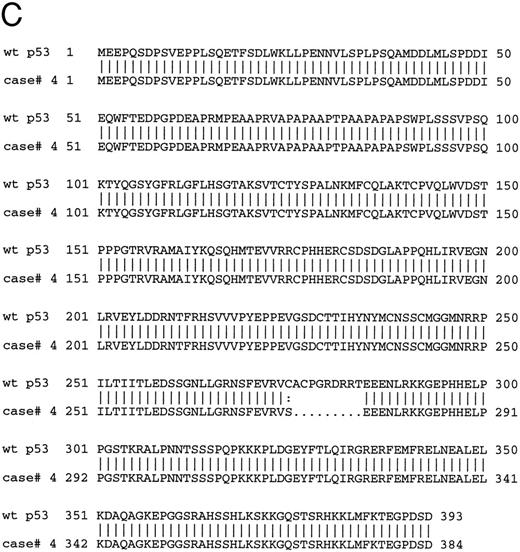
The 8 cases of B-PLL with positive immunostaining comprised 4 cases with missense mutation, 2 cases with short deletions, and 2 cases with normal p53 sequence. The 2 cases carrying deletions (cases no. 4 and 10) and 1 of the cases without mutation (case no. 11) were further assessed by Western blot using PAb 1801 and DO-1 antibodies as a probe. A band of 53 kD that comigrated with the p53 band in the CEM cell line control was recognized by the antibody in samples from cases nos. 4 and 11 but not in samples from case no. 10 (Fig 5A) The same results were observed in cases no. 4 and 10 when using an MoAb directed to the C-terminus of p53 (Pab421; Fig 5B). Figure 5C showed the predicted amino acid sequence of the mutated protein of case no. 4 compared with the wild-type p53 protein sequence.
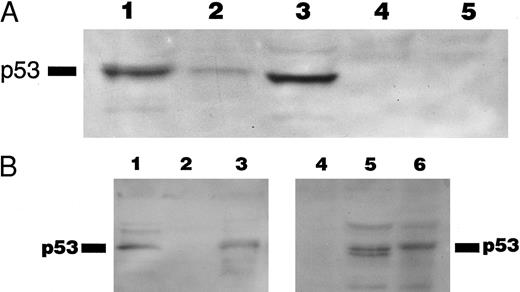
(A) Immunoblotting with DO-1 antibody. Lane 1, CEM positive control; lane 2, case no. 11 with no p53 mutation; lane 3, case no. 4 with a 27-bp intragenic deletion; lane 4, case no. 10 with a 35-bp deletion; lane 5, case no. 5 with a 5-bp deletion at exon 7 and p53 protein negative by immunocytochemistry. (B) Immunoblotting of whole cell lysates from cases no. 4 (lanes 1 and 5) and case no. 10 (lanes 2 and 4) with PAb 1801 and PAb 421 antibodies. Case no. 4 (lanes 1 and 5) showed a 53-kD band with both antibodies and no band is observed with any of the antibodies in case no. 10 (lanes 2 and 4). Lane 3 and 6 correspond to CEM-positive control. (C) Comparison of the predicted amino acid sequence of case no. 4 with the wild-type p53 protein sequence.
(A) Immunoblotting with DO-1 antibody. Lane 1, CEM positive control; lane 2, case no. 11 with no p53 mutation; lane 3, case no. 4 with a 27-bp intragenic deletion; lane 4, case no. 10 with a 35-bp deletion; lane 5, case no. 5 with a 5-bp deletion at exon 7 and p53 protein negative by immunocytochemistry. (B) Immunoblotting of whole cell lysates from cases no. 4 (lanes 1 and 5) and case no. 10 (lanes 2 and 4) with PAb 1801 and PAb 421 antibodies. Case no. 4 (lanes 1 and 5) showed a 53-kD band with both antibodies and no band is observed with any of the antibodies in case no. 10 (lanes 2 and 4). Lane 3 and 6 correspond to CEM-positive control. (C) Comparison of the predicted amino acid sequence of case no. 4 with the wild-type p53 protein sequence.
DISCUSSION
Mutations of p53 are the most common genetic abnormality in cancer.12,22 They have been extensively studied in various malignancies of mature B cells, including NHL,23-26 CLL,27-30 and myeloma.31 In NHL and CLL, it is clear that p53 mutation is a secondary event often associated with progression or transformation.26,27,32 33 In this study, we report the frequency and nature of p53 abnormalities in B-PLL, a rare and aggressive form of leukemia.
First, allelic loss at 17p (eg, LOH) was studied by Southern blot using two highly polymorphic probes that detect minisatellite sequences at 17p13.3. Fifty-three percent of B-PLL cases showed only one band. Because there is a great chance (95%) of detecting a difference between the restriction fragment lengths with these probes if two alleles are present, it is likely that cases with a single band have one allele of chromosome 17p13.3 deleted.11 23
Mutations of p53 gene were studied using direct sequencing of PCR products. We limited our analysis to the highly conserved region between exons 5 and 9 that prior studies14,34 35 showed to contain 80% to 95% of all p53 mutations in a variety of human cancers, including hematologic malignancies.
The overall frequency of p53 mutation in B-PLL was 53%, the highest incidence of p53 mutation reported within lymphoid neoplasias, reaching figures found in solid tumors. Concerning the mutational spectrum of p53, we found that, unlike other B-cell malignancies in which substitution accounted for 90% of p53 mutations (data obtained from the p53 mutational database36 ), 40% of the mutations detected in B-PLL were either deletions or insertions. Three cases exhibited deletions of 5, 27, and 35 bp and 1 case exhibited a 2-bp insertion. These mutations were localized in the central part of the molecule (exons 7 and 8). Among the missense mutations, 4 cases were transversions and 2 cases were transitions. This pattern is different from that found by us in CLL (data not shown) and reported in other lymphoid malignancies that consist primarily of G → A transitions at cathespin G (CpG) dinucleotides.14,34,35,37 In addition, according to the p53 mutational database,36 8 of the 10 changes observed in the p53 gene in B-PLL had not been previously reported in hematologic malignancies. This difference in the spectrum of p53 mutations may reflect the etiologic contribution of distinct exogenous or endogenous factors to B-PLL pathogenesis.
Deletions/insertions are a type of mutations that were previously reported to be scattered along the p53 gene and not only confined to exons 5 through 9. Thus, we performed SSCP analysis in cases in which mutations were not found in exons 5 through 9 to screen for mutations in the rest of the p53 coding region. No altered migration was observed in any of these cases, suggesting that p53 mutations in B-PLL may be preferentially localized in the highly conserved DNA-binding region.
Several observations support the concept that p53 mutation is accompanied by loss of the remaining allele.11,38 In this series, no additional signal corresponding to the normal p53 sequence was detected in any of the mutated cases, implying deletion of the nonmutated allele. These events may occur simultaneously or one may precede the other during the disease progression. When p53 mutation and LOH results were compared, 3 cases with LOH without mutation in the coding region of p53 gene were detected, which may suggest that in B-PLL deletion preceded mutation. In contrast, 3 cases with mutation and no remaining signal of wild-type sequence showed two bands by Southern blot with p144-D6 or pYNZ22 probes. This finding would imply that the break of the deletion in these cases was between the p53 locus at 17p13.1 and 17p13.3 where pYNZ22 and p144-D6 map. Interstitial deletions involving p53 but not the distal part of 17p have been previously reported in Burkitt's lymphomas and in breast cancer.23 39
Positive immunostaining is generally thought to reflect the presence of p53 mutations, because mutation usually results in protein stabilization and an increase in its half life. Eight of 17 B-PLL cases studied were positive for p53 expression by immunocytochemistry. However, in this series, there was not a strict correlation between mutation and detectable p53 expression. Thus, in 2 cases with positive immunostaining, we were unable to detect p53 mutations either by direct sequencing of exons 5 through 9 or by SSCP of exons 2, 3, 4, 10, and 11. In 1 of these cases (no. 11), the immunocytochemistry was confirmed by Western blotting, which showed the presence of apparently wild-type protein. In these cases, mechanisms other than mutation of the protein coding sequences may result in overexpression and/or stabilization of p53 protein. Similarly, 2 cases with missense mutation were unexpectedly negative for p53 protein. One possible explanation for this is a false-negative immunocytochemical result due to poor preservation of the antigen. Another possibility is that these particular point mutations may result in loss of p53 function without protein stabilization.
It has also been suggested from previous studies that nonsense or frameshift mutations may result in negative immunostaining.37,40 In this series, in 2 cases, 1 with a deletion of 5 bp and another with an insertion of 2 bp, this was indeed the case and no positively staining cells were observed. However, we unexpectedly found 2 cases (cases no. 4 and 10 with deletions of 27 and 35 bp, respectively) that both exhibited a high percentage of positively staining cells. To ensure that this result was not artifactual, the staining was repeated with different MoAbs that recognize a different epitope within the amino terminus of the p53 protein and similar results were obtained. To corroborate these results, Western blotting with N- and C-terminal MoAbs was performed. In case no. 4, all MoAbs detected a 53-kD protein confirming the immunocytochemistry. Thus, in this case, it appears that the deletion of 27 bp from exon 8 resulted in the deletion of 9 amino acids from the DNA-binding region of p53, but yet also resulted in protein stabilization. In contrast, case no. 10 was negative by Western blotting using the same reagents. The reasons for this discrepancy are not clear but may reflect a false-positive immunocytochemical reaction, because this was performed on cryopreserved cytospin preparations that may not be optimal. However, overall, our results indicate that in B-PLL immunocytochemistry may not always correlate with p53 mutation. Similar results have been reported by others in other lymphoid malignancies.39,41 42
In summary, our results indicate that the overall abnormalities of the p53 gene occur in 75% of cases of B-PLL; this is the highest frequency of p53 abnormalities reported in any hematologic malignancy. Therefore, disruption of the p53 pathway is probably an important event in the pathogenesis of B-PLL. Moreover, both the pattern and the distribution of mutations were distinctive from those observed in other B-cell malignancies, implying the operation of distinct etiologic mechanisms. B-PLL is known for its aggressive clinical course and poor response to chemotherapy,5 with a median survival of 3 years.41 It is likely that the abnormalities of p53 shown in this report may underline the resistance to treatment and aggressive course of this disease.
ACKNOWLEDGMENT
We are grateful to “BREAKTHROUGH Breast Cancer” for the use of the ABI sequencer.
Supported by grants from the Kay Kendall Leukaemia Trust, the European Community, and the D.C. Collen Foundation.
Address reprint requests to Daniel Catovsky, MD, Academic Department of Haematology and Cytogenetics, The Royal Marsden Hospital, 203 Fulham Rd, London SW3 6JJ, UK.