Abstract
We have previously reported a mutated protein C, designated protein C Nagoya (PCN), characterized by the deletion of a single guanine residue (8857G). This frameshift mutation results in the replacement of the carboxyl-terminal 39 amino acids of wild-type protein C (G381-P419) by 81 abnormal amino acids. This elongated mutant was not effectively secreted, and was retained in the endoplasmic reticulum. To determine why PCN is not secreted, we constructed a series of mutants from which some or all of the 81 amino acids were deleted. None of these shortened proteins were secreted from producing cells, indicating that the carboxyl-terminal extension is not mainly responsible for the intracellular retention of PCN, and that the 39 carboxyl-terminal amino acids of wild-type protein C are required for secretion. To determine which residues are essential for the secretion of protein C, deletion mutants of the carboxyl-terminal region (D401-P419) were prepared. Metabolic labeling showed that mutants of protein C truncated before W417, Q414, E411, or K410 were efficiently secreted. On the other hand, the mutants truncated before D409 were retained and degraded intracellularly. Immunofluorescence and immunoelectron microscopy showed that truncation before D409 blocks the movement from rough endoplasmic reticulum to the Golgi apparatus. To understand the conformational change in the carboxyl-terminal region, two models of truncated activated protein C were constructed using energy optimization and molecular dynamics with water molecules.
PROTEIN C IS A VITAMIN K-dependent glycoprotein and plasma serine protease precursor that acts as an anticoagulant and plays an important role in hemostasis.1The native human protein C molecule is a disulfide-linked heterodimer composed of light and heavy chains. It is synthesized in the liver as a 461-amino acid precursor protein that undergoes extensive cotranslational and post-translational modification.2Protein C deficiency is an autosomally inherited disorder that is associated with a high risk of recurrent venous thrombosis. The protein C mutation database contains 160 unique mutations in the protein C gene from a total of 315 unrelated probands.3 Most are missense mutations, with the remainder consisting mainly of nonsense mutations, splice-site abnormalities, frameshift mutations, and others.
We previously reported on a mutated protein C, designated protein C Nagoya (PCN), characterized by the deletion of a single guanine residue ( 8857 G) among four consecutive guanine nucleotides [W380 (TGG)-G381 (GGT)].4 This results in a frameshift mutation with replacement of the carboxyl-terminal 39 amino acids of wild-type protein C (wtPC) (G381-P419) by 81 abnormal amino acids. This elongated variant is mostly retained and degraded within the endoplasmic reticulum (ER), and is associated with 78-kD glucose regulated protein (GRP78)/Immunoglobulin heavy chain binding protein (BiP) and 94-kD glucose regulated protein (GRP94).5 These data suggest that the carboxyl-terminal portion of this protein is important for its efficient secretion.
In the present study, we produced a series of deletion mutants of both PCN and wtPC using site-directed mutagenesis to identify amino acid sequences that contribute to the impaired secretion of protein C. The results of pulse-labeling, immunofluorescence, and immunoelectron microscopy indicated that the carboxyl-terminal region of protein C plays an essential role in its intracellular transport. We constructed structural models of the protease domains of truncated protein C and discussed the role of the carboxyl-terminal region in the secretion of protein C.
MATERIALS AND METHODS
Construction of carboxyl-terminal deletion mutants.
Complementary DNA (cDNA) fragments coding for wtPC and PCN were ligated into the multicloning site of the pBluescript II KS (+) (Stratagene, La Jolla, CA) as described previously,4 and designated pBPC and pBPCN, respectively. The carboxyl-terminal deletion mutants of PCN and wtPC were constructed by oligonucleotide-directed mutagenesis. The oligonucleotides used to construct the deletion mutants of PCN and wtPC are shown in Table 1. The mutant bases are indicated by lower case letters. The mutated fragments were constructed by polymerase chain reaction (PCR) amplification with C1 and other antisense primers. Each pair of PCR fragments were then subcloned in pBluescript II KS (+) by T-A cloning.6 After digestion with ApaI and XbaI, the amplified fragments were inserted in pBPCN or pBPC. The resulting mutations were confirmed by the dideoxy chain termination sequencing method.7 The mutated cDNAs were digested from pBluescript II KS (+) by EcoRI and ligated into the pED vector.8 Mutated proteins were designated as shown in Fig 1A.
(A) The alignment of carboxyl-terminal amino acids of wtPC, PCN, and their deletion mutants. The carboxyl-terminal amino acids are indicated by standard one-letter designations. Hydrophobic regions of PCN are underlined. The hydropathy plot was performed with the LASERGENE Navigator (DNASTAR Inc, Madison, WI) determined by the methods of Kyte and Doolittle.34 (B) Northern blot analysis of the stably transfected CHO cells. Total RNA (5 μg) from each cell line was separated by 1% formaldehyde-agarose gel electrophoresis, transferred to a Zeta-Probe membrane (Bio-Rad), and hybridized with 32P-labeled PC cDNA probe.4
(A) The alignment of carboxyl-terminal amino acids of wtPC, PCN, and their deletion mutants. The carboxyl-terminal amino acids are indicated by standard one-letter designations. Hydrophobic regions of PCN are underlined. The hydropathy plot was performed with the LASERGENE Navigator (DNASTAR Inc, Madison, WI) determined by the methods of Kyte and Doolittle.34 (B) Northern blot analysis of the stably transfected CHO cells. Total RNA (5 μg) from each cell line was separated by 1% formaldehyde-agarose gel electrophoresis, transferred to a Zeta-Probe membrane (Bio-Rad), and hybridized with 32P-labeled PC cDNA probe.4
Stable expression of recombinant protein C.
CHO-DUKX-B11 cells9 were stably transfected with expression vectors using Lipofectin reagent (GIBCO-BRL, Gaithersburg, MD) as previously described.5 Pulse-labeling and immunoprecipitations were performed as previously described.5 Data are presented as the mean ± standard deviation (SD) for three observations.
Northern blot analysis.
Northern blot analysis was performed as previously described.10 In brief, total RNA (5 μg) from each clone was separated by 1% formaldehyde-agarose gel electrophoresis in a minigel system (ADVANCE Co, Ltd, Tokyo, Japan), transferred to a Zeta-Probe membrane (Bio-Rad, Hercules, CA), and then crosslinked using a UV Stratalinker 1800 (Stratagene). The membrane was hybridized with32P-labeled PC cDNA probe,4 and exposed to Kodak-XR5 film (Eastman Kodak, Rochester, NY) for 20 hours.
Measurements of protein C antigen and activity.
The protein C antigen level and anticoagulant activity were determined as previously described.5 Statistical analysis of secreted protein C antigen and activity level was performed between wtPC and other secreted proteins by Mann-Whitney test. A P value of less than .05 was considered to be significant.
Immunofluorescence microscopy and immunoelectron microscopy.
Immunofluorescence microscopy and immunoelectron microscopy were performed as previously described.5
Molecular model of protein C.
Molecular models of activated protein C were constructed using the homology modeling methods, as described previously.11Briefly, coagulation factor Xa (Protein Data Bank code: 1HCG)12,13 was used as the template protein for model construction of the second EGF-like and protease domains of activated protein C. In the present study, the catalytic domain was modeled and energetically optimized for the position 170-41211 and, hence, the wtPC model was called PC412. To obtain the models of two deletion mutants of protein C named PC408 and PC410, the carboxyl-terminal amino acids corresponding to D409-A412 and E411-A412 were deleted, respectively. To obtain the best structural deletion models, the full set of protein C protease domain coordinates was subjected to energy optimization and molecular dynamics (MD) calculation in a sphere of 76Å diameter packed with 5746 and 5738 water molecules for PC408 and PC410, respectively. Energy minimization and MD calculation procedures were carried out using a program APRICOT14 with AMBER united atom force field.15 Constraints by SHAKE algorithm16were used on bonds with hydrogens and lone pairs.
RESULTS
Pulse-labeling of the deletion mutants of PCN.
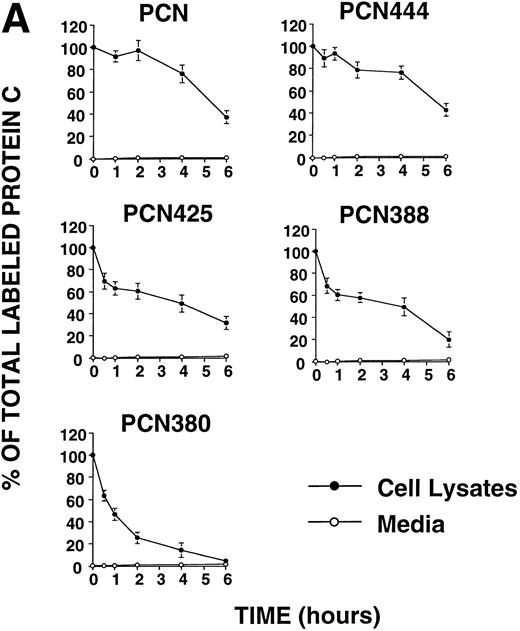
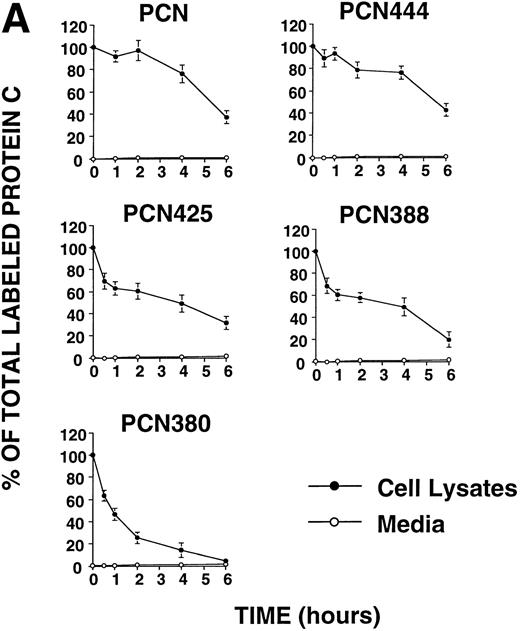
The 81 carboxyl-terminal amino acids of PCN contain highly hydrophobic regions at residues 445-458 (Fig 1A). Previous study showed that the carboxyl-terminal long hydrophobic amino acids of pregnancy-specific glycoprotein (PSG) specified intracellular retention and stability.17 To investigate the effects of the carboxyl-terminal hydrophobic region of PCN on its secretion, we constructed four deletion mutants truncated at 444, 425, 388, and 380, which is immediately before the frameshift mutation. Chinese hamster ovary (CHO) cells stably transfected with the different constructs were pulse-labeled with [35 S]Met and chased for up to 6 hours. None of the truncated variants were efficiently secreted from CHO cells (Fig 2A). The protein C messenger RNA (mRNA) levels of each cell line were comparable (Fig 1B), indicating that impaired secretion of the mutant proteins was not due to the depressed levels of transcripts. These results showed that the carboxyl-terminal extension is not predominantly responsible for the intracellular retention of PCN, and that the 39 carboxyl-terminal amino acids of wtPC are required for secretion.
(A) Pulse-chase analysis of PCN and its deletion mutants. Cells were labeled and chased as described previously.5 The data are expressed as percent of the intracellular35S-labeled protein C present at the zero time point. (•, intracellular protein C; ○, secreted protein C). Values represent the means ± standard deviation of three independent experiments. (B) Pulse-chase analysis of wild-type protein C and its deletion mutants. Cells were labeled and chased as described previously.5 The data are expressed as percent of the intracellular35S-labeled protein C present at the zero time point (•, intracellular protein C; ○, secreted protein C; ×, total protein C, ie, the summation of secreted and intracellular material). Values represent the means ± standard deviation of three independent experiments.
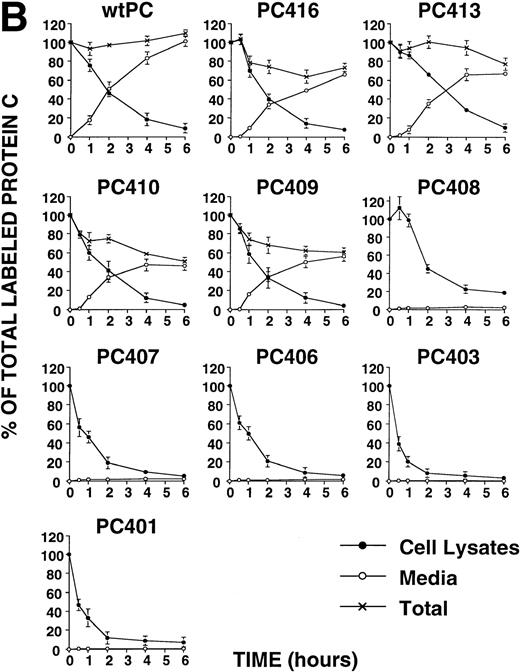
(A) Pulse-chase analysis of PCN and its deletion mutants. Cells were labeled and chased as described previously.5 The data are expressed as percent of the intracellular35S-labeled protein C present at the zero time point. (•, intracellular protein C; ○, secreted protein C). Values represent the means ± standard deviation of three independent experiments. (B) Pulse-chase analysis of wild-type protein C and its deletion mutants. Cells were labeled and chased as described previously.5 The data are expressed as percent of the intracellular35S-labeled protein C present at the zero time point (•, intracellular protein C; ○, secreted protein C; ×, total protein C, ie, the summation of secreted and intracellular material). Values represent the means ± standard deviation of three independent experiments.
Truncation of wtPC before D409 prevents its secretion.
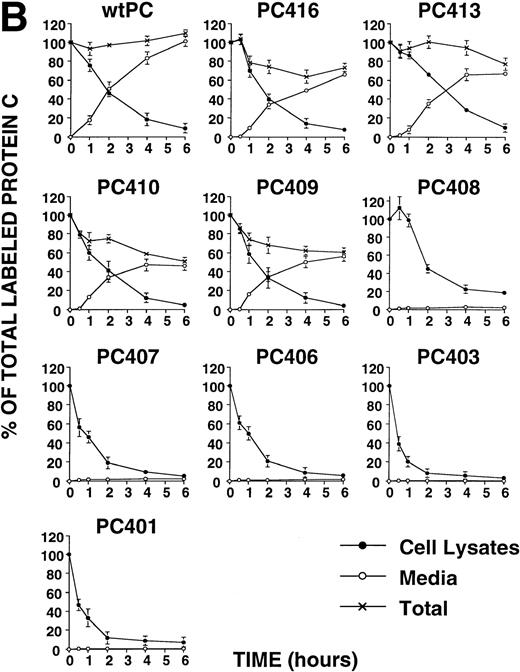
To obtain information on the structural requirements necessary to the secretion, we expressed a series of recombinant molecules containing limited deletion of the sequence present in the carboxyl-terminal of wtPC. The results showed that PC416, PC413, PC410, and PC409 were efficiently secreted from the cells. The secreted antigen levels of these mutants were significantly lower than that of wtPC (Table 2). The relative activities of secreted protein C mutants were not significantly different from wtPC (Table 2). On the other hand, PC408, PC407, PC406, PC403, and PC401 appeared to undergo intracellular retention and degradation. In all of these cases, intracellular degradation was apparent by 6 hours after initiation of the chase (Fig 2B).
Immunofluorescence microscopy.
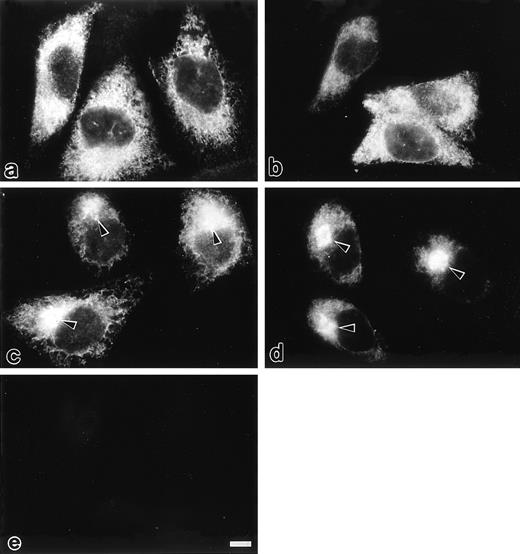
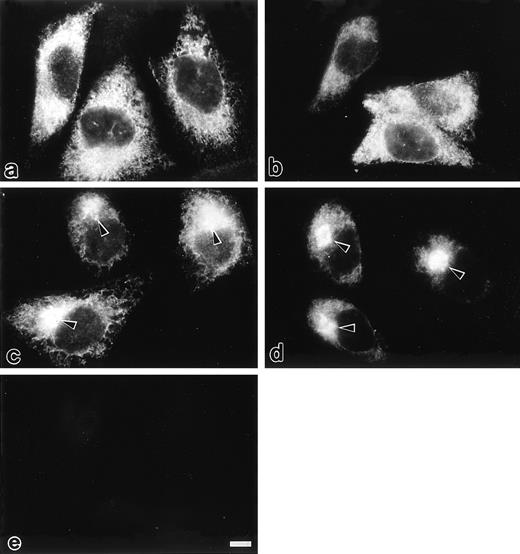
Intracellular localization of selected mutants of protein C was examined by immunofluorescence microscopy (Fig 3). In transfected CHO cells expressing PC407 and PC408, cytoplasmic reticular staining was observed, whereas a perinuclear spot was not observed (Fig 3a and 3b). In the cells expressing PC409 (Fig 3c) and wtPC (Fig 3d), both perinuclear spots with prominent brightness and cytoplasmic reticular staining were observed, which correspond to the rough ER and the Golgi apparatus, respectively.5,18,19 20 No immunofluorescence was detected in mock-transfected CHO cells (Fig 3e). Thus, PC407 and PC408 appear to be retained in the rough ER, whereas PC409 exhibits normal secretion kinetics.
Immunofluorescence microscopic localization of different protein C variants transfected in the CHO cells protocols are described under Materials and Methods. In the cells expressing PC407 (a) and PC408 (b), cytoplasmic reticular staining was observed. In the cells expressing PC409 (c) and wtPC (d), a perinuclear spot with prominent brightness (arrow heads) was observed in addition to the cytoplasmic reticular staining. No immunofluorescence was detected in the mock-transfected CHO cells (e). Scale bars, 10 μm.
Immunofluorescence microscopic localization of different protein C variants transfected in the CHO cells protocols are described under Materials and Methods. In the cells expressing PC407 (a) and PC408 (b), cytoplasmic reticular staining was observed. In the cells expressing PC409 (c) and wtPC (d), a perinuclear spot with prominent brightness (arrow heads) was observed in addition to the cytoplasmic reticular staining. No immunofluorescence was detected in the mock-transfected CHO cells (e). Scale bars, 10 μm.
Immunoelectron microscopy.
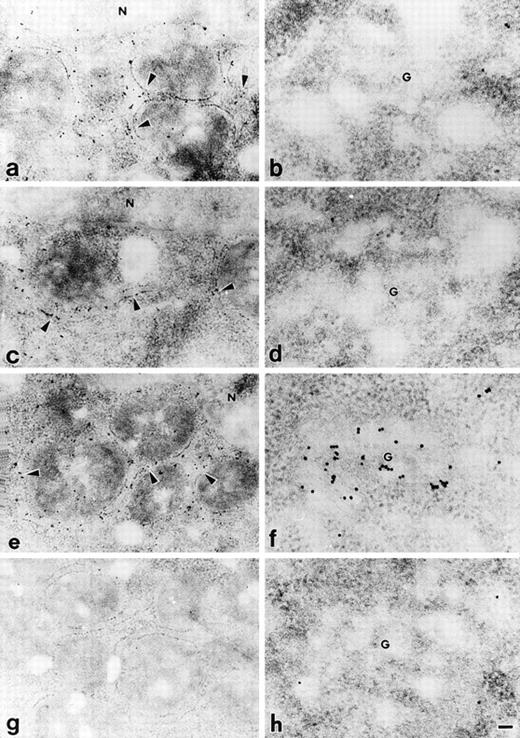
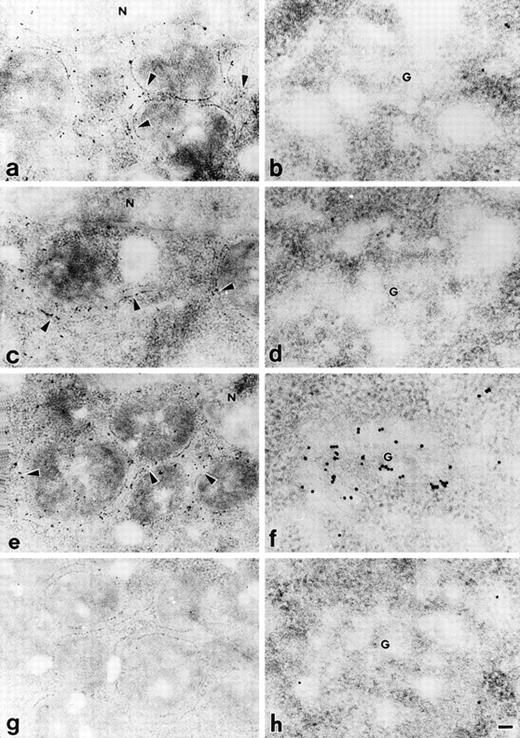
Precise localization of PC407, PC408, and PC409 was determined by postembedding immunoelectron microscopy using a 10 nm colloidal gold-conjugated second antibody. In the CHO cells expressing PC407 and PC408, gold particles were mainly distributed in the rough ER (Fig 4a and 4c), and no gold particles were localized to the Golgi apparatus (Fig 4b and 4d). In the cells expressing PC409, the Golgi apparatus was heavily labeled with gold particles (Fig 4f) in addition to the localization of particles in the rough ER (Fig 4e). Mock-transfected CHO cells did not show any specific immunolabelling (Fig 4g and h). From these results, we conclude that the transport of PC407 and PC408 from rough ER to the Golgi apparatus is blocked and that these mutant proteins are retained in the rough ER, whereas PC409 was transported through the Golgi apparatus and secreted normally.
Immunoelectron microscopic localization of different protein C variants transfected in the CHO cells. Transfected CHO cells were examined by postembedding immunoelectron microscopy using 10 nm colloidal gold-conjugated second antibody. In the cells expressing PC407 (a, b) and PC408 (c, d), gold particles were mainly distributed in rough ER (a, c), and no gold particles were localized to the Golgi apparatus (b, d). In the cells expressing PC409, gold particles were distributed in both rough ER (e) and Golgi apparatus (f). The mock-transfected CHO cells showed no specific immunolabelling (g, h). Arrowheads, rough ER; G, Golgi apparatus; N, nucleus. Scale bar, 100 nm for a, c, e and g; 50 nm for b, d, f and h.
Immunoelectron microscopic localization of different protein C variants transfected in the CHO cells. Transfected CHO cells were examined by postembedding immunoelectron microscopy using 10 nm colloidal gold-conjugated second antibody. In the cells expressing PC407 (a, b) and PC408 (c, d), gold particles were mainly distributed in rough ER (a, c), and no gold particles were localized to the Golgi apparatus (b, d). In the cells expressing PC409, gold particles were distributed in both rough ER (e) and Golgi apparatus (f). The mock-transfected CHO cells showed no specific immunolabelling (g, h). Arrowheads, rough ER; G, Golgi apparatus; N, nucleus. Scale bar, 100 nm for a, c, e and g; 50 nm for b, d, f and h.
Molecular modeling and MD calculation of the catalytic domain of two truncated protein C mutants.
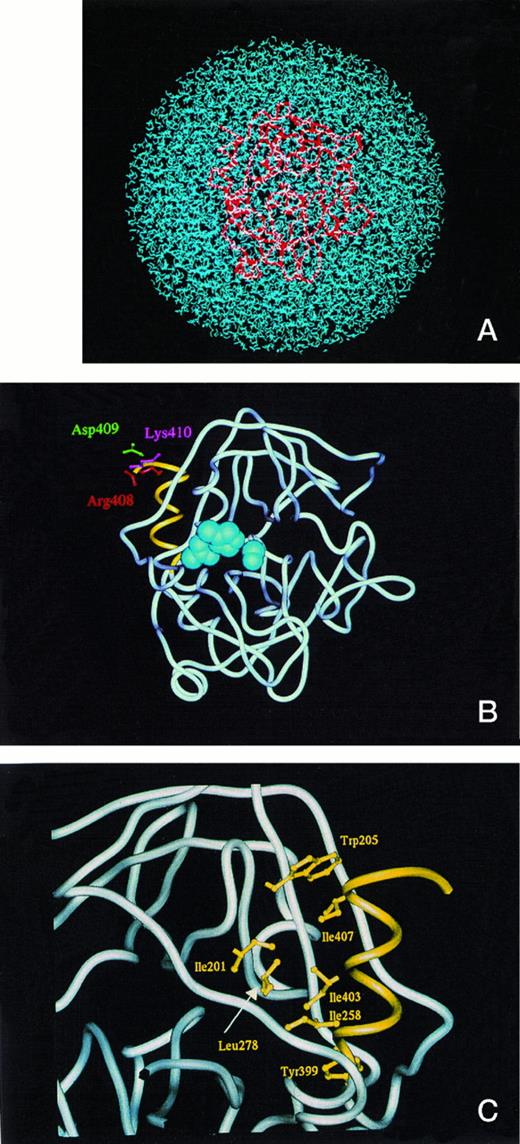
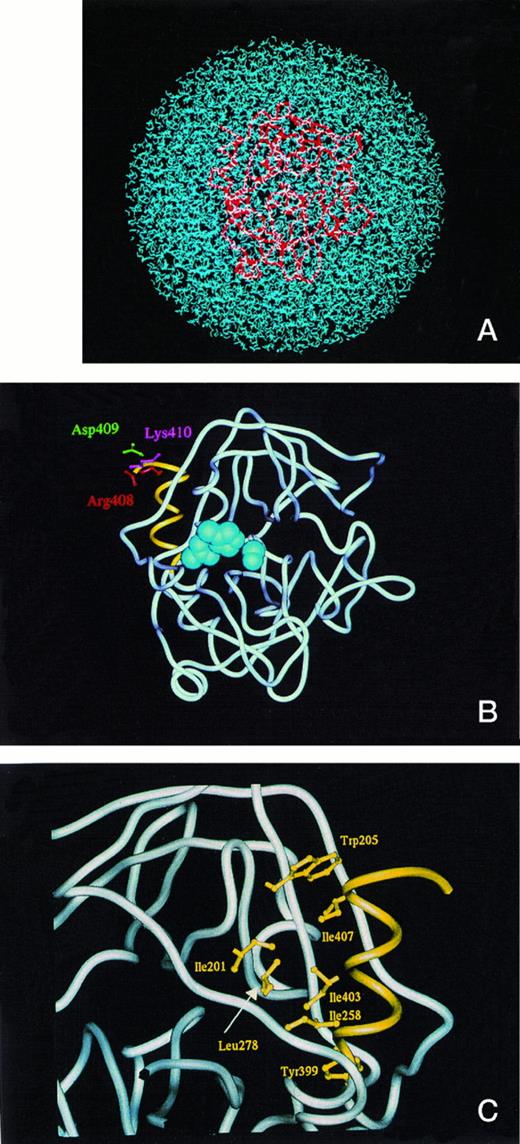
To distinguish the structural differences between the secreted form PC410 and the ER retention form PC408, we constructed models of two deletion mutants of activated protein C. They were constructed with water molecules to obtain the information about conformational stability at room temperature and to describe the fluctuation of the hydrophobic core regions during the MD calculation. Figure 5A shows the full set of PC410 coordinates in a sphere of 76Å diameter packed with 5,738 water molecules. The overall tertiary structural model of PC410 was superimposed with that of wild-type PC412 except for the carboxyl-terminal two amino acids deletion in root mean square deviation for main chain Cα atoms of 1.80Å (data not shown). The PC410 model constructed with water molecules predicts that the carboxyl-terminal region of the protease domain has an alpha-helix with three turns from Y399 to K410 (Fig 5B and 5C). This structural feature in the carboxyl-terminal region is widely observed among serine protease family proteins, which are 1TLD (bovine beta-trypsin), 1PFX (porcine factor IXa), 1TRN (human trypsin), 1PPB (human alpha-thrombin), 1HCG (human coagulation factor Xa), 1LMW (human urokinase-type plasminogen activator), 5CHA (bovine alpha-chymotrypsin A), 2PKA (porcine kallikrein A), 3EST (porcine pancreas elastase), 3RP2 (rat mast cell protease), 1HNE (human leukocyte elastase), 1TON (rat tonin), and 1FUJ (a neutrophil serine protease antigen of Wegener's granulomatosis antibodies) in the Protein Data Bank (Table 3). The alpha-helix with three turns forms the hydrophobic core, and four hydrophobic interactions exist in this hydrophobic core of these serine proteases. In the hydrophobic core of PC410, three hydrophobic amino acid residues Y399, I403, and I407 on the inner face of the alpha-helix interact specifically with the side chains of I201, W205, I258, and L278 (Fig 5C). Table 3 shows four hydrophobic distances in this hydrophobic core. The hydrophobic distances were defined to be the nearest distance between two hydrophobic amino acid residues. This hydrophobic core formation is important to maintain the tertiary structure. Therefore, it is worth examining if the carboxyl-terminal alpha-helix is conserved in the ER retention form PC408. We constructed the model of PC408 using the same procedures. The resulting PC408 model was superimposed with that of PC412 (data not shown). In the comparison of PC408 with PC410, the root mean square deviation for Cα atoms of the catalytic domain was 1.77Å. The PC408 model showed the same four hydrophobic distances as those in PC410 (Table 3). Trypsin also showed the same hydrophobic distances that are positioned in the equivalent places on the sequence alignment. Because hydrophobic distances are in a range from 3.4 to 6.4,21 all three residues of I407, I403, and Y399 can still form the expected bonds with hydrophobic amino acid residues in the catalytic domain in both secretory form PC410 as well as ER retention form PC408, indicating that the hydrophobic core is intact. Furthermore, the amino acid deletion at the carboxyl-terminal region did not expose any hydrophobic regions on either PC410 or PC408.
(A) The model of the secreted form of truncated protein C, PC410, in the presence of water molecules. The full set of protein C protease domain coordinates was subjected to energy optimization in a sphere of 76Å diameter packed with 5,738 water molecules to obtain the best structural deletion models. The activated protein C was shown by tube model (red) and the water molecules were shown by blue. (B) The model of the secreted form, PC410. The average main chain coordinates during 45 and 60 ps obtained from MD calculation was shown by the tube model structure. The active site triad was shown by the CPK shells (blue), and the carboxyl-terminal three-rounds alpha-helix from Tyr399 to Lys410 was shown by yellow. The side chains of Arg408, Asp409, and Lys 410 were shown by the ball and stick model by red, green, and blue, respectively. (C) Hydrophobic interaction of the carboxyl-terminal alpha-helix with the surrounding residues. The carboxyl-terminal structure of PC410 was enlarged and the residues involved in the hydrophobic interaction with the carboxyl-terminal alpha-helix from Tyr399 to Lys410 were shown by the ball and stick model in yellow. This figure was taken from the backside of Fig 5B.
(A) The model of the secreted form of truncated protein C, PC410, in the presence of water molecules. The full set of protein C protease domain coordinates was subjected to energy optimization in a sphere of 76Å diameter packed with 5,738 water molecules to obtain the best structural deletion models. The activated protein C was shown by tube model (red) and the water molecules were shown by blue. (B) The model of the secreted form, PC410. The average main chain coordinates during 45 and 60 ps obtained from MD calculation was shown by the tube model structure. The active site triad was shown by the CPK shells (blue), and the carboxyl-terminal three-rounds alpha-helix from Tyr399 to Lys410 was shown by yellow. The side chains of Arg408, Asp409, and Lys 410 were shown by the ball and stick model by red, green, and blue, respectively. (C) Hydrophobic interaction of the carboxyl-terminal alpha-helix with the surrounding residues. The carboxyl-terminal structure of PC410 was enlarged and the residues involved in the hydrophobic interaction with the carboxyl-terminal alpha-helix from Tyr399 to Lys410 were shown by the ball and stick model in yellow. This figure was taken from the backside of Fig 5B.
DISCUSSION
Several studies have shown the significance of the carboxyl-terminal region of protein C. Peptide inhibition studies indicate that potential sites of the activated protein C factor Va interaction are located on the exposed portion of the carboxyl-terminal alpha helix, K395-H404.22 The crystal structure of protein C supported this finding.23 A database of naturally occurring mutations and their clinical phenotypes has been established,3 and analysis of these mutants of protein C showed that natural type I mutants I403L and I403M occur in the carboxyl-terminal helix H3 (L400-R408) within the hydrophobic patch, and the likely perturbation of the helix would interfere with the proper interaction of the serine protease domain with the second EGF domain.24,25 26
Earlier results indicated the existence of a complex containing GRP78, GRP94, and PC.5 Whereas these two stress proteins are strongly associated with PCN, only low amounts form a complex with wtPC. Our interpretation of these data is that PCN does not acquire its proper folding and is available for longer periods of time than wtPC to associate with these two ER chaperones.
In the present study, to examine why PCN is not secreted, we constructed a series of deletion mutants of the 81 abnormal amino acids at the carboxyl-terminal of PCN. This rationale was based on a previous study showing that mutant PSG with carboxyl-terminal hydrophobic amino acids was retained in the ER, and deletion of this region caused effective secretion. The extra hydrophobic amino acids constituted an ER retention signal.17 Moreover, several studies indicated that the carboxyl-terminal region of certain proteins is essential for secretion and stability.27 28 In the present study, however, none of the truncated PCN proteins were secreted. Although the presence of 81 abnormal amino acids may contribute to the intracellular retention, these results indicated that the loss of normal carboxyl-terminal amino acids (G381-P419) is responsible for the intracellular retention of PCN rather than the extension by abnormal amino acids.
Deletion mutagenesis of wtPC shows that forms of protein C differing in length by only one amino acid (K408 versus D409 residues) have very different fates. Pulse-labeling analysis showed that the shorter proteins were degraded intracellularly. Immunoelectron microscopy showed these molecules were not transported from the ER to the Golgi apparatus. The longer molecules, extended by one residue to include D409, were mostly secreted. A part of these longer molecules was degraded intracellularly, indicating that they did not show an entirely normal secretion pattern (Fig 2b). These data showed that changes in this carboxyl-terminal region may have significant effects on the secretion of this protein.
Previous studies have reported that carboxyl-terminal amino acids play important roles in the secretion of many proteins.17,29,30,31 Among vitamin K-dependent glycoproteins, the carboxyl-terminal amino acid residues 403-415 of factor IX (398-410 in protein C) were studied for their role in protein secretion.32 The mutations of T415 (K410 in protein C) showed only small (T415L, T415S and T415R) or moderate (T415G and T415I) reductions both in the intracellular and secreted factor IX levels. On the other hand, another group (Y404H, Y404P, W407R, I408N, T412K and T412N) had high to moderate levels of intracellular factor IX with no detectable or only basal levels of secreted factor IX. The specific activities observed for secreted mutant factor IX were similar to the normal protein. Severe phenotypes observed for hemophilia B patients with K411stop and W407stop (H406 and W402 in protein C, respectively) further support the importance of the carboxyl-terminal region.33 These findings are consistent with our results.
In this study, we constructed two structural models of deletion mutants of activated protein C, the secretion form PC410 and the ER retention form PC408, using energy optimization and MD calculation with water molecules. Although the other models of protein C were previously published,11, 24-26 none of them were constructed with the water molecules. The overall tertiary structural models of both PC410 and PC408 were superimposed with that of wild-type PC412. In general, the serine protease family has a common structural feature of an alpha-helix with three turns at the carboxyl-terminal region. In activated protein C, the helix is calculated to be from Y399 to K410. Therefore, before model construction, we speculated that the ER retention form PC408 has the structural abnormalities on the carboxyl-terminal alpha-helix. However, even though the model was constructed with the water molecules, the PC408 model showed that the hydrophobic distances in the hydrophobic core in relation to the alpha-helix with three rounds from Y399 to K410 were the same as those in PC410 and in trypsin (Table 3). Accordingly, once the native folding of PC408 occurred, the structure of PC408 was estimated to be thermally stable in the same manner as that of PC410. Therefore, it was not apparent from the model construction that the removal of carboxyl-terminal amino acids should result in structural changes that may lead to improper folding and consequently, defective secretion. Although the overall conformation was not so different, the ER resident molecular chaperones may recognize very subtle changes of the PC408 molecules. Alternatively, the molecular chaperones may recognize the intermediate structure of the folding process of PC408 that would not be predicted by the modeling algorithm.
ACKNOWLEDGMENT
We thank Drs J. Evan Sadler, Maurice J. Keller, Tadashi Matsushita, and Koji Yamamoto for their critical reading of the manuscript and Dr Kotoku Kurachi for helpful discussions. We also thank Tomoko Ichihara, Satoshi Suzuki, Mieko Suzuki, and Chika Wakamatsu for their excellent technical help.
Supported in part by the Grants-in-Aid from the Ministry of Education, Science, and Culture, Japan, and the Surveys and Research on Specific Diseases from the Ministry of Health and Welfare, Japan.
Address reprint requests to Akira Katsumi, MD, Howard Hughes Medical Institute Research Laboratories, Washington University School of Medicine, 660 South Euclid Avenue, Box 8022, St Louis, MO 63110; email:akira@darwin.wustl.edu.
The publication costs of this article were defrayed in part by page charge payment. This article must therefore be hereby marked “advertisement” in accordance with 18 U.S.C. section 1734 solely to indicate this fact.