Abstract
Stem cell factor (SCF) is an important mast cell growth, differentiation, and survival factor. We investigated whether SCF influenced the response of mouse mast cells to an IgE-independent stimulus, eosinophil-derived granule major basic protein (MBP). Mouse bone marrow cultured mast cells (BMCMC) were derived in either concanavalin-stimulated mouse spleen conditioned medium (CM) or SCF. The cloned growth, factor-independent mast cell line Cl.MC/C57.1 was also studied. BMCMC in SCF exhibited cytochemical staining properties, protease and histamine content, and increased serotonin uptake consistent with more mature differentiated mast cells as compared with BMCMC in CM or Cl.MC/ C57.1 cells. BMCMC in SCF released serotonin,14C-labeled arachidonic acid metabolites and tumor necrosis factor-α (TNF-α) on stimulation with MBP, while no response was seen from either BMCMC in CM or Cl.MC/C57.1 cells. All three mast cell populations released mediators on stimulation with the cationic MBP analog, poly-L-arginine, indicating that the cationic charge did not explain the selective response of BMCMC in SCF to eosinophil-derived granule MBP. These findings show that SCF significantly influences mast cell differentiation and the responsiveness of mast cells to eosinophil-derived granule MBP.
© 1998 by The American Society of Hematology.
MAST CELLS PRODUCE a variety of biologically active mediators that play an important role in hypersensitivity and other allergic inflammatory reactions. Mast cell degranulation is accompanied by the release of preformed mediators, such as histamine, serotonin (in murine species), proteases, proteoglycans, and tumor necrosis factor-α (TNF-α). As well, mast cells can generate newly synthesized mediators, such as products of arachidonic acid metabolism, platelet activating factor, and a variety of multifunctional cytokines.1
The activation of mast cells by antigen cross-linking of IgE antibodies bound to high-affinity IgE receptors (FcεRI) on the cells' surface is one of the most extensively studied mechanisms of mast cell degranulation.2 However, a number of alternative pathways that are independent of IgE antibodies can elicit mast cell mediator release. For example, complement fragments, bradykinin, bacterial products, and certain neuropeptides, such as substance P, can induce mast cell degranulation.3,4 As well, certain proteins contained within the cytoplasmic granules of eosinophils have actions on mast cells and basophils. The eosinophil-derived granule major basic protein (MBP) has been shown to elicit histamine release from rodent mast cells and human basophils.5 6 Thus, mast cell activation by IgE-independent mechanisms may be important in the pathogenesis of a number of allergic or inflammatory conditions.
The response of mast cells to stimuli not involving IgE antibodies is critically dependent on the population of mast cells examined. Mast cells in different anatomic locations in rodent species and humans exhibit significant phenotypical and functional heterogeneity.7 Mast cells in rodent species can be broadly divided into at least two populations: mast cells located at mucosal surfaces termed mucosal-type mast cells and mast cells in the skin, peritoneal cavity, and submucosa of the intestinal tract termed connective tissue-type mast cells. These mast cell populations differ in a number of characteristics, including biochemical and histochemical properties, the types of mediators produced upon degranulation, as well as their functional response to agents that induce mast cell degranulation through IgE-independent mechanisms.8 Thus, the response of mast cells at one anatomic site to a given stimulus does not necessarily predict the response of mast cells at a different anatomic location to the same stimulus.
The factors controlling mast cell heterogeneity have not been completely defined. A number of cytokines have been shown to influence mast cell proliferation and maturation, including interleukin-3 (IL-3), IL-4, IL-9, IL-10, and nerve growth factor, but recent studies suggest that stem cell factor (SCF) plays a critical role in mast cell development and survival. For example, mice genetically deficient in SCF have an absence of mast cells in both connective tissues and at mucosal surfaces.9 SCF acting alone can induce mast cell proliferation and maturation in vitro10 and influence the development of both mucosal and connective tissue-type mast cells in vivo.11 In addition to its effects on mast cell differentiation, SCF also has profound effects on mast cell function. For example, while SCF has little effect on mast cell mediator release in vitro, we reported that the intradermal injection of SCF elicits mast cell degranulation and mast cell-dependent cutaneous inflammation.12
In the current study, we investigated whether soluble recombinant rat SCF influences the response of mast cells to an IgE-independent stimuli. We now report that bone marrow cultured mast cells (BMCMC) derived in SCF acquire the ability to release preformed and newly synthesized mediators in response to eosinophil-derived granule MBP.
MATERIALS AND METHODS
Cells.
Studies were conducted with three different populations of murine mast cells. A cloned, growth factor–independent mast cell line (Cl.MC/C57.1) that was originally derived from BMCMC isolated from BALB/c mice13 was maintained in Dulbecco's modified Eagle's medium (GIBCO, Grand Island, NY) with 10% heat-inactivated fetal calf serum (Intergen, Purchase, NY), 50 μmol/L 2-mercaptoethanol, and 2 mmol/L L-glutamine at 37°C in 5% CO2. In addition, femoral bone marrow cells were obtained from BALB/c mice and maintained in medium as above, but supplemented with either Concanavalin A-stimulated mouse spleen cell-conditioned medium (BMCMC in conditioned medium [CM]), as previously described,14 or recombinant rat SCF164 (50 ng/mL). Mast cells derived in 20% WEHI-3–conditioned medium (a source of IL-3) were also examined. After 4 to 6 weeks, mast cells represented more than 98% of the total cells as determined by neutral red staining.
Histochemical analysis and histamine content of different mast cell populations.
The different mast cell populations were washed and suspended at 3 × 106 cells/mL in their respective media. Cytocentrifuge slides were prepared from each mast cell population and stained in 0.5% alcian blue (Rowley Biochemical Institute, Rowley, MA), pH 0.1 at 60°C for 10 minutes. Slides were then immediately counterstained in 0.1% safranin (Rowley Biochemical Institute) for 5 minutes at room temperature. The slides were then rinsed in water, 95% ethanol, 100% ethanol, and xylene.
One-milliliter aliquots (3 × 106 mast cells) were taken and centrifuged at 800 rpm for 10 minutes. Pellets and supernatants were taken for determination of specific histamine release as previously described.15
Reverse transcriptase-polymerase chain reaction (RT-PCR) analysis of mast cell protease expression.
Total RNA was isolated from mast cells using Ultraspec RNA (Biotecx Laboratories Inc, Houston, TX) and treated with 1 U of Heparinase I (Sigma Chemical Co, St Louis, MO) per microgram RNA and 40 U of RNase inhibitor (Sigma Chemical Co) at 25°C for 2 hours. One microgram of total RNA from each sample was reverse transcribed with oligo dT and then 1/20 of this product was amplified with 100 pmol of primers for individual mast cell proteases in 50 μL of PCR Supermix (GIBCO-BRL, Grand Island, NY). The primer sequences for murine mast cell protease 2 (mMCP2), murine mast cell protease 4 (mMCP4), and murine mast cell carboxypeptidase A (mMCCPA) were as follows: 5′ primers: 5′-GTGATGACT GCTGCACACTG, 5′-GTAATTCCTCTGCCTCGTCCT, 5′-ACACAGGATCGAATG TGGAG; 3′ primers: 5′-CTTGAAGAGTCTGACTCAGG, 5′-ACCCAGGGTTAT CAGAGCTC, 5′-TAATGCAGGACTTCATGAGC, respectively. PCR was performed as follows: 94°C, 3 minutes; 30 cycles of 94°C, 1 minute; 52°C, 2 minutes; 72°C, 2 minutes; 72°C, 8 minutes. The PCR products were identified by electrophoresis in a 1.5% agarose gel and verified by molecular size.
Isolation of human eosinophils and purification of MBP.
Human eosinophils and eosinophil granule proteins were isolated as previously described.16 Briefly, whole blood was obtained from healthy and hypereosinophilic donors in the Clinical Research Center at the Beth Israel Deaconess Medical Center (Boston, MA). Venipuncture procedures were approved by and performed within the guidelines of the Institutional Care and Use Committee at the Beth Israel Deaconess Medical Center. The buffy coat was collected and separated by density centrifugation in Ficoll-Paque (Pharmacia, Piscataway, NJ). Red blood cells were lysed in NH4Cl, and the remaining granulocytes were washed in phosphate-buffered saline (PBS) with 2% fetal bovine serum, and counted. Eosinophils were then isolated by negative selection using the magnetic cell separation system.17 The granulocytes were incubated with CD 16 antibodies conjugated to magnetic particles (50 μL of particles per 5 × 107cells) for 30 minutes at 6°C, then passed through a separation syringe containing iron fibers. Eosinophil number and purity were assessed by Randolph's stain with eosinophil preparations of greater than 95% purity typically obtained. The eosinophils were lysed in hypertonic sucrose (0.25 mol/L) and centrifuged at 400g for 10 minutes. The cellular lysates were then centrifuged at 13,000gfor 20 minutes to pellet eosinophil granules. The granules were lysed in 10 mmol/L HCl and the lysates were fractionated by column chromatography using Sephadex G-50 (Pharmacia) equilibrated with 0.025 mol/L acetate buffer (pH 4.3), 0.15 mol/L NaCl. MBP was identified by its distinctive chromatographic filtration pattern. The purity of MBP was assessed by protein gel electrophoresis and the concentration determined by spectrophotometric absorbance at 277 nm.16
The effects of MBP or poly-L-arginine on the release of3H-5-hydroxytryptamine from murine mast cells.
The effect of either MBP or poly-L-arginine, a highly charged synthetic analog of eosinophil-derived granule proteins,18 on mast cell mediator release was determined using a3H-5-hydroxytryptamine (3H-5HT) release assay, as previously described.19 Briefly, mast cells were incubated with 1 mCi/mL of 5[1,2-3H](N)-hydroxytryptamine creatine sulfate (NEN Dupont, Boston, MA) for 2 hours. The cells were washed three times and then challenged with their respective media alone (unstimulated) or different concentrations of either MBP or poly-L-arginine (Sigma Chemical Co) for 10 minutes at 37°C. Each mast cell population was maintained in its respective medium throughout the incubation, wash, and challenge periods. The cells were then centrifuged at 1,000 rpm for 10 minutes. Radioactive counts were measured in the supernatants and pellets, and the percentage of specific 3H-5-HT release was calculated as follows: % Specific Release = 100 × [(cpm Stimulated Supernatants/cpm Stimulated Cells + Stimulated Supernatants) − (cpm Unstimulated Supernatants/cpm Unstimulated Cells + Unstimulated Supernatants)]. In some experiments after3H-5HT incubation, mast cells were centrifuged and radioactive counts determined as a measure of total serotonin uptake.
Effects of MBP, poly-L-arginine, or IgE/antigen on arachidonic acid mediator release from BMCMC in SCF.
We also examined the effects of MBP or poly-L-arginine on the release of 14C-labeled arachidonic acid from BMCMC in SCF as previously described.20 The mast cells were incubated with14C arachidonic acid (1 mCi/mL) for 12 hours at 37°C. In some experiments, murine monoclonal IgE antidinitrophenyl (DNP) antibodies (3 μg/mL) were added for the final 2 hours of incubation. The cells were washed three times, then incubated with different concentrations of either MBP, poly-arginine, 20 ng/mL DNP30-40-human serum albumin (Sigma Chemical Co), or calcium ionophore (Sigma Chemical Co) for 10 minutes. Radioactive counts were measured in the supernatants and pellets and the percent specific release of 14C-labeled arachidonic acid was calculated as follows: % Specific Release = 100 × [(cpm Stimulated Supernatants/cpm Stimulated Cells + Stimulated Supernatants) − (cpm Unstimulated Supernatants/cpm Unstimulated Cells + Unstimulated Supernatants)].
Effects of MBP or poly-L-arginine on the production of TNF-α by mast cells.
Mast cells were incubated with medium alone or different concentrations of poly-L-arginine or MBP for 30 minutes at 37°C. The supernatants were collected and immediately stored at −80°C. Immunoreactive TNF-α protein was measured by enzyme-linked immunosorbent assay (ELISA) (Endogen, Woburn, MA).
Statistical analysis.
The results of differences in TNF-α production between the MBP or poly-L-arginine and unstimulated cells or specific3H-5HT release from different mast cell populations were analyzed for statistical significance (defined as P < .05) by the Student's t-test (two-tailed).
RESULTS
BMCMC in SCF exhibit positive staining with safranin, mCCPA, mMCP-2, and mMCP-4 protease expression, increased histamine content, and increased uptake of 3H-serotonin.
We initially compared the phenotypic characteristics of BMCMC grown in SCF as the only exogenous growth factor (BMCMC in SCF) with BMCMC derived in Concanavalin A-stimulated spleen conditioned medium, a source of multiple growth factors (BMCMC in CM), or a cloned, growth factor–independent mast cell line, Cl.MC/C57.1. We used a histochemical approach for defining mast cell heterogeneity by comparing the staining characteristics with alcian blue and safranin.21 Such an approach can identify mucosal-type mast cells by their cytoplasmic staining with alcian blue and their absence of cytoplasmic staining with safranin, while connective tissue-type mast cells exhibited strong cytoplasmic staining with safranin. We found that BMCMC in CM (Fig 1A) or conditioned medium and Cl.MC/C57.1 mast cells (not shown) exhibited cytoplasmic staining with alcian blue, but failed to stain with safranin. In contrast, BMCMC in SCF exhibited heterogeneous cytoplasmic staining characteristics (Fig 1B). The majority of cells exhibited mixed alcian blue and safranin staining of their cytoplasmic granules (55% ± 6% of total mast cells), while cells with either predominantly alcian blue cytoplasmic staining cells or predominantly safranin staining accounted for the remainder of the cells (25% ± 5% and 20% ± 4%, respectively).
Histochemical staining properties of different populations of mast cells in vitro. Cytocentrifuge preparations of BMCMC grown in 20% Concanavalin A stimulated spleen conditioned medium (BMCMC in CM, [A]) or BMCMC grown in 50 ng/mL of SCF (BMCMC in SCF, [B]) were stained with alcian blue and safranin O. Photomicrographs were taken at 400× magnification. White arrow denotes alcian blue positive (safranin negative) mast cell, black arrow denotes safranin positive mast cell (alcian blue negative), and black arrowhead denotes mast cells with mixed alcian blue and safranin positive granules. The growth factor independent cloned mast cell line, Cl.MC/C57.1 exhibited similar staining characteristics as BMCMC in CM (data not shown).
Histochemical staining properties of different populations of mast cells in vitro. Cytocentrifuge preparations of BMCMC grown in 20% Concanavalin A stimulated spleen conditioned medium (BMCMC in CM, [A]) or BMCMC grown in 50 ng/mL of SCF (BMCMC in SCF, [B]) were stained with alcian blue and safranin O. Photomicrographs were taken at 400× magnification. White arrow denotes alcian blue positive (safranin negative) mast cell, black arrow denotes safranin positive mast cell (alcian blue negative), and black arrowhead denotes mast cells with mixed alcian blue and safranin positive granules. The growth factor independent cloned mast cell line, Cl.MC/C57.1 exhibited similar staining characteristics as BMCMC in CM (data not shown).
Mast cell heterogeneity has also been defined by the expression of various neutral proteases.22 Therefore, we examined whether the differences in histochemical staining characteristics between BMCMC in SCF and BMCMC in CM were accompanied by changes in protease gene expression. We found that BMCMC in CM expressed mCCPA and mMCP-2, but not mMCP-4 (Fig 2A), consistent with previous reports.23 24 In contrast, BMCMC in SCF expressed mCCPA, mMCP-2, and mMCP-4 (Fig 2A). Thus, in addition to the changes in histochemical staining properties, BMCMC in SCF also exhibited a change in protease gene expression.
Mast cell protease expression, histamine content, and uptake of 3H-5HT in different populations of mast cells in vitro. RT-PCR analysis of mast cell protease expression was performed on BMCMC in CM and BMCMC in SCF for the murine mast cell proteases (mMCP)-2 and -4, and the exopeptidase carboxypeptidase A (mMCCPA) (A). Total histamine content of Cl.MC/C57.1, BMCMC in CM, and BMCMC in SCF was determined by fluorometric assay and expressed as pg of histamine per cell (B). The ability of the three mast cell populations to take up3H-5HT was assessed. Mast cells were incubated with3H-5HT for 2 hours, then washed. Total radioactive counts in the cell pellets were measured and expressed as cpm (C). Data in (B and C) are shown as mean ± SEM (n = 2 to 4/group). Similar results were obtained in eight repeat experiments.
Mast cell protease expression, histamine content, and uptake of 3H-5HT in different populations of mast cells in vitro. RT-PCR analysis of mast cell protease expression was performed on BMCMC in CM and BMCMC in SCF for the murine mast cell proteases (mMCP)-2 and -4, and the exopeptidase carboxypeptidase A (mMCCPA) (A). Total histamine content of Cl.MC/C57.1, BMCMC in CM, and BMCMC in SCF was determined by fluorometric assay and expressed as pg of histamine per cell (B). The ability of the three mast cell populations to take up3H-5HT was assessed. Mast cells were incubated with3H-5HT for 2 hours, then washed. Total radioactive counts in the cell pellets were measured and expressed as cpm (C). Data in (B and C) are shown as mean ± SEM (n = 2 to 4/group). Similar results were obtained in eight repeat experiments.
We also determined the histamine content of these mast cell populations and the ability of the different mast cell populations to take up exogenous 5-hydroxytryptamine (5-HT). BMCMC in CM and the Cl.MC/C57.1 line contained approximately 0.1 pg of histamine per cell, which is consistent with previous reports,10,25 but the BMCMC in SCF contained approximately 25 times more histamine on a per cell basis (Fig 2B). In addition, we found that BMCMC in SCF took up approximately 10 times the amount of 5-HT compared with either BMCMC in CM or the Cl.MC/C57.1 line (Fig 2C). The increased histamine content and uptake of exogenous 5-HT exhibited by BMCMC in SCF are consistent with a maturational/phenotypical change in the cells, as previously reported for BMCMC in CM that had been changed and maintained in medium with SCF as the only exogenous growth factor for 4 to 6 weeks.10
MBP induces 3H-5HT release from mouse mast cells grown in SCF.
We next examined the ability of eosinophil granule MBP to induce mast cell degranulation and preformed mediator release from the different mast cell populations. MBP elicited a dose-dependent release of3H-5HT from BMCMC in SCF, but had no effect on either the Cl.MC/C57.1 line, BMCMC in CM (Fig 3A), or BMCMC derived in WEHI-3-conditioned medium, a source of IL-3 (not shown). Cell viability, as determined by Trypan blue exclusion, was not significantly changed in BMCMC in SCF challenged with MBP compared with cells challenged with a similar dilution of column elution buffer (98% v 96%, respectively, at the highest concentration of MBP). Thus, MBP, at the concentrations used, had no inherent cytotoxic effects on any of the mast cell populations examined.
Effect of MBP on 3H-5HT release in different populations of murine mast cells. BMCMC grown in stem cell factor (BMCMC in SCF), BMCMC grown in Concanavalin A stimulated spleen conditioned medium (BMCMC in CM), or the growth factor independent cloned mast cell line, Cl.MC/C57.1, was incubated with3H-5HT for 2 hours, washed and then challenged with different concentrations of MBP (A) or poly-L-arginine (B) for 10 minutes. Specific 5-HT release was calculated as described in Materials and Methods. Results are expressed as mean ± SD (n = 3). ***P < .001 by the two-tailed paired Student'st-test. Similar results were obtained in a repeat experiment.
Effect of MBP on 3H-5HT release in different populations of murine mast cells. BMCMC grown in stem cell factor (BMCMC in SCF), BMCMC grown in Concanavalin A stimulated spleen conditioned medium (BMCMC in CM), or the growth factor independent cloned mast cell line, Cl.MC/C57.1, was incubated with3H-5HT for 2 hours, washed and then challenged with different concentrations of MBP (A) or poly-L-arginine (B) for 10 minutes. Specific 5-HT release was calculated as described in Materials and Methods. Results are expressed as mean ± SD (n = 3). ***P < .001 by the two-tailed paired Student'st-test. Similar results were obtained in a repeat experiment.
Poly-L-arginine stimulates the release of 3H-5HT from mouse mast cells.
MBP has a high content of arginine residues (15%) in its amino acid sequence that results in a highly charged cationic protein. Several laboratories have reported that some of the stimulatory effects of MBP are related to its charge and these effects can be imitated by poly-L-arginine, a highly charged cationic analog of MBP.26 27 Therefore, we examined the effect of poly-L-arginine on mast cell mediator release.
In contrast to the response with MBP, all three populations of murine mast cells examined released 3H-5HT after stimulation with poly-L-arginine (Fig 3B), and there was no adverse effect on mast cell viability (data not shown). The concentration of poly-L-arginine necessary to elicit 5-HT release was high (10−5mol/L) and there appeared to be a threshold effect in that almost no specific serotonin release occurred at lower concentrations. Nevertheless, this effect of poly-L-arginine was seen in each of the mast cell populations tested (Fig 3B).
MBP and poly-L-arginine stimulates the release of arachidonic acid metabolites and TNF-α from mast cells grown in SCF.
In addition to preformed mediators such as serotonin and histamine, immunologically stimulated murine mast cells can generate products of arachidonic acid metabolism and a number of multifunctional cytokines. Therefore, we investigated the effects of MBP on the release of newly synthesized arachidonic acid mediators and cytokine production. Like the histochemical and biochemical heterogeneity exhibited by mast cells, the pathways of arachidonic acid metabolism used by mast cells is characteristic of different mast cell populations. Mucosal mast cells use the lipoxygenase pathway and produce mainly leukotriene C4, while the cyclooxygenase pathway predominates in connective tissue-type mast cells, resulting in the production of prostaglandin D2.28To define whether arachidonic acid metabolites derived from either pathway were produced, we prelabeled BMCMC in SCF with14C-arachidonic acid and determined if MBP or poly-L-arginine elicited the release of arachidonic acid mediators. We found that MBP induced a dose-dependent release of14C-arachidonic acid from BMCMC in SCF (Fig 4). MBP was more effective than poly-L-arginine in eliciting 14C-arachidonic acid release (Fig 4). The release of 14C-arachidonic acid from BMCMC in SCF by MBP at 10-7 mol/L was similar to that seen when the cells were sensitized with IgE anti-DNP antibodies and challenged with 20 ng/mL of DNP30-40-human serum albumin (25.5% ± 0.2% specific release) or stimulated with calcium ionophore at 4 μmol/L (28.1% ± 2% specific release).
Effect of MBP or poly-L-arginine on 14C arachidonic acid release from BMCMC maintained in SCF. BMCMC grown in SCF (BMCMC in SCF) were incubated with 14C-arachidonic acid for 12 hours. BMCMC in SCF were incubated with different concentrations of MBP or poly-L-arginine for 10 minutes. The data are expressed as mean ± SD (n = 3). Specific arachidonic acid release was calculated as described in Materials and Methods.
Effect of MBP or poly-L-arginine on 14C arachidonic acid release from BMCMC maintained in SCF. BMCMC grown in SCF (BMCMC in SCF) were incubated with 14C-arachidonic acid for 12 hours. BMCMC in SCF were incubated with different concentrations of MBP or poly-L-arginine for 10 minutes. The data are expressed as mean ± SD (n = 3). Specific arachidonic acid release was calculated as described in Materials and Methods.
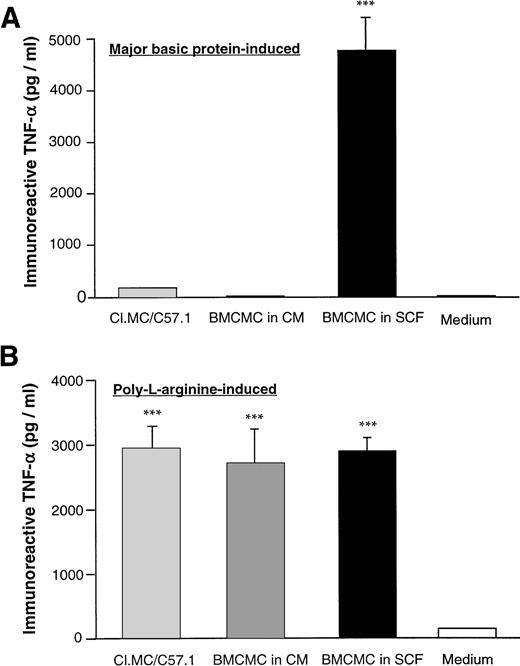
We also examined the effect of MBP or poly-L-arginine on the production of TNF-α in all three mast cell populations. We found that MBP induced significant TNF-α release from BMCMC in SCF, but had little effect on TNF-α production in either BMCMC in CM or the Cl.MC/C57.1 cell line (Fig 5A). In contrast, poly-L-arginine induced the production of TNF-α in all three mast cell populations (Fig 5B).
Production of TNF-α from different populations of mast cells stimulated with either MBP or poly-L-arginine. BMCMC in SCF, BMCMC in CM, or Cl.MC/C57.1 mast cells were stimulated with either MBP (10−6 mol/L) (A) or poly-L-arginine (10-5mol/L) (B) for 30 minutes. Supernatants were then collected and analyzed for TNF-α by ELISA. Cell viability was greater than 90% as determined by Trypan blue exclusion. The data are expressed as mean ± SD (n = 3). ***P < .05 by the two-tailed paired Student's t-test.
Production of TNF-α from different populations of mast cells stimulated with either MBP or poly-L-arginine. BMCMC in SCF, BMCMC in CM, or Cl.MC/C57.1 mast cells were stimulated with either MBP (10−6 mol/L) (A) or poly-L-arginine (10-5mol/L) (B) for 30 minutes. Supernatants were then collected and analyzed for TNF-α by ELISA. Cell viability was greater than 90% as determined by Trypan blue exclusion. The data are expressed as mean ± SD (n = 3). ***P < .05 by the two-tailed paired Student's t-test.
DISCUSSION
The mechanisms involved in mast cell activation by stimuli other than IgE antibodies have not been completely defined. We found that SCF is an important factor in the development of mast cell responsiveness to the eosinophil granule-associated protein MBP. Now, in addition to its role as a growth and survival factor for mast cells in vitro and in vivo,10,11 29 our study shows that SCF also influences mast cell/eosinophil interactions.
The population of mast cells derived in SCF from the bone marrow of BALB/c mice had several phenotypical characteristics of mature connective tissue-type mast cells. The mast cells exhibited positive staining for safranin, increased histamine content, increased uptake of 5-HT compared with the negative safranin staining, low histamine and 5-HT uptake typically seen in BMCMC in CM. These findings are similar to a previous report in which BMCMC in CM were transferred into medium containing SCF as the only exogenous growth factor.10 After 4 weeks in culture, the mast cells exhibited alterations in their phenotype similar to the BMCMC in SCF, but not identical to peritoneal mast cells.10 However, in this study, mast cell function was not examined.
As well, we found that BMCMC in SCF expressed the protease mMCP-4, which is typically found in connective tissue-type mast cells and not mucosal-type mast cells.30 31 Thus, phenotypically mature mast cells could be derived directly from mouse bone marrow cells using soluble SCF as the only exogenous growth factor. However, it is important to note that while these cells had characteristics similar to mature connective tissue-type mast cells, BMCMC in SCF represent a heterogeneous population of mast cells. Thus, BMCMC in SCF are similar, but not identical to connective tissue mast cells derived from the peritoneal cavity of mice.
We found that MBP elicited the release of a mast cell granule-associated mediator (serotonin) and newly synthesized mediators (arachidonic acid metabolites) from BMCMC in SCF. In addition, MBP induced significant TNF-α production by BMCMC in SCF. These responses did not occur in BMCMC in CM. Thus, in addition to the biochemical and morphological changes seen in BMCMC in SCF, the mast cells also underwent a functional change in their responsiveness to MBP. Ogasawara, et al32 reported that the coculture of BM-derived mast cells with Swiss 3T3 fibroblasts for 4 to 6 days resulted in the cells acquiring responsiveness to polycationic activators, such as compound 48/80 or substance P, but that short-term culture with soluble SCF (6 days) did not confer responsiveness to compound 48/80. We have found that BMCMC in CM that were switched into SCF-containing medium and maintained for 6 weeks acquired responsiveness to substance P also release mediators in response to challenge with substance P (manuscript in preparation). Thus, SCF either as a soluble dimer and, presumably in its membrane bound form associated with fibroblasts or smooth muscle cells, can alter mast cell responses to certain agents that activate mast cells.
MBP is thought to damage cell membranes33,34 and is a toxin to helminths and mammalian cells in vitro.35 But in our study, MBP did not affect mast cell viability. This is similar to other reports demonstrating noncytolytic mediator release from rodent mast cells and human basophils5,6 by MBP. It has been suggested that the effects of MBP are the result, at least in part, of the high cationic charge of MBP.26 However, cationic charge alone cannot explain the effects of MBP on mast cells. If cationic charge only was responsible for mast cell activation, then MBP would be predicted to elicit mediator release from each of the different mast cell populations examined. However, we found that MBP induced mediator release only from BMCMC in SCF and not BMCMC in CM or Cl.MC/C57.1 cells. This is in contrast to a synthetic MBP analog, poly-L-arginine, which elicited mediator release from all populations. This suggests that poly-L-arginine is not an ideal alternative to authentic native MBP for the study of MBP/mast cell interactions.
Previous studies have shown that MBP stimulates the preformed mediator release from connective tissue-type mast cells isolated from the peritoneal cavity of rats.5,6 Our findings show that SCF is an important factor in the development of mast cell responsiveness to MBP. At this time, it is not clear whether SCF induces the growth of a population of mast cells that respond to MBP or, alternatively, induces a direct change in MBP unresponsive mast cells that leads to responsiveness, such as by the induction of an as yet unidentified MBP receptor or receptor-like complex.36 37 We favor the former hypothesis, because BMCMC in SCF after 3 weeks in culture (mast cells represented >75% of the total cell population) did not release serotonin to challenge with MBP (data not shown). This suggests that SCF is not simply facilitating the action of MBP, but may, in fact, be driving the proliferation and differentiation of a population of mast cells that have the capacity to respond to MBP.
The findings of our study may have important implications regarding mast cell/eosinophil interactions in vivo. As previously discussed, SCF has important effects on mast cell growth and differentiation. The responsiveness of mast cell populations under the influence of SCF to eosinophil-derived granule proteins may be important in inflammatory and allergic reactions. For example, MBP-induced mast cell degranulation may lead to an amplification of the inflammatory response through the generation of preformed and newly synthesized mediators by mast cells. As well, granule release by mast cells may promote the downregulation of the inflammatory response by the release of heparin from connective tissue-type mast cells, which has been shown to neutralize the biological activity of MBP.38 39 Thus, the ability of mast cells to release mediators in response to MBP may have multiple, complex biological consequences.
In conclusion, our study shows that BMCMC in SCF express phenotypic characteristics similar to mature connective tissue mast cells as defined by histochemical staining, protease expression, histamine content, and 5-HT uptake. BMCMC in SCF release preformed 5-HT, products of arachidonic acid metabolism, and TNF-α in response to MBP. These findings suggest that SCF is an important factor in the development of a functional response of mast cells to eosinophil-derived granule MBP.
ACKNOWLEDGMENT
We thank Dr Keith E. Langley of AMGEN, Inc (Thousand Oaks, CA) for providing rrSCF164.
Supported by National Institutes of Health Grants No. DK33506 and Core B (Morphology), DK40561 (Clinical Nutrition Research Center at Harvard), DK46819 (to B.K.W.) and AI25230 (to S.J.A.), and a Career Development Award from the Crohn's and Colitis Foundation of America (to G.T.F.).
Address reprint requests to Barry K. Wershil, MD, Combined Program in Pediatric Gastroenterology and Nutrition, Massachusetts General Hospital, 149 13th St (1493404), Charlestown, MA 02129.
The publication costs of this article were defrayed in part by page charge payment. This article must therefore be hereby marked "advertisement" is accordance with 18 U.S.C. section 1734 solely to indicate this fact.

![Fig. 1. Histochemical staining properties of different populations of mast cells in vitro. Cytocentrifuge preparations of BMCMC grown in 20% Concanavalin A stimulated spleen conditioned medium (BMCMC in CM, [A]) or BMCMC grown in 50 ng/mL of SCF (BMCMC in SCF, [B]) were stained with alcian blue and safranin O. Photomicrographs were taken at 400× magnification. White arrow denotes alcian blue positive (safranin negative) mast cell, black arrow denotes safranin positive mast cell (alcian blue negative), and black arrowhead denotes mast cells with mixed alcian blue and safranin positive granules. The growth factor independent cloned mast cell line, Cl.MC/C57.1 exhibited similar staining characteristics as BMCMC in CM (data not shown).](https://ash.silverchair-cdn.com/ash/content_public/journal/blood/92/3/10.1182_blood.v92.3.1055/5/m_blod415fur01z.jpeg?Expires=1769207006&Signature=v~VJ1sa1K7gMd8rnaoiBDMvBd4b7E9P-A2ld0v8Rjb-AVmUw3hgM-f6q08AAOk1v2mYFjVJaBVQ-B5gTzWUEwVIHwtA3KRYb3afnTsyMRJpgc9QAiZ07vZnJPCjhIlKX0eeyigczOxq~cujbZQsh0IU6cSlTt6A~bawd9P6NnW4DkCSL4UM83ggyUjNCHtZZ~gU43sdMCZKLWVpU~s6Zt8JdR7SGY0-n2MtFOK465OkXVUPd9ic4oVaKBYfbG9UG9uEZ0XGVNuoBh~twA7h5mYva-~e1xv7oz4UNeOXck7MUrg13Y-xn2axtql79BSL2jmgQaZzgKhOjI8lxz0no5Q__&Key-Pair-Id=APKAIE5G5CRDK6RD3PGA)