Abstract
Eotaxin is a potent chemoattractant for eosinophils during inflammation and allergic reactions in the adult, but its role in the embryonic development of the hematopoietic system has not been examined. We report here that eotaxin and its receptor, CCR-3, are expressed by embryonic tissues responsible for blood development, such as fetal liver (FL), yolk sac (YS), and peripheral blood. We found that eotaxin acts synergistically with stem cell factor to accelerate the differentiation of embryonic mast cell progenitors, and this response can be suppressed by pertussis toxin, an inhibitor of chemokine-induced signaling through Gi protein and chemotaxis. Eotaxin promotes the differentiation of fetal mast cell progenitors into differentiated mast cells as defined by the expression of mast cell specific proteases. Furthermore, in combination with stem cell factor (SCF), it promotes the growth of Mac-1+myeloid cells from embryonic progenitors. These studies suggest that eotaxin may be involved in the growth of granulocytic progenitors and the differentiation and/or function of mast cells during embryogenesis and/or pathological conditions that induce high levels of eotaxin, such as allergic responses.
© 1998 by The American Society of Hematology.
CHEMOKINES PLAY AN important role in the regulation of leukocyte trafficking and migration.1,2 In most instances, the expression and function of chemokines has been studied in adult mice during inflammatory responses. However, the ability of chemokines to influence cell migration suggests that these molecules may also play a role in embryonic development. For example, hematopoietic progenitor cells must be recruited from early, transient sites of blood development, such as the fetal liver (FL) and yolk sac (YS), to more definitive, adult sites of hematopoiesis, like the bone marrow and spleen.3 While this developmental shift in the site of hematopoiesis has been well documented, little is known about the molecular signals that direct cellular trafficking in the developing embryo.
Hematopoietic stem cells in mice were thought to originate in the YS. However, recent evidence derived from studies in other species suggests that the earliest precursors are located within the body of the embryo proper.4-7 For example, chimeras constructed with chick YS and quail bodies showed that yolk sac-derived cells populated the organism only transiently, while long-term hematopoietic progeny were derived from within the body of the developing embryo.6These progeny appeared to colonize tissues over relatively short distances suggesting that secreted chemoattractants may be involved. In mice, hematopoiesis involves the formation of “blood islands” in the extraembryonic mesoderm of the visceral YS at 7.5 days postcoital (dpc).8 Hematopoietic progenitors begin to fill these islands at 7 to 8 dpc, before the circulatory systems and extraembryonic tissues of the embryo merge (at 8.5 dpc), and give rise to primitive nucleated red blood cells (RBC). However, the greatest number of definitive stem cells in the mouse were found in the aorta-gonad-mesonephros (AGM) region of the embryonic hindgut.9-11 The appearance of intraembryonic stem cell activity in avian tissue was found to coincide with the clustering of hematopoietic cells in the AGM region, implying that the AGM region was the source of the stem cells.12 The factors controlling the migration of these stem cells to extramedullary sites of hematopoiesis are currently being investigated.
Several recent studies provide evidence that chemokines play a role in hematopoietic development. The chemoattractant stromal-derived factor-1 (SDF-1) has been shown to induce the migration of CD34+progenitors isolated from cord blood.13 Furthermore, SDF-1–deficient mice exhibit defective myelopoiesis in the fetal bone marrow and defective B-cell development in the fetal liver and bone marrow.14 Another chemokine that influences hematopoiesis is macrophage inflammatory protein-1α (MIP-1α ), which has been shown to inhibit colony formation by burst-forming unit-erythroid (BFU-E), but not by colony-forming unit-erythroid (CFU-E) in a methylcellulose culture of human CD34+ bone marrow cells.15 Finally, during development, fetal rabbit tonsils are seeded by primitive cells that are thought to be blood-borne, suggesting that mechanisms and signals, such as chemokines, that control cellular trafficking and diapedesis exist in utero.16
Our group and others have identified and characterized eotaxin, a potent and specific chemoattractant for eosinophils during inflammation.17 18 Given the evidence that chemokines may participate in hematopoietic development, we examined the expression and function of eotaxin in progenitor cell proliferation and differentiation. We now report that eotaxin is expressed at sites of hematopoiesis in the embryo and that eotaxin, in combination with stem cell factor (SCF), promotes the growth and differentiation of mast cell progenitors.
MATERIALS AND METHODS
Mice and Tissue Collection
Male and female C57BL/6 mice were placed in cages and the next morning plugged mice were considered at day 0 of gestation. A minimum of two pregnant mice were killed for each experiment by CO2narcosis. Embryos were isolated under a dissecting microscope and bled via the umbilicus until pale. Samples of YS, FL, and fetal blood (FBL) were collected on ice in Ultraculture medium (GIBCO BRL, Grand Island, NY) containing penicillin (50 U/mL) and streptomycin (50 mg/mL). YS and FL suspensions were prepared by gentle mechanical disruption through a 70-μm sieve (Fisher Scientific, Pittsburgh, PA) and viability determined by trypan blue exclusion. The number of embryos used per experiment ranged from 11 to 39 and individual organs from the different embryos were pooled. These studies were approved and performed in accordance with the guidelines of the Institutional Animal Care and Use Committee at the Center for Blood Research and Harvard Medical School, Boston, MA.
Colony Growth and In Vitro Chemotaxis Assays
The ability of eotaxin to stimulate colony growth from embryonic tissues was assessed in methylcellulose cultures.13 FL cells (1 × 104), cells from fetal YS (5 × 104), or blood cells (5 × 104) were plated in methylcellulose (0.9%) containing 20 ng/mL of murine recombinant SCF (PeproTech, Inc, Rocky Hill, NJ) and/or 50 ng/mL of eotaxin (PeproTech, Inc). In some experiments,pertussis toxin (100 ng/mL) was added in addition to SCF and eoxtaxin. Chemokine receptors are seven-transmembrane-spanning, G-protein–linked molecules whose signaling is affected bypertussis toxin.2 Colonies of greater than 50 cells were counted between 9 to 13 days or at day 18. Controls included methylcellulose cultures seeded with maternal peripheral white blood cells (5 × 104/plate) and cultures of fetal cells grown in the absence of SCF or eotaxin. No scorable colonies were induced in the absence of SCF or eotaxin, and the frequency of colonies induced from maternal white blood cells is ≈1 colony/80,000 cells. Cell migration was assessed in 24-well chemotaxis chambers (Corning Costar, Cambridge, MA). A total of 5 × 105 cells were placed in the upper chamber and tested for chemoattraction to media alone (negative control), MS-5 supernatant (undiluted), or eotaxin at 100 ng/mL. The chambers were incubated for 3 hours at 37°C. Cells migrating into the lower chamber were assessed on cytocentrifuge preparations and in methylcellulose culture containing interleukin (IL)-3 (10% vol), SCF (20 ng/mL), and human recombinant erythropoietin (Epoetin Alfa Procrit, 4.3 U/mL; Amgen, Inc, Thousand Oaks, CA). In the case of FBL cells, nucleated erythrocytes could not be excluded from cell counts by microscopic inspection.
Supernatant was harvested from the murine stromal cell line MS-519 for use as a positive control in chemotaxis assays. The adherent line was grown to subconfluence in Iscove’s modified Dulbecco’s medium (IMDM) containing 10% fetal calf serum (GIBCO), then switched to serum-free Ultraculture medium. The serum-free medium was collected after 48 hours, spun, filter-sterilized, and frozen in aliquots at −20°C.
For replating experiments, individual colonies were collected under a dissecting microscope using a flame-drawn pasteur pipette. Pooled cells were washed once with UltraCULTURE medium and divided into two pools, one with 10% of the cells and a second fraction containing 90%. Each pool was plated into methylcellulose containing erythropoietin, IL-3, and SCF, as described above. Colonies were counted after 10 days in culture.
Growth of Bone Marrow Cultured Mast Cells
Femoral bone marrow cells were obtained from BALB/c mice and maintained in Dulbecco’s modified Eagle’s medium (GIBCO) with 10% heat-inactivated fetal calf serum (Intergen, Purchase, NY), 50 mmol/L 2-mercaptoethanol, and 2 mmol/L L-glutamine at 37°C in 5% CO2 and supplemented with either concanavalin A-stimulated mouse spleen cell-conditioned medium (CM), as a source of IL-3 as previously described,20 or SCF (50 ng/mL). After 4 to 6 weeks, mast cells represented >98% of the total cells as determined by neutral red staining.
Identification of Eotaxin RNA in Tissues by In Situ Hybridization
Eotaxin mRNA expression in embryonic tissues was examined by in situ hybridization, as previously described.17 A 265-kb antisense RNA probe lacking the most conserved domains of the CC chemokine coding regions was prepared by T7 transcription of a Sac 1-digest of a murine eotaxin cDNA obtained by polymerase chain reaction (PCR) and subcloned into pBluescript. A sense probe was generated by SP6 transcription of an EcoRV-digest of the subclone. The probes were labeled with digoxigenin-uridine triphosphate (UTP) (Boehringer-Mannheim, Indianapolis, IN) and visualized by immunohistochemistry using antidigoxigenin antibodies coupled to alkaline phosphatase. Slides were counterstained with neutral red.
Histochemical Staining
Cytocentrifuge preparations were fixed in ice cold acetone for 10 minutes, then air dried. Slides were treated with the substrate for chloroacetate esterase, as described,21 1% toluidine blue in methanol for 5 minutes, or modified Giesma (1:20 dilution) for 60 minutes.
Immunoperoxidase Staining
Whole embryos were fixed in 10% formalin overnight, then dehydrated in serial concentrations of ethanol, embedded in paraffin, and 4-μm sections prepared. The slides were baked at 60°C for 1 hour to deparaffinize, washed in xylene, hydrated through serial concentrations of ethanol, then washed in phosphate-buffered saline (PBS). The slides were treated for 25 minutes with 0.1% trypsin in 0.1% calcium chloride, then preincubated with 10% fetal calf serum in PBS for 20 minutes. They were then incubated overnight at 4°C with a 1:10 dilution of monoclonal rat antimouse eotaxin antibodies. Endogenous peroxidase activity was quenched with 0.6% hydrogen peroxide in methanol and endogenous biotin activity blocked (Vector, Burlingame, CA) Biotinylated mouse antirat-IgG antibodies (Dako, Carpinteria, CA) were applied for 1 hour at room temperature (RT), the slides were washed, and then incubated with a complex of streptavidin-biotin for 30 minutes. Antibody deposition was visualized by incubation with diaminobenzidine and hydrogen peroxide (Vector) for 2 to 4 minutes, and the slides were subsequently washed in PBS and counterstained in hematoxylin. Immunoperoxidase staining for c-kit was performed using the anti–c-kit antibody, ACK2, at 5 mg/mL.22 Frozen sections (5 μm) were stained with anti-CD3423 (undiluted supernatant) and antiendoglin (10 μg/mL).24
Immunofluorescent Analysis
Cells in methylcellulose were collected in calcium/magnesium-free PBS containing 5 mmol/L EDTA, washed in PBS containing 0.1% bovine serum albumin and 0.1% sodium azide, and incubated for 15 minutes on ice with a monoclonal antibody to CD16/CD32(FcγIII/II receptor; Pharmingen, San Diego, CA). After three washes, they were incubated with fluorescein isothiocyanate- or phycoerythrin-conjugated monoclonal rat IgG antibodies (Pharmingen) for 30 minutes on ice, and stained cells were analyzed on a FACSsort flow cytometer (Becton Dickinson, Carpenteria, CA).
Reverse Transcriptase-Polymerase Chain Reaction
Total RNA was prepared from fetal organs using the lithium chloride method.25 Mast cell protease mRNA expression was examined in colonies harvested on day 10 or 11 of culture. The cells were washed and suspended in 20 to 40 μL of di-ethyl pyrocarbonate (DEPC)-treated water and 40 U of RNase inhibitor was added. The pellet was boiled for 5 minutes, spun, and the supernatant was snap frozen for use in reverse transcriptase-polymerase chain reaction (RT-PCR) assays. RT-PCR was performed as previously described.17 The primer sequences for CCR-326 are GACAGCTTTGGAGAGTTTTCCTGCAGTCCTCGCTAT (5′) and AATATATTTTCCCAGGTAAACTGCCACATTTCT (3′). To confirm the specificity of the CCR-3 primers, PCR product from several embryonic tissues was sequenced. As a control, RNA samples were subjected to the same cycling cοnditions used to generate cDNA, except RT was absent from the reaction. No PCR-amplified product was found, except for a faint band that was present in yolk sac at 11 dpc, which was much weaker than that found in YS amplified appropriately (data not shown). Primer sequences for the serine proteases are: (1) mouse mast cell-carboxypeptidase A (mMC-CPA): ACACAGGATCGAATGTGGAG (5′) and TAATGCAGGACTTCATGAGC (3′); (2) mouse mast cell protease (mMCP)-2: GTGATGACTGCTGCACACTG (5′) and CTTGAAGAGTCTGACTCAGG (3′); (3) mMCP-4: GTAATTCCTCTGCCTCGTCCT (5′) and ACCCAGGGTTATCAGAGCTC (3′).
RESULTS
Eotaxin mRNA is expressed in cells lining the primitive vessels of the YS.
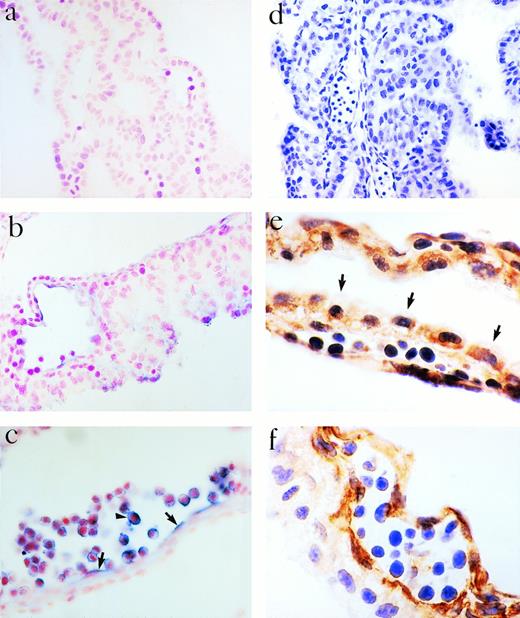
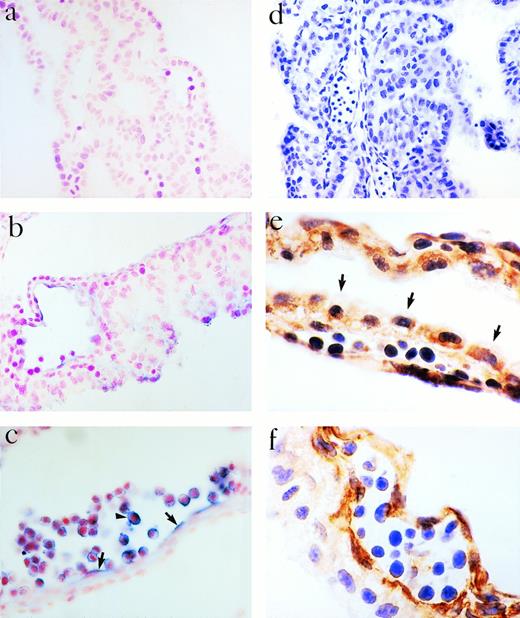
The YS is considered an important site of blood formation and/or vascularization in the embryo. We therefore examined the expression of eotaxin in the embryonic YS of mice using RNA in situ hybridization, RT-PCR, and immunoperoxidase staining. Cells lining primitive vessels within the YS at 10.5 dpc expressed eotaxin mRNA (Fig 1b and c) by in situ hybridization. These cells also expressed the endothelial markers CD34 (Fig 1f) and endoglin (data not shown), confirming that they represent endothelium.23,24 Cells within the lumen of the transected vessels also expressed eotaxin mRNA and the CD34 protein antigen (Fig1c and f). The murine YS is composed of mesoderm and visceral endoderm cells.27 By 9.5 dpc, epithelial-like cells arise from the endoderm and primitive endothelium arises from the mesoderm. The visceral layer of the YS is thought to be crucial for the differentiation of mesodermally-derived cells into blood cells.27 We found that the visceral (epithelial) layer was negative for CD34, but positive for eotaxin by immunostaining (Fig 1e). The expression of eotaxin in the visceral layer appeared polarized with more staining seen along the edge of the cells oriented toward the lumen of the vessels.
Expression of eotaxin in YS at 10.5 dpc. Eotaxin mRNA was assessed by in situ hybridization using a sense probe (a) or antisense probe (b and c). The endothelium (arrows, c) and cells in the vessel lumen (arrowhead, c) are positive. Immunoperoxidase staining was performed with a control antibody (d), antieotaxin MoAb (e), or a MoAb to CD34 (f). The visceral layer of cells, the endothelium, and primitive blood cells express eotaxin (e). In (e), the arrows identify the edge of the visceral cell layer. Magnifications are 100× (d), 400× (a, c, and f), and 1,000× (b and e).
Expression of eotaxin in YS at 10.5 dpc. Eotaxin mRNA was assessed by in situ hybridization using a sense probe (a) or antisense probe (b and c). The endothelium (arrows, c) and cells in the vessel lumen (arrowhead, c) are positive. Immunoperoxidase staining was performed with a control antibody (d), antieotaxin MoAb (e), or a MoAb to CD34 (f). The visceral layer of cells, the endothelium, and primitive blood cells express eotaxin (e). In (e), the arrows identify the edge of the visceral cell layer. Magnifications are 100× (d), 400× (a, c, and f), and 1,000× (b and e).
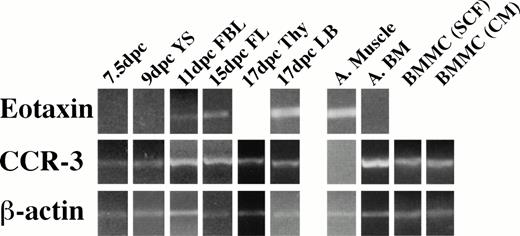
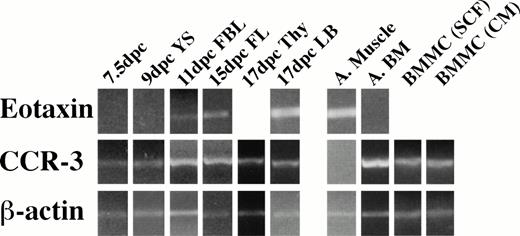
We next examined the expression of eotaxin and its receptor CCR-3 in embryonic tissues by RT-PCR. Eotaxin mRNA was not detectable in YS at day 9 dpc, and CCR-3 was detected weakly at 7.5 dpc in the embryo and at 9 dpc in the YS (Fig 2). Embryos at day 8.5 did not express eotaxin by immunoperoxidase staining (data not shown), but eotaxin and CCR-3 mRNA could be detected in all tissues tested at 11 dpc and thereafter. We were not able to amplify eotaxin and CCR-3 from RNA samples representing AGM harvested at 10 to 11 dpc.
Eotaxin and CCR-3 mRNA expression in embryonic tissues. RT products were prepared from RNA isolated from frozen tissues. Briefly, 1 μg of total RNA was reverse transcribed, and equalized amounts of the cDNA generated were amplified with β-actin (20 cycles), eotaxin (30 cycles), and CCR-3 (32 cycles) primers to generate fragments of 348, 108, and 454 bp, respectively. Adult tissues (A) were used as a control.
Eotaxin and CCR-3 mRNA expression in embryonic tissues. RT products were prepared from RNA isolated from frozen tissues. Briefly, 1 μg of total RNA was reverse transcribed, and equalized amounts of the cDNA generated were amplified with β-actin (20 cycles), eotaxin (30 cycles), and CCR-3 (32 cycles) primers to generate fragments of 348, 108, and 454 bp, respectively. Adult tissues (A) were used as a control.
Eotaxin and SCF synergistically induce colony formation from fetal hematopoietic tissues.
Given the expression of eotaxin in fetal hematopoietic tissues, we next asked whether eotaxin could induce the specific migration of progenitors isolated from YS and FBL using a chemotaxis chamber (Fig 3).28 The migratory (output) population was quantitated by colony-forming assays using cytokines that support the growth of erythroid and myeloid progenitors, but not lymphoid or megakaryocytic cells. No progenitors with colony-forming ability migrated to eotaxin in multiple experiments using fetal cells from gestational days 11-15 (Fig 3a). As a positive control, we found strong chemotaxis of fetal liver progenitors to the stromal cell supernatant, MS-5 (Fig 3a). MS-5 supernatant contains the progenitor chemokine SDF-1α,13 but it may also contain other chemokines that are capable of inducing the migration of progenitor cells. We are currently investigating which factors within the MS-5 supernatant induce migration of progenitors. In this study, the MS-5 supernatant was used solely to ensure that embryonic cells were capable of migrating through a porous membrane when presented with a proper stimulus.
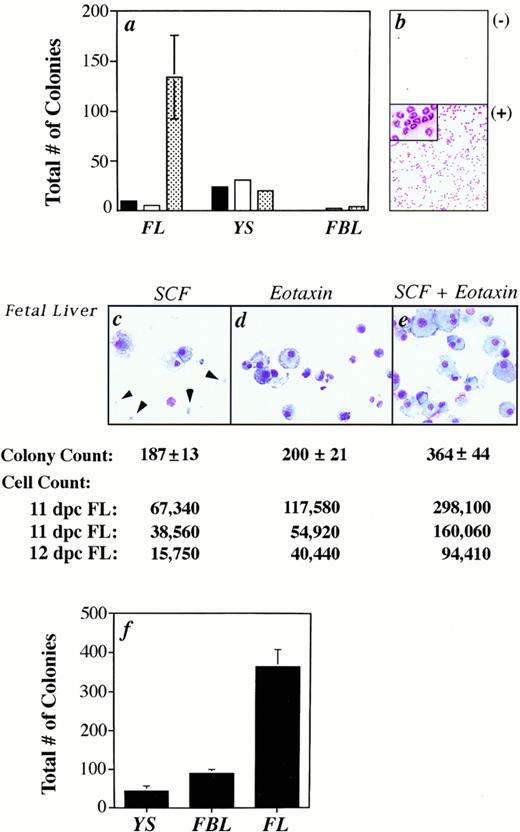
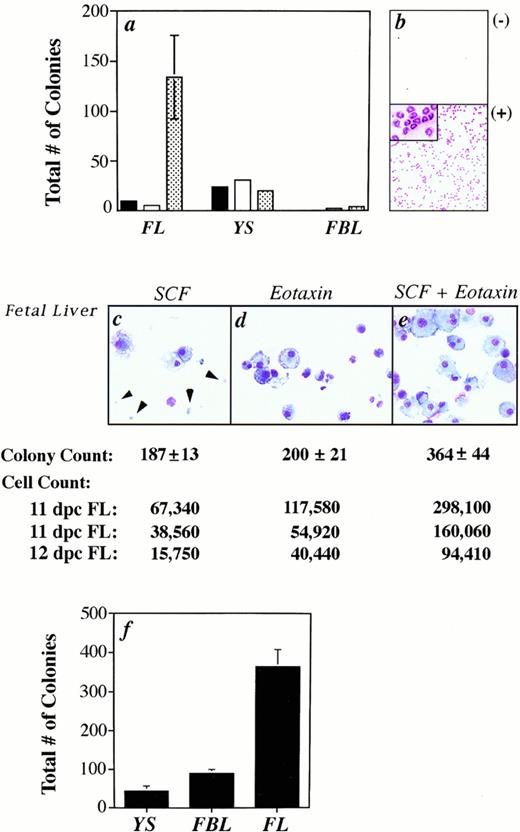
Eotaxin acts synergistically with SCF to induce colony formation, but not migration. (a) The total number of colonies (myeloid and erythroid) formed from fetal cells that migrated to either media alone (black bar), eotaxin (white bar), or MS5 supernatant (patterned bar) is shown. Standard error of the mean was determined, but in some cases was too small to be seen in the graph. No colonies were produced from the FBL output population in response to media alone. (b) As a control, eotaxin induced the migration of peritoneal eosinophils from IL-5 transgenic mice. (c through e) The cytospins are of cells harvested from 11 dpc FL colonies. The arrows identify erythrocytes. The total number of colonies (from the same experiment as the cytospins) and cells induced (in three separate experiments) is shown. (f) The total number of colonies generated from 11 dpc YS, FBL, and FL cells grown with SCF plus eotaxin is plotted for one representative experiment.
Eotaxin acts synergistically with SCF to induce colony formation, but not migration. (a) The total number of colonies (myeloid and erythroid) formed from fetal cells that migrated to either media alone (black bar), eotaxin (white bar), or MS5 supernatant (patterned bar) is shown. Standard error of the mean was determined, but in some cases was too small to be seen in the graph. No colonies were produced from the FBL output population in response to media alone. (b) As a control, eotaxin induced the migration of peritoneal eosinophils from IL-5 transgenic mice. (c through e) The cytospins are of cells harvested from 11 dpc FL colonies. The arrows identify erythrocytes. The total number of colonies (from the same experiment as the cytospins) and cells induced (in three separate experiments) is shown. (f) The total number of colonies generated from 11 dpc YS, FBL, and FL cells grown with SCF plus eotaxin is plotted for one representative experiment.
Several studies have shown that SCF acts as a potent comitogen for progenitor cells when combined with other cytokines, including IL-3, IL-6, and IL-1,29 thus, we also examined the effect of SCF combined with eotaxin on hematopoietic colony formation. The receptor for SCF, c-kit, has been shown by others to be expressed on progenitor cells in YS30 and FL.31 We tested fetal tissues at 11 dpc for c-kit expression and found 35% of liver cells, 3.8% of YS cells, and 3.2% of FBL cells were positive.
A synergistic response in the total number of colonies induced by SCF combined with eotaxin was found with fetal liver (Fig 3c through e), while an additive effect was seen with YS (data not shown) and FBL (Fig 4). The synergistic effect of these factors was not always represented by the colony count, but was seen when the total number of cells produced in methylcellulose cultures were harvested and counted (Fig 3). In three experiments, we obtained cell counts that were 1.61, 1.67, and 1.68 times greater than the sum of (cells grown in the presence of eotaxin alone)+(cells grown in the presence of SCF alone). Pertussis toxin decreased the cytospun cell count by 52% (544 cells/25 fields v 265 cells withpertussis toxin) and the colony count by 30% (± 8.7%) for FBL cultures, in three experiments.
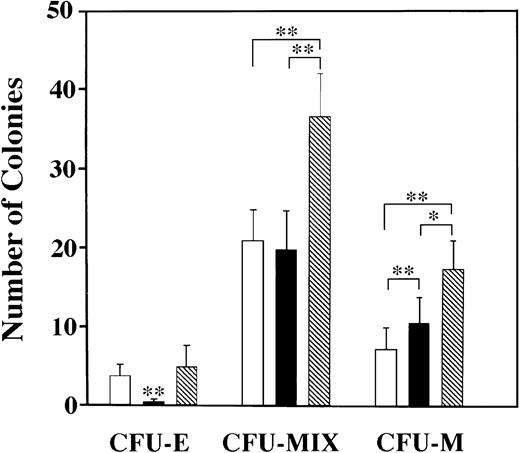
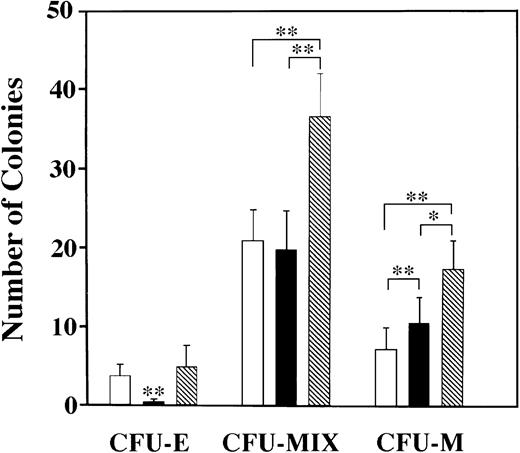
Myeloid colonies are predominantly induced by SCF combined with eotaxin. Three types of colonies were induced with SCF and/or eotaxin: CFU-E, CFU-MIX (containing cells of varying size, granulation, and nuclear shape/size, with and without RBCs) and CFU-M. Colony type was confirmed by preparing cytospun preparations from individual colonies with distinct morphological characteristics. Standard error bars are shown, and statistical analysis was performed using the Friedman test; ** = P < .001 and * = P< .01. (□) SCF; (▪) Eotaxin; (▧) SCF + Eotaxin.
Myeloid colonies are predominantly induced by SCF combined with eotaxin. Three types of colonies were induced with SCF and/or eotaxin: CFU-E, CFU-MIX (containing cells of varying size, granulation, and nuclear shape/size, with and without RBCs) and CFU-M. Colony type was confirmed by preparing cytospun preparations from individual colonies with distinct morphological characteristics. Standard error bars are shown, and statistical analysis was performed using the Friedman test; ** = P < .001 and * = P< .01. (□) SCF; (▪) Eotaxin; (▧) SCF + Eotaxin.
CFU-MIX are preferentially induced with SCF and eotaxin.
The type of colonies induced in our culture system was analyzed by preparing cytospun preparations of individual colonies with varying morphologies. We scored three types of colonies: CFU-E, CFU-MIX, and CFU-macrophage (CFU-M). CFU-MIX contained cells of varying morphology, including occasionally red-pigmented progeny, and the colonies were diffusely scattered.13 Cytospun preparations confirmed that colonies scored as CFU-MIX contained cells of varying size, nuclear shape, and granulation (data not shown), and the majority of cells were macrophages, mast cells, and immature, weakly-granulated cells. In Fig4, data from pooled experiments (n = 6) are shown for FBL at day 11. Similar patterns were obtained with FL and YS. We conclude that erythroid colonies are not induced by eotaxin alone in any tissue type. CFU-MIX are most prevalent and additively increase in number with the addition of eotaxin to SCF, as did CFU-M. The one exception to this pattern was seen with YS, where the numbers of CFU-M were similar for each growth condition tested. Statistical analysis was performed using the Friedman test for paired data. As stated above, synergistic increases in the total number of colonies and their size were seen for FL only (Fig 3c through e), which likely reflects the rapid cell proliferation in the liver at 11 dpc.
Phenotypic analysis of colonies grown with SCF and eotaxin.
The surface phenotype of the colony progeny produced was analyzed by direct immunofluorescent staining with lineage-specific monoclonal antibodies (MoAb) to myeloid cells (Mac-1; CD1132), RBC (TER-11933), and granulocytes (Gr-134; Table 1). In three of six experiments, there was a synergistic increase of 1.63 times (± 0.4) in the number of Mac-1+ cells from colonies grown in the presence of both factors. In every case, however, synergy was only seen when the source of progenitors for the assay was FBL. This might reflect differences in Mac-1 expression in progenitors from different hemaotopoietic sites, differential chemokine-induced adhesion molecule expression (Mac-1) in organ resident (FL, YS) versus circulating progenitors (FB), or simply limitations of the experimental system used here. Mononuclear cells of variable size were the predominant cells induced with SCF and/or eotaxin (see Fig 3c through e). The combined percentage of lineage-positive cells was always less than 100%, ranging from 15% to 51%. Thus, some of the lineage-negative cells may be progenitors or stromal cells. To test this hypothesis, we hand-pulled colonies of mixed morphology from 10-day old, YS-seeded cultures and replated these cells into methylcellulose containing IL-3, SCF, and erythropoietin. Multiple colonies arose from these replated cells with a plating efficiency of 19%, suggesting that undifferentiated progenitors can be maintained in our original cultures with SCF and eotaxin (data not shown). Significant numbers of YS and FBL-derived progeny were also positive for the SCF receptor,c-kit, a marker of progenitor cells29 and mast cells at all stages of differentiation (Table 1).
Our culture system does not support the growth of lymphoid cells, as few progeny induced by both factors expressed the B- and T-cell markers, B220, and Thy-1, respectively (Table 1).
Eotaxin influences mast cell development from embryonic progenitors.
When cytospun preparations of YS- and FBL-derived colonies induced with SCF alone, eotaxin alone, or both factors were closely examined, we identified a unique subpopulation of heavily granulated cells (Fig 5C). These granulated cells were not obtained from cultures grown in eotaxin alone or SCF alone at these concentrations. However, cultures of progenitor cells in SCF alone at concentrations of 50 ng/mL gave rise to cells with the same morphological characteristics (Fig 5B). These prominent cytoplasmic granules stained positively with toluidine blue (Fig 5D), a specific stain for mast cells and basophils.35 The cells were also positive for chloroacetate esterase, an enzyme found in cells of the granulocytic lineage, including mast cells, but not eosinophils or basophils.21 The number of granules per cell, cell size, and nuclear profile (both mononuclear and multinucleated cells were seen) were characteristic of mature mast cell populations derived in vitro.36
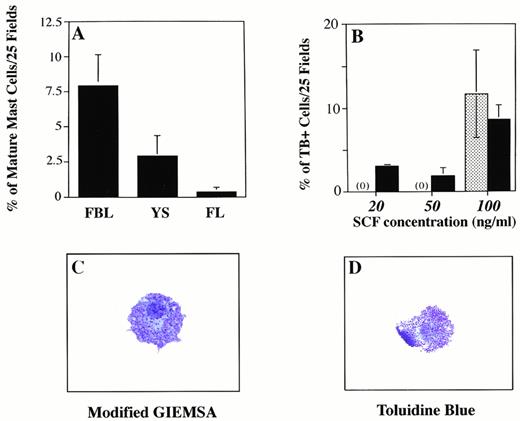
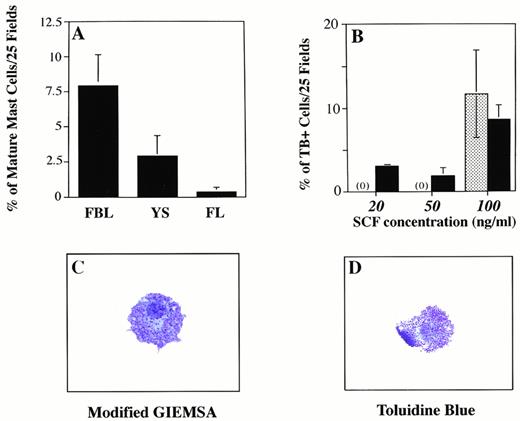
Eotaxin influences mast cell differentiation in FBL. Graph A shows the percentage of more differentiated mast cells identified on cytospins stained with modified giemsa (MG), based on granular content. The percentage of heavily-granulated cells (toluidine blue (TB)+, MG+, or CAE+) was determined by counting the number of these cells (in 25 or 100 fields at 400× magnification) on cytospun preparations made by harvesting all colony progeny induced in methylcellulose. Total cell counts ranged from 84 to 964. (B) Increased numbers of TB+ mast cells are induced as the concentration of SCF increases. The patterned bar represents the percentage of TB+ cells induced with SCF alone (at 100 ng/mL). Because no TB+ cells were seen with SCF alone at 20 and 50 ng/mL, the patterned bar is prepresented by (0). The solid bar shows the percentage of TB+ cells induced with eotaxin (50 ng/mL) combined with increasing concentrations of SCF. (C and D) Mature mast cells generated after 10 days in culture with eotaxin and SCF are shown stained with TB or MG.
Eotaxin influences mast cell differentiation in FBL. Graph A shows the percentage of more differentiated mast cells identified on cytospins stained with modified giemsa (MG), based on granular content. The percentage of heavily-granulated cells (toluidine blue (TB)+, MG+, or CAE+) was determined by counting the number of these cells (in 25 or 100 fields at 400× magnification) on cytospun preparations made by harvesting all colony progeny induced in methylcellulose. Total cell counts ranged from 84 to 964. (B) Increased numbers of TB+ mast cells are induced as the concentration of SCF increases. The patterned bar represents the percentage of TB+ cells induced with SCF alone (at 100 ng/mL). Because no TB+ cells were seen with SCF alone at 20 and 50 ng/mL, the patterned bar is prepresented by (0). The solid bar shows the percentage of TB+ cells induced with eotaxin (50 ng/mL) combined with increasing concentrations of SCF. (C and D) Mature mast cells generated after 10 days in culture with eotaxin and SCF are shown stained with TB or MG.
In three experiments, the percentage of mature mast cells obtained from 11 dpc FBL colonies grown in SCF and eotaxin were 10.6%, 5.2%, and 8.3% (mean = 8% ± 3%), while similar cells obtained from YS cultures represented 2.8% ± 1.7% of the total cells (Fig 5A) produced in methylcellulose after 10 days. The percentages were obtained by quantitating the number of mast cells on cytospun preparations of the entire contents of methylcellulose dishes. Immunohistochemical staining for c-kit was performed on colonies derived from 11 dpc YS and FBL and mast cells were found to be positive; no c-kit positive cells were identified in colonies grown in SCF or eotaxin alone.
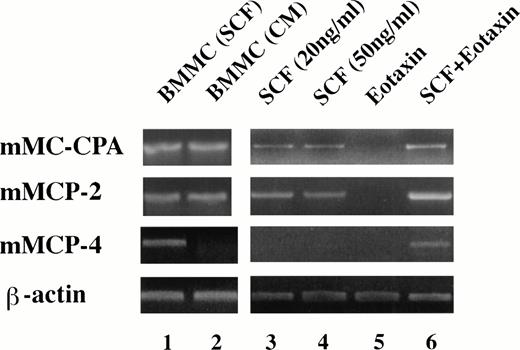
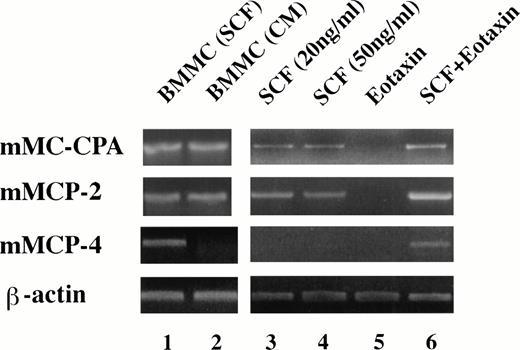
To further confirm that the population of heavily-granulated cells obtained in methylcellulose culture with SCF and eotaxin were mast cells, we performed RT-PCR analysis for the expression of serine proteases specific for mast cells.37 The expression of at least eight different serine proteases and an exopeptidase, carboxypeptidase A, have been used to define different mast cell populations.38 As shown in Fig6, mast cells obtained from cultures of mouse bone marrow cells (BMCMC) in mouse spleen cell-conditioned medium are immature mast cells expressing mMCP-2 and mMC-CPA, while mast cells obtained in medium with SCF as the only exogenous growth factor expressed mMCP-2 and mMC-CPA, but in addition express mRNA for mMCP-4. We found that colonies grown from 11 dpc FBL in SCF alone expressed mMCP-2 and mMC-CPA, but not mMCP-4 mRNA (Fig 6). However, if the progenitors were grown in SCF in combination with eotaxin, then mMCP-2, 4, and mMC-CPA mRNA were expressed.
Eotaxin is required for the differentiation of mast cell progenitors expressing mMCP-4. RT-PCR was used to determine the pattern of mast cell proteases (carboxypeptidase A, mMC-CPA; cellular protease-2, mMCP-2; and cellular protease 4, mMCP-4) expressed in FBL colonies after exposure to the factors indicated in the figure (see Materials and Methods for details). Lanes 1 and 2 represent BMCMC derived from BALB/c mice, in SCF alone (lane 1) or Con-A–stimulated conditioned medium as a source of IL-3 (CM; lane 2). Lanes 3 to 6 represent FBL-derived colonies (11 dpc).
Eotaxin is required for the differentiation of mast cell progenitors expressing mMCP-4. RT-PCR was used to determine the pattern of mast cell proteases (carboxypeptidase A, mMC-CPA; cellular protease-2, mMCP-2; and cellular protease 4, mMCP-4) expressed in FBL colonies after exposure to the factors indicated in the figure (see Materials and Methods for details). Lanes 1 and 2 represent BMCMC derived from BALB/c mice, in SCF alone (lane 1) or Con-A–stimulated conditioned medium as a source of IL-3 (CM; lane 2). Lanes 3 to 6 represent FBL-derived colonies (11 dpc).
Mast cells are more prevalent in colonies derived from FBL than from FL.
We also examined the relative number of mast cells in FBL and FL cultured in SCF and eotaxin. We found that the number of mature mast cells in 10-day old methylcellulose cultures was greater in FBL (8% ± 3%) compared with fetal liver (<1%) at 11 dpc (Fig 5A). A previous study showed that the FL contains approximately 10 times more multipotent precursors for granulocytes, macrophages, and megakaryocytes than FBL at 11 dpc.39 In our assay system, two to four times more total colonies were produced from FL in the presence of SCF and eotaxin than from FBL and YS (Fig 3f), despite using fivefold fewer FL cells to seed the cultures. The mean number of total blood and FL cells per organism at 11 dpc was 8.5 ± 5.4 × 105 (n = 8) and 3.7 ± 2.6 × 105 (n = 5), respectively, which compares favorably with reported estimates.39 The estimated frequency of FBL cells at 11 dpc responsive to SCF in combination with eotaxin ranged from 0.02% (1/3,703 FBL) to 0.18% (1/555) with a mean of 0.05% (1/2,096 ± 1,312, n = 6) or ≈500 FBL-derived colonies per embryo. In three independent experiments with 11 dpc FL, the variability was much greater, with estimates of 0.06% (1/1,666 FL cells), 0.75% (1/133), and 3.7% (1/27) or ≈1,600 FL-derived colonies/embryo on average in response to both growth factors. The wide variability likely reflects the rapid, exponential expansion of progenitors that occurs in the FL at 11 to 12 dpc and variability in embryo age, given that sibling embryos can differ by as much as 12 hours of development.
DISCUSSION
Few studies have examined the role of chemoattractants in embryonic hematopoietic development. Our study showed the chemokine eotaxin and mRNA for its receptor, CCR-3, are expressed during embryogenesis. We also found that eotaxin in combination with SCF synergistically induces the differentiation from embryonic progenitors of granulated cells with morphological features characteristic of mature mast cells, in vitro. Mast cells were definitively identified by the expression pattern of mast cell-specific proteases. There are several possibilities for this observation. Eotaxin, acting through CCR-3 or another receptor, could induce the secretion of additional SCF or other cytokines in culture that accelerate mast cell proliferation, differentiation, and/or survival. Alternatively, eotaxin could compensate for low levels of c-kit occupancy by acting through a signaling pathway that is shared by c-kit or IL-3. Ogasawara et al,40 recently reported that mouse BM-derived mast cells initially grown in IL-3 undergo exocytosis and cytokine expression in response to G-protein-activating polybasic compounds after coculture with Swiss 3T3 fibroblasts and soluble c-kitligand. However, these responses were suppressed by pertussistoxin; furthermore, they were not seen when BM-derived mast cells were cultured with soluble SCF or 3T3 fibroblasts alone. Phenotypically, the BM-mast cells cocultured with SCF and fibroblasts resembled connective tissue-type mast cells, as they expressed mMCP-4.41 This study suggested that a fibroblast-derived mast cell maturation factor acted synergistically with SCF to mediate the functional differentiation of BM-derived mast cells to connective tissue-type mast cells that respond to polybasic compounds. While the number of mature mast cells that developed in our methylcellulose culture was insufficient to examine functional responses, our findings suggest that eotaxin similarly aids SCF in promoting or maintaining mast cell differentiation towards a mMCP-4–expressing mast cell.
Rodewald et al42 recently described a committed fetal mast cell precursor identified by Thy-1loc-kithi expression and mRNA for mast cell-associated proteases. Purified Thy1loc-kithi cells were also able to generate functional peritoneal mast cells when injected intoKitW/KitW-v mast cell-deficient mice. They showed that both IL-3 and SCF (10 ng/mL) were needed to support their growth, but no chemoattractants were tested. They also found that mast cell precursors were present exclusively in the FBL at mid to late stages of gestation. It is not known if these were committed cells released from the FL. Eotaxin does not induce migration of mature mast cells, and several studies have failed to identify eotaxin expression in IL-3–dependent mast cells.17 18
Mast cell precursor activity has also been detected in the YS at 10 dpc.43 Keller et al43 described the kinetics of mast cell development from embryoid bodies, YS, FL, and adult bone marrow. They showed that mast cell precursors were the last cell type to develop from embryoid bodies, appearing only after 10 days in culture. They responded preferentially to SCF alone (250 ng/mL), and IL-3/IL-1–responsive mast cell precursors were not present until the fourteenth day of differentiation. Mast cell precursors from YS at 10 dpc and FL at 12 dpc responded better to SCF alone than to IL-3 combined with IL-1. There are conflicting reports on whether the IL-3 receptor gene is expressed by the FL earlier than 13 dpc44(and Gutierrez-Ramos, unpublished data). In contrast, the number of mast cell colonies derived from adult bone marrow was slightly higher in the presence of IL-3 plus IL-1, compared with SCF alone. Although we saw very low numbers of mature mast cells arising in our cultures from FL cells grown with both factors, our studies do not suggest that mast cell precursors are absent from FL at 11 dpc. Rather, the percentage of mast cell precursors may be much greater in FBL, or their response to cytokines or chemokines may be age- or tissue-specific.
We have examined the effect of eotaxin on embryonic progenitors between 10 and 14 dpc and found that eotaxin’s ability to induce colony formation in combination with SCF was greatest for progenitors at 11 dpc, but then declined sharply at 12 dpc, with no or few colonies induced from progenitors at 13 to 14 dpc.45 This was true for all three tissues tested, although the decline in the number of FL-derived colonies after 11 dpc is not nearly as sharp or as complete as with FBL and YS tissues. The reduced effect after 11 dpc may reflect a change in the number of responsive progenitors, or a relative reduction secondary to a dilutional effect from the expansion of other cell types, or simply the result of migration out of a developmental compartment. However, when the total number of colonies per embryo equivalent is plotted, the decline in colony number still occurs sharply between 12 to 13 dpc.
Overall, the highest frequency of responding colony-forming cells (myeloid and erythroid) is in FL. Between 10 and 12 dpc, the number of multipotent hematopoietic cells increases linearly in the circulation and exponentially in the FL.39 In addition, there may be wide variability in cell estimates and progenitor potential during embryogenesis, due to a difference of several hours in the time of development between individual embryos in the same mother. Comparing our activity with the temporal scheme of fetal lineage commitment proposed by others,9-11 we can only speculate on which subset of progenitor(s) eotaxin may be inducing with SCF.
In adult mice, evidence suggests that eotaxin in combination with SCF functions as a granulocyte-macrophage colony-stimulating factor.46 The increase in Mac-1+ cells in our cultures suggests that eotaxin also promotes myelopoiesis from embryonic progenitors. Peled et al46 also found a large increase in bone marrow-derived Gr-1+ cells with both factors, which we did not find. However, day 12 YS and splanchnopleural cultures could not support the growth of GR-1+ cells in culture.10 The increase in Mac-1 may be due to its upregulated expression on stem cells or early progenitors, given that it is coexpressed with c-kit and CD34 on stem/progenitor cells in FL and the AGM,31 but not on mature mast cells. Interestingly, the adhesion molecule very late antigen-4 (VLA-4)47 was downregulated on colony progeny induced with SCF and eotaxin, and pertussis toxin upregulated VLA-4 expression when it was combined with SCF and eotaxin (Table 1). In human fetuses at 23 to 34 weeks of gestation, blood obtained by percutaneous umbilical cord sampling had a surprisingly large proportion of eosinophils (42% ± 26%).48 Eotaxin may, thus, play a role in the expansion of this granulocytic subset during fetal development in the absence of inflammation or infection.
We found RBC, mast cells, and Mac-1+ cells (presumably macrophages, based on morphology) induced in our methylcellulose cultures. SCF alone can induce extensive proliferation of human erythroid progenitors,49 but chemokine-induced proliferation has not been studied. In our system, eotaxin alone has minimal effect on induction of CFU-E (Fig 4). Thus, the progenitor we expand may be capable of multipotency or we are simultaneously expanding different committed progenitors.
As for committed progenitors, they are mainly erythroid and myeloid precursors that peak in the YS at 9 to 10 dpc and then migrate to the FL as part of the first wave of migrating cells. We have not analyzed YS from 9 dpc, but our peak of colony-forming activity (11 to 12 dpc) correlated better temporally with a second wave of progenitors into the FL, which is comprised of multipotent progenitors and long-term repopulating cells from the AGM and YS.10,11 The fact that adult BM progenitors (lacking mature lineage markers) can also be synergistically induced with SCF and eotaxin implies that a definitive progenitor is responding.46
Mice genetically deficient in eotaxin have been described, but embryonic and mast cell populations in these mice were not examined.50 These mice have reduced numbers of circulating eosinophils and the number of eosinophils “recruited” into inflamed airways was also diminished. There was no reduction in the number of eosinophil progenitors in the bone marrow, however. As with the MIP-1α–deficient mice, which have no overt hematopoietic abnormalities despite MIP-1α’s ability to inhibit stem cell proliferation in vitro,15 51 there is redundancy in the immune system that compensates for the loss of one cytokine/chemokine during embryogenesis. Similarly, chemokine gradients likely do not exist singularly in vivo, and recent studies have focused on deciphering how one chemokine gradient can be “dominant” in a tissue where multiple chemokines and their receptors are simultaneously expressed.52 We are currently analyzing the number of mast cells present in eotaxin-deficient mice and the response of mast cell populations in pathological conditions associated with high levels of eotaxin. We speculate that eotaxin production during allergic or inflammatory responses, such as antigen-induced lung inflammation, may influence the differentiation and/or function of mast cells at local sites of inflammation.
In summary, our study shows that eotaxin can modulate granulopoiesis, specifically mast cell differentiation, from fetal progenitors and suggests that the study of chemokines during development is a novel and unique approach to understanding many pathological processes in the adult, as well.
ACKNOWLEDGMENT
We wish to acknowledge Mark Ryan for his technical assistance in the FACSsort analysis and Dr Clare Lloyd for her excellent assistance with the immunohistochemical techniques. Dr L. Kremer (Centro Nacional de Biotecnologia, Universidad Autonoma de Madrid) very kindly provided the MoAb to eotaxin, and Dr Michelle Letarte (The Hospital for Sick Children, University of Toronto) provided the antiendoglin MoAb. Embryonic day 7.5 RNA was obtained from Dr Tak Mak at the University of Toronto. We also wish to thank Dr Daniel Friend for his very helpful comments and recommendations.
Supported in part by Grants No. DK33506 and Core B (Morphology), DK46819, DK40561 (Clinical Nutrition Research Center at Harvard) from the National Institutes of Health, Bethesda, MD.
Address reprint requests to Jose-Carlos Gutierrez-Ramos, PhD, Millennium Pharmaceuticals, Inc, 640 Memorial Dr, Cambridge, MA 02139.
The publication costs of this article were defrayed in part by page charge payment. This article must therefore be hereby marked "advertisement" is accordance with 18 U.S.C. section 1734 solely to indicate this fact.