Abstract
The mechanisms underlying the growth-inhibitory effect of retinoids on normal human B lymphocytes are not well understood. We addressed this issue by examining the effect of retinoic acid on the cell cycle machinery involved in G1/S transition. When retinoic acid was administered to B cells stimulated into mid to late G1 by anti-IgM antibodies (anti-μ) and Staphylococcus aureus crude cell suspension (SAC), the phosphorylation of pRB required for S-phase entry was prevented in a time- and dose-dependent manner. Thus, 2-hour treatment with retinoic acid at the optimal concentration of 1 μmol/L prevented phosphorylation of pRB, and effects were noted at concentrations as low as 10 nmol/L. Based on our results, we suggest that the rapid effect of retinoic acid on pRB phosphorylation is due primarily to the reduced expression of cyclin E and cyclin A in late G1. This could lead to the diminished cyclin E– and cyclin A–associated kinase activities noted as early as 2 hours after addition of retinoic acid. Furthermore, our results imply that the transient induction of p21Cip1 could also be involved. Thus, retinoic acid induced a rapid, but transient increased binding of p21Cip1 to CDK2. The retinoic acid receptor (RAR) agonist TTNPB mimicked the key events affected by retinoic acid, such as pRB phosphorylation, cyclin E expression, and expression of p21Cip1, whereas the RAR-selective antagonist Ro 41-5253 counteracted the effects of retinoic acid. This implies that retinoic acid mediates its growth-inhibitory effect on B lymphocytes via the nuclear receptors.
AN IMPORTANT ASPECT of the in vivo function of vitamin A is its protection against infections.1,2 It is believed that this in part is due to the requirement of retinoids in maintaining normal differentiation of the epithelium along the body linings.3 The role of retinoids on the immune system per se, however, is unclear. Although most in vivo studies conclude that retinoids are required for a functional immune system,1,4 high intake of vitamin A has been reported to impair normal immune responses.5 In vitro studies on cultured lymphocytes have shown both stimulatory6,7 and inhibitory8,9 effects of retinoids. We have previously documented that physiological levels of retinoids inhibit the proliferation of human peripheral blood B lymphocytes8 as well as of human B-cell precursors, murine splenic lymphocytes, and murine B-cell precursors.10 Still, however, it appears that the inhibitory effect of retinoids on normal lymphocytes is a controversial issue.
To further strengthen our data on the inhibitory role of physiological levels of retinoids on B-cell growth, we wished to examine the effect of retinoids on the cell cycle machinery. Unraveling the components of the cell cycle machinery has during recent years lead to a profound new understanding of the mechanisms involved in cell cycle progression.11,12 Furthermore, the identification of cyclin-dependent kinases (CDKs) as the key driving force in cell cycle progression has helped to identify important substrates that are phosphorylated by CDKs. Among these key substrates, the protein product of the retinoblastoma gene (pRB) appears as particularly important.12pRB is a tumor suppressor gene that is frequently inactivated in human tumors.13 The physiological role of pRB in normal cells is, however, to prevent the cells from entering into S phase from G1.12 This feature of pRB is accomplished by its role as a pocket protein, binding transcription factors such as E2F and DP that are known to regulate S-phase–promoting genes.14 The phosphoprotein pRB is active as a sequestrator of transcription factors only in its hypophosphorylated state. Upon sequential phosphorylation in G1 by CDK4/CDK6 and CDK2, pRB is inactivated, leading to release of E2F, transcription of S-phase genes, and subsequent entry into S phase.12 It is believed that pRB phosphorylation identifies the restriction point in G1 of the cell cycle, as a point of no return in stimulation of cells to proliferate.12 Thus, the regulation of CDKs that are responsible for pRB phosphorylation appears as a key event in the processes that determine the proliferative state of a cell. The main regulation of CDK activity is at the level of interaction with specific cyclins, CDK inhibitors (CKIs), and by specific phosphorylation and dephosphorylation of critical amino acids on the CDK catalytic subunit.11
In the present study, we have examined the effect of physiological doses of all-trans-retinoic acid on regulation of the cell cycle machinery in human peripheral blood B lymphocytes, with specific emphasis on the events leading to pRB dephosphorylation in the G1 phase of the cell cycle. It has been an issue of debate whether or not nuclear receptors mediate the effects of retinoids on growth, differentiation, and apoptosis in lymphocytes,15 as well as in other cell types.16 In this study we therefore included a synthetic retinoid known to act via nuclear retinoic acid receptors (RAR), which we recently showed could mimic the effects of all-trans-retinoic acid on regulation of both growth and apoptosis in human B-lymphocytes.17 Furthermore, we included an RAR-selective antagonist, previously shown to counteract the effects of retinoic acid in several different cell systems.18
MATERIALS AND METHODS
Reagents and antibodies.
Retinoids are defined as retinol and derivatives of retinol, including those without biological activity. Vitamin A is commonly used to define naturally occurring retinoids with biological activity similar to that of retinol, including retinol itself. Retinoic acid is the oxidized form of retinol, and the isomer all-trans-retinoic acid was purchased from Sigma Chemical Co (St Louis, MO). The RAR agonist Ro13-7410 (TTNPB) and the RAR-selective antagonist Ro 41-5253 were kindly provided by Dr M. Klaus (Hoffman-La Roche, Basel, Switzerland).
Anti-cyclin E (HE12; for Western blot analysis, HE111; for immunoprecipitation), anti-cyclin A (C-19), anti-CDK2 [(M2)-G], anti-cyclin D2 (34B1-3), anti-cyclin D3 (D-7), anti-CDK4 (C-2), anti-CDK6 (C-21), anti-p21Cip1 (C-19), and anti-p27Kip1 (C-19) were purchased from Santa Cruz Biotechnology (Santa Cruz, CA). Anti-pRB (G3-254) was obtained from Pharmingen (San Diego, CA).
Cell purification, cell culture, and proliferation assay.
Resting human B lymphocytes were isolated from platelet-depleted buffy coats obtained from the Blood Bank (Ullevål Hospital, Oslo, Norway), as described by Funderud et al.19 Each buffy coat (50 mL) was diluted in 25 mL RPMI 1640 medium (GIBCO, Grand Island, NY) containing 0.01 mol/L EDTA and mixed with 2 × 108 Dynabeads coated with anti-CD19 antibody (Product No. 111.04; Dynal, Oslo, Norway). After incubation of the mixture on a rotating wheel for 1 hour at 4°C, the rosetted cells were attracted to a samarium cobalt magnet and the nonrosetting cells were removed. Cell rosettes were subsequently washed five times with 10 mL RPMI 1640 medium containing 1% heat-inactivated fetal bovine serum (FBS; GIBCO) to remove the residual nonrosetting cells. To detach the beads from the cells, cell rosettes were cultured in RPMI 1640 medium containing 1% FBS overnight at 37°C in a CO2 incubator, and then the beads were attracted to a samarium cobalt magnet and the B cells were recovered from the supernatant. Under these conditions, less than 1% T cells, natural killer (NK) cells, and monocytes were detected in the cell population.
B cells were cultured in RPMI 1640 medium supplemented with 1% FBS, 2 mmol/L glutamine, 125 U/mL penicillin, 125 μg/mL streptomycin at a density of 1.5 × 106 cells/mL as previously described.19 Cells were stimulated to enter the cell cycle by the addition of the combination of anti-μ [37.5 μg/mL of F(ab′)2 fragments of rabbit polyclonal antibodies to human IgM heavy chain; Dako, Copenhagen, Denmark] and 0.005% of formalin-fixed SAC (Sigma). To examine the effect of retinoic acid or the RAR-selective agonist TTNBP on B lymphocytes, different concentrations of retinoic acid or TTNBP were added to cell cultures for the indicated number of hours before harvesting the cells 32 hours poststimulation. For measurement of DNA synthesis, cells were cultured in microtiter plates at an initial density of 7.5 × 104 cells/0.2 mL. Cells were pulsed with 0.2 mCi of [3H]thymidine (Amersham, Buckinghamshire, UK) for the last 20 hours of a 68-hour incubation. The cells were then harvested on a cell harvester and counted on a liquid scintillation counter (Topcount; Packard, Meriden, CT).
Immunoblot analysis and immunoprecipitations.
Total cell lysates were prepared by lysis with Laemmli sample buffer.20 After boiling the samples for 5 minutes, cellular debris was removed by centrifugation at 4°C and the protein content of the supernatant was determined by Bradford analysis (Bio-Rad, Hercules, CA). Equal amounts of proteins (30 to 100 μg) were resolved on a 7.5% (for pRB) or 10% to 12% (for other proteins) polyacrylamide gel under reducing conditions, and transferred onto nitrocellulose membrane (Amersham) using a semidry transfer cell (Bio-Rad). After blocking in TBS-T (0.1% Tween 20) containing 5% nonfat dry milk for 1 hour at room temperature, the membrane was incubated with 1 μg/mL of the indicated antibody in TBS-T for 2 hours at room temperature. The membrane was then washed four times with TBS-T, incubated with a 1:6000 dilution of horseradish peroxidase-linked secondary antibody (Bio-Rad), and the immunoreactive proteins were visualized with the enhanced chemiluminescence detection system (Amersham).
For immunoprecipitations followed by immunoblotting, whole cell extracts were prepared essentially as described by Gorospe et al.21 In brief, the cells were lysed for 20 minutes on ice in Triton X-100 lysis buffer (50 mmol/L Tris [pH 7.5], 250 mmol/L NaCl, 0.1% Tween X-100, 10 μg of leupeptin per mL, 9.5 μg of aprotinin per mL, 35 μg of phenylmethylsulfonyl floride per mL, 5 mmol/L NaF, 0.1 mmol/L ortho-vanadate, 10 mmol/L β-glycerophosphate), and the lysate was cleared by centifugation at 4°C. Cell lysates corresponding to equal amounts of protein (500 μg) were immunoprecipitated for 2 hours at 4°C with the desired antibody. The immunocomplexes were absorbed to 30 μL of a 1:1 slurry of protein G sepharose beads (Pharmacia, Sweden) for 1 hour at 4°C, collected by centrifugation at 2000g for 5 minutes, and washed twice with the lysis buffer. The beads were then resuspended in Laemmli sample buffer, boiled, and subjected to Western analysis.
Kinase assays.
For histone H1 kinase assays, whole cell extracts were prepared with Triton X-100 lysis buffer as described.21 After preclearing with 20 μL of a 1:1 slurry of protein G-sepharose for 30 minutes at 4°C, 200 μg of each lysate was immunoprecipitated with 2 μg of the indicated antibody as described above. After immunoprecipitation, the beads were washed twice with lysis buffer, and once with kinase buffer (50 mmol/L Tris [pH 7.5], 10 mmol/L MgCl2, 1 mmol/L dithiothreitol [DTT], 2 mmol/L EGTA, 1 mmol/L NaF, 0.1 mmol/L ortho-vandate, 10 mmol/L β-glycerophosphate). The beads were resuspended in 15 μL of kinase buffer containing 30 μmol/L adenosine triphosphate (ATP), 5 μg histone H1 (Upstate Biotechnology, NY), 10 μCi of [γ-32P]ATP per reaction mixture, and incubated for 30 minutes at 30°C. Reactions were stopped with addition of 7.5 μL 3× Laemmli sample buffer. The samples were boiled for 5 minutes and subjected to sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE). After electrophoresis, gels were stained with Coomassie blue, dried, and subjected to autoradiography.
pRB kinase assays were performed essentially as described previously by Matsushime et al.22 In brief, whole cell extracts were prepared by resuspending the cell pellets on ice in Tween 20 lysis buffer (50 mmol/L HEPES [pH 7.5], 150 mmol/L NaCl, 1 mmol/L EDTA, 2.5 mmol/L EGTA, 0.1 % Tween 20, 10% glycerol, 1 mmol/L DTT, 10 μg of leupeptin per mL, 9.5 μg of aprotinin per mL, 35 μg of phenylmethylsulfonyl fluoride per mL, 5 mmol/L NaF, 0.1 mmol/L ortho-vandate, 10 mmol/L β-glycerophosphate), and vortexed at 5-minute intervals for 20 minutes. After removing the insoluble material by centrifugation, the lysates were precleared by incubation with 25 μL of a 1:1 slurry protein G-sepharose for 30 minutes at 4°C and analyzed for protein content by the Bradford method (Bio-Rad). CDK6-associated complexes were recovered by immunoprecipitation of 600 μg of cell lysates with anti-CDK6 polyclonal antibodies. After washing the beads twice with Tween 20 lysis buffer and three times with pRB kinase buffer (50 mmol/L HEPES [pH 7.5], 10 mmol/L MgCl2, 1 mmol/L DTT, 2 mmol/L EGTA, 1 mmol/L NaF, 0.1 mmol/L ortho-vandate, 10 mmol/L β-glycerophosphate), the reactions were initiated by addition of 50 μL pRB kinase buffer containing 20 μmol/L ATP, 1 μg glutathione S-transferase (GST)–tagged RB(GST-RB) (Santa Cruz Biotechnology), and 10 μCi of [γ-32P]ATP per reaction mixture and incubated for 30 minutes at 30°C. After addition of the 3× Laemmli sample buffer, samples were boiled and subjected to SDS-PAGE analysis and autoradiography.
Northern blot analysis.
Poly (A)+ mRNA was purified from 25 × 106cells for each treatment using Dynabeads oligo (dT)25magnetic beads (Dynal, Oslo, Norway) following manufacturer’s instructions, fractionated on a formaldehyde/agarose gel, transferred onto Hybond-N filter (Amersham, UK) in 20× SSC and ultraviolet crosslinked.23 The filter was then hybridized with the indicated cDNA probes at 42°C in 50% formamide, washed to final stringency of 0.1× SSC, 0.1% SDS at 50°C, and autoradiographed. The probes (p21Cip1, NotI fragment of PC-WAF1-S 24; cyclin E, HindIII fragment of Rc-cycE25; cyclin A, HindIII-XbaI fragment of Rc-cycA25) were labeled with [α-32P]dCTP (Amersham Megaprime Labeling System, Amersham) according to the Amersham Megaprime protocol. The p21Cip1 probe was a kind gift from B. Vogelstein (Baltimore, MD). The cyclin E and cyclin A probes were kind gifts from R.A. Weinberg (Cambridge, MA).
RESULTS
Retinoic acid inhibits DNA synthesis also when added near the restriction point in normal B-lymphocytes.
The purpose of the present study was to examine early changes in cell cycle parameters upon treatment of normal human B lymphocytes with retinoic acid. We have previously shown that the restriction point in human B lymphocytes is located approximately 30 to 35 hours into G1, ie, approximately 10 hours before S-phase entry.26 We therefore measured the effect of retinoic acid on DNA synthesis when added at the beginning of the cell culture, or when added after 32 hours of stimulation with anti-μ and SAC. As shown in Fig 1, retinoic acid inhibited the DNA synthesis of B lymphocytes at both the physiological level (10 nmol/L) and the pharmacological level (1 μmol/L), in agreement with our previous report.8 Thus at the optimal dose of 1 μmol/L retinoic acid, DNA synthesis was reduced by 61%. Interestingly, retinoic acid added as late as 32 hours after stimulation of the cells still inhibited the DNA synthesis, although the effect was somewhat reduced.
Effect of retinoic acid on DNA synthesis. Human B cells were isolated and stimulated with μ (37.5 μg/mL) and SAC (0.005%) as described in Material and Methods. Cells (0.75 × 106 cells/mL) were cultured in 96-well culture plates in triplicates. Retinoic acid (10 or 1000 nmol/L) was added at the indicated times (T) after stimulation. [3H]Thymidine incorporation (mean ± SD) was measured as described in Material and Methods. One representative experiment of three is shown. μ, F(ab′)2 fragment of rabbit antihuman IgM; SAC,Staphylococcus aureus crude cell suspension; RA, retinoic acid.
Effect of retinoic acid on DNA synthesis. Human B cells were isolated and stimulated with μ (37.5 μg/mL) and SAC (0.005%) as described in Material and Methods. Cells (0.75 × 106 cells/mL) were cultured in 96-well culture plates in triplicates. Retinoic acid (10 or 1000 nmol/L) was added at the indicated times (T) after stimulation. [3H]Thymidine incorporation (mean ± SD) was measured as described in Material and Methods. One representative experiment of three is shown. μ, F(ab′)2 fragment of rabbit antihuman IgM; SAC,Staphylococcus aureus crude cell suspension; RA, retinoic acid.
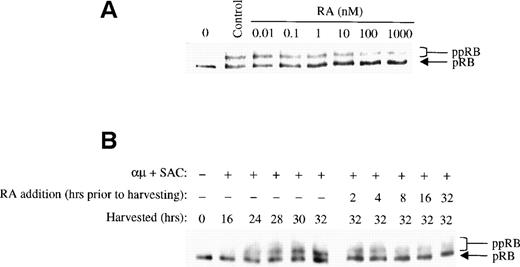
Retinoic acid prevents phosphorylation of pRB in a dose- and time-dependent manner in late G1.
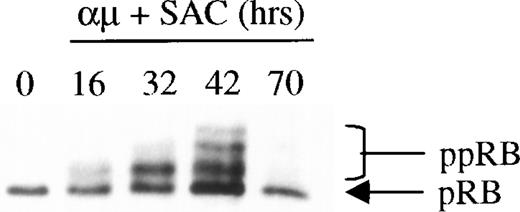
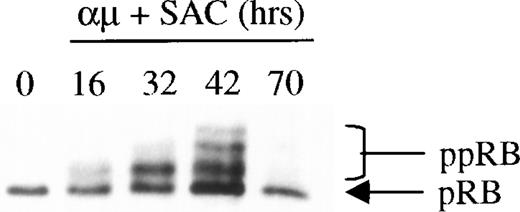
The phosphorylation of pRB by CDK2 in complex with cyclin E is believed to be a key event in the regulation of S-phase entry, and appears to define the restriction point in late G1.12 It was therefore of particular interest to examine whether retinoic acid would prevent phosphorylation of pRB as part of its growth-inhibitory effect on human B lymphocytes. The Western blot in Fig 2shows the kinetics of pRB phosphorylation during stimulation of B-lymphocytes with anti-μ and SAC. The results showed that the maximum level of pRB phosphorylation was obtained after 42 hours of stimulation (ie, approximately at the time of entrance into S phase26). When the cells entered the G1 phase of the following cell cycle after stimulation for 70 hours, pRB again appeared in its hypophosphorylated state.
Expression and phosphorylation of pRB in stimulated human B cells. Freshly isolated human B cells (1.5 × 106cells/mL) were stimulated with a combination of μ (37 μg/mL) and SAC (0.005%) for the indicated times. Total cell lysates were prepared and 30 μg per lane was subjected to Western blot analysis as described in Material and Methods. Hypophosphorylated pRB and hyperphosphorylated pRB (ppRB) were detected with the RB-PMG3-245 monoclonal antibody. The lane indicated as 0 hrs refers to a sample harvested before mitogenic stimulation of the cells. μ, F(ab′)2 fragment of rabbit antihuman IgM; SAC,Staphylococcus aureus crude cell suspension.
Expression and phosphorylation of pRB in stimulated human B cells. Freshly isolated human B cells (1.5 × 106cells/mL) were stimulated with a combination of μ (37 μg/mL) and SAC (0.005%) for the indicated times. Total cell lysates were prepared and 30 μg per lane was subjected to Western blot analysis as described in Material and Methods. Hypophosphorylated pRB and hyperphosphorylated pRB (ppRB) were detected with the RB-PMG3-245 monoclonal antibody. The lane indicated as 0 hrs refers to a sample harvested before mitogenic stimulation of the cells. μ, F(ab′)2 fragment of rabbit antihuman IgM; SAC,Staphylococcus aureus crude cell suspension.
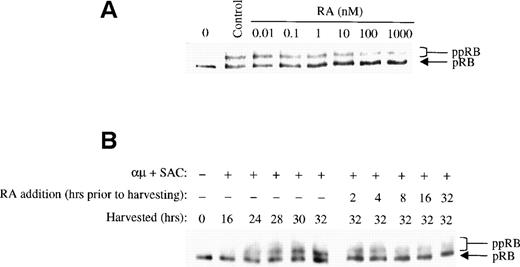
We examined the effect of retinoic acid on pRB phosphorylation by adding different concentrations of retinoic acid to the cell cultures together with the stimulatory agent at time 0. The cells were harvested after 32 hours, and the level of pRB phosphorylation was assessed by Western blot analysis. As shown in Fig 3A, phosphorylation of pRB was prevented by retinoic acid at low concentrations. In fact, concentrations as low as 1 to 10 nmol/L, ie, physiological concentrations,27 showed considerable inhibition of pRB phosphorylation. However, an even stronger effect on pRB phosphorylation was noted at 1 μmol/L of retinoic acid, and this concentration was therefore used in the following experiments.
Retinoic acid inhibits pRB phosphorylation. (A) Human B cells (1.5 × 106 cells/mL) were stimulated with μ (37 μg/mL) and SAC (0.005%) for 32 hours in the absence (control) or presence of retinoic acid at the indicated concentrations. Total cell lysates were prepared and 30 μg of total protein was analyzed for pRB protein by Western blotting. The lane indicated as 0 hrs refers to a sample harvested before mitogenic stimulation of the cells. (B) Left panel: Cells (1.5 × 106 /mL) were stimulated with μ (37 μg/mL) and SAC (0.005%) and harvested at the indicated times after stimulation. Right panel: Cells were stimulated with μ (37 μg/mL) and SAC (0.005%). Retinoic acid (1000 nmol/L) was then added to the cell cultures at each indicated time before harvesting the cells at 32 hours. Total cell lysates were prepared and subjected to Western blot analysis for pRB. μ, F(ab′)2 fragment of rabbit antihuman IgM; SAC, Staphylococcus aureus crude cell suspension; RA, retinoic acid.
Retinoic acid inhibits pRB phosphorylation. (A) Human B cells (1.5 × 106 cells/mL) were stimulated with μ (37 μg/mL) and SAC (0.005%) for 32 hours in the absence (control) or presence of retinoic acid at the indicated concentrations. Total cell lysates were prepared and 30 μg of total protein was analyzed for pRB protein by Western blotting. The lane indicated as 0 hrs refers to a sample harvested before mitogenic stimulation of the cells. (B) Left panel: Cells (1.5 × 106 /mL) were stimulated with μ (37 μg/mL) and SAC (0.005%) and harvested at the indicated times after stimulation. Right panel: Cells were stimulated with μ (37 μg/mL) and SAC (0.005%). Retinoic acid (1000 nmol/L) was then added to the cell cultures at each indicated time before harvesting the cells at 32 hours. Total cell lysates were prepared and subjected to Western blot analysis for pRB. μ, F(ab′)2 fragment of rabbit antihuman IgM; SAC, Staphylococcus aureus crude cell suspension; RA, retinoic acid.
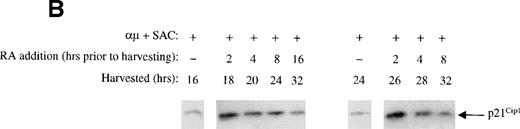
Because addition of retinoic acid at time 0 appeared to partially but not totally prevent pRB phosphorylation, we assessed the effect of retinoic acid when administered to the cells later in G1. To do so, retinoic acid was added to the cultures at different times during G1, but before the restriction point. All the cells were harvested at 32 hours, and the phosphorylation state of pRB was analyzed by Western blotting. In the right panel of Fig 3B the effect of retinoic acid on pRB phosphorylation is shown. Note that the phosphorylation patterns of pRB shown in the right panel should be compared with that of the 32-hour control in the left panel, because all the cell cultures were harvested at 32 hours. Any reduced level of pRB phosphorylation in retinoic acid-treated cells as compared with that of the 32-hour control would indicate that retinoic acid has prevented pRB phosphorylation. The other controls in the left panel of Fig 3B show the phosphorylation state of pRB at the various times in G1 when retinoic acid was added.
The results in Fig 3B indicated that pRB phosphorylation was prevented by retinoic acid in a time-dependent manner. When retinoic acid was added to the cultures between 8 and 2 hours before harvesting the cells at 32 hours into G1, retinoic acid appeared to totally prevent further pRB phosphorylation. When added earlier in G1, ie, between time 0 and 16 hours into G1, the phosphorylation of pRB was only partially prevented. These results are in agreement with our previous findings that retinoic acid blocks B lymphocytes in mid to late G1, ie, after approximately 24 hours of stimulation into G1.8
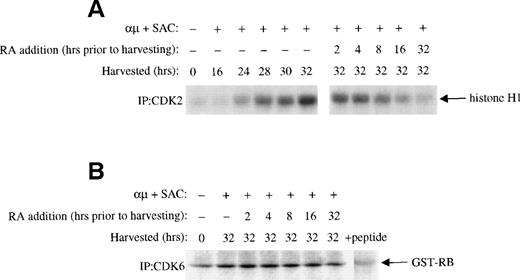
Retinoic acid inhibits the kinase activity of CDK2.
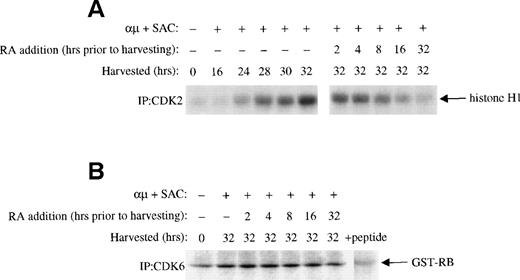
The phosphorylation of pRB in G1 is mediated by CDK4 and CDK6 in complex with cyclin D, and later in G1 by CDK2 in complex with cyclin E and cyclin A.12 The prevention of pRB phosphorylation in the presence of retinoic acid could therefore be due to inhibition of one or more of these complexes. We therefore examined the effect of retinoic acid on the activities of CDK4, CDK6, and CDK2. As shown in Fig 4A, the activation-induced increase in CDK2 activity was reduced by retinoic acid in a time-dependent manner. Again an effect was noted even when retinoic acid was added as late as 2 hours before harvesting the cells. No effect of retinoic acid was noted on the kinase activity of CDK6 (Fig 4B), whereas CDK4 kinase activity in our hands was below detectable levels in the isolated B-lymphocytes using GST-RB as substrate (data not shown).
Effect of retinoic acid on G1 CDKs. Mitogenic stimulation and retinoic acid treatment of the cells were performed as described in the legend to Fig 3B. Whole cell extracts were prepared as described in Material and Methods. (A) Equal amounts of whole cell extracts (200 μg) were immunoprecipitated with antibody to CDK2 followed by kinase assay with histone H1 as substrate as described in Material and Methods. (B) Cells were stimulated with μ (37 μg/mL) and SAC (0.005%) and harvested at 32 hours. For retinoic acid treatment of the cells, retinoic acid (1000 nmol/L) was added to the cell cultures at each indicated time before harvesting the cells at 32 hours. CDK6 was immunprecipitated from 600 μg of whole cell extracts and the immunoprecipitates were then assayed for kinase activity with GST-RB as substrate as described in Materials and Methods. + peptide, background activity was determined by blocking the CDK6 antibody with the specific antigenic peptide. μ, F(ab′)2fragment of rabbit antihuman IgM; SAC, Staphylococcus aureuscrude cell suspension; RA, retinoic acid.
Effect of retinoic acid on G1 CDKs. Mitogenic stimulation and retinoic acid treatment of the cells were performed as described in the legend to Fig 3B. Whole cell extracts were prepared as described in Material and Methods. (A) Equal amounts of whole cell extracts (200 μg) were immunoprecipitated with antibody to CDK2 followed by kinase assay with histone H1 as substrate as described in Material and Methods. (B) Cells were stimulated with μ (37 μg/mL) and SAC (0.005%) and harvested at 32 hours. For retinoic acid treatment of the cells, retinoic acid (1000 nmol/L) was added to the cell cultures at each indicated time before harvesting the cells at 32 hours. CDK6 was immunprecipitated from 600 μg of whole cell extracts and the immunoprecipitates were then assayed for kinase activity with GST-RB as substrate as described in Materials and Methods. + peptide, background activity was determined by blocking the CDK6 antibody with the specific antigenic peptide. μ, F(ab′)2fragment of rabbit antihuman IgM; SAC, Staphylococcus aureuscrude cell suspension; RA, retinoic acid.
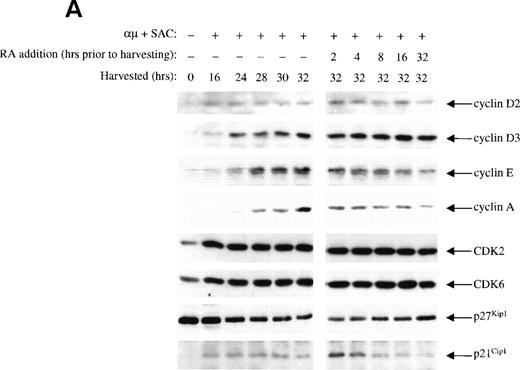
Retinoic acid changes the protein expression of cyclin E, cyclin A, and p21Cip1.
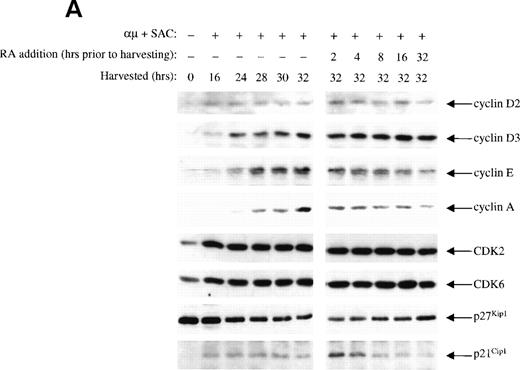
The inhibitory effect of retinoic acid on the CDK2 activity could be due to either reduced levels of cyclins or changes in the level of CKIs. By Western blot analysis we measured the kinetics of retinoic acid–mediated changes in protein levels of the various G1 cyclins, CDKs, and CKIs. As presented in the left panel of Fig 5A, we first analyzed the levels of G1 cyclins during progression through G1, showing that cyclin D2 and D3 were expressed before cyclin E, which was expressed before cyclin A. The expression of cyclin A was in particular distinct, appearing at 24 hours and exhibiting a substantial increase at 30 to 32 hours after stimulation.
Effect of retinoic acid on the expression of various cell cycle regulatory proteins in human B cells. (A) Mitogenic stimulation and retinoic acid treatment of the cells were performed as described in the legend to Fig 3B. Total cell lysates were prepared and equal amounts of total protein (60 μg) were subjected to Western blot analysis with antibodies against the proteins indicated on the right. (B) Cells (1.5 × 106 cells/mL) were stimulated for 16 or 24 hours before addition of retinoic acid (1000 nmol/L). The cells were then treated with retinoic acid for the indicated times and analyzed for the expression of p21Cip1 by Western blot analysis. μ, F(ab′)2 fragment of rabbit antihuman IgM; SAC, Staphylococcus aureus crude cell suspension; RA, retinoic acid.
Effect of retinoic acid on the expression of various cell cycle regulatory proteins in human B cells. (A) Mitogenic stimulation and retinoic acid treatment of the cells were performed as described in the legend to Fig 3B. Total cell lysates were prepared and equal amounts of total protein (60 μg) were subjected to Western blot analysis with antibodies against the proteins indicated on the right. (B) Cells (1.5 × 106 cells/mL) were stimulated for 16 or 24 hours before addition of retinoic acid (1000 nmol/L). The cells were then treated with retinoic acid for the indicated times and analyzed for the expression of p21Cip1 by Western blot analysis. μ, F(ab′)2 fragment of rabbit antihuman IgM; SAC, Staphylococcus aureus crude cell suspension; RA, retinoic acid.
The effect of retinoic acid on the level of the various cell cycle proteins are shown in the right panel of Fig 5A. Retinoic acid prevented the activation-induced increase in cyclin E and cyclin A levels in a time-dependent manner that could explain the reduced kinase activity of CDK2. The levels of cyclin D2, cyclin D3, CDK2, CDK4, and CDK6 were not affected by retinoic acid. We did not detect cyclin D1 in the B cells (data not shown).
We did not detect protein expression of the CKIs p15/p16Ink4 in the isolated human B lymphocytes (data not shown). The level of CKI p27Kip1 was high in unstimulated cells, in accordance with previous reports on other cell types.28 29 Upon stimulation with anti-μ and SAC, the expression of p27Kip1 was gradually reduced. Treatment of the cells with retinoic acid did not prevent the activation-induced decline in the levels of p27Kip1.
More surprising were the changes noted in the protein levels of p21Cip1. As reported in other cell types,28 30the level of p21Cip1 was low in unstimulated cells. Stimulation of the cells was accompanied by induction of p21Cip1, and it was further enhanced by retinoic acid when added 2 hours before harvesting the cells. However, the effect of retinoic acid on the expression of p21Cip1 was only transient. Retinoic acid present for more than 4 to 8 hours reduced the level of p21Cip1 to that observed in control cells (Fig5A).
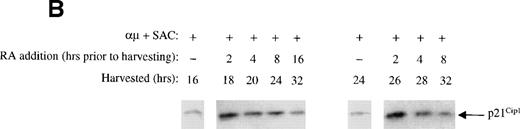
The transient expression of p21Cip1 induced by retinoic acid occurs in all parts of G1.
The transient increased expression of p21Cip1 that was observed when retinoic acid was added to the stimulated cells 2 to 4 hours before the restriction point in G1 could represent a window in G1 in which retinoic acid could induce the expression of p21Cip1. Alternatively, the noted effect could represent a general ability of retinoic acid to transiently increase the level of p21Cip1 independent of the position in the cell cycle in which retinoic acid was added to the cultures. To distinguish between these two possibilities, we added retinoic acid to the cell cultures at various times in G1, and followed the expression of p21Cip1 for the next 8 to 16 hours. Interestingly, we showed that retinoic acid was able to transiently increase the level of p21Cip1 independent of the position in G1 (Fig 5B). The immunoblot presented in Fig 5B was also reprobed with cyclin E and cyclin A, and reduced levels of both cyclins were noted when retinoic acid was added as early as 16 hours into G1 (data not shown).
Retinoic acid reduces the kinase activity associated with cyclin E and cyclin A.
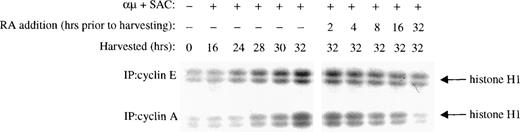
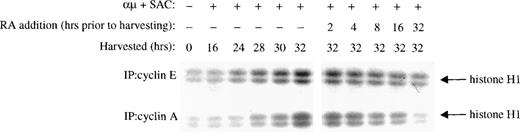
Near the restriction point in G1, pRB is phosphorylated by CDK2 in complex with cyclin E and later in complex with cyclin A.12The inhibition of CDK2 kinase activity noted in the presence of retinoic acid could therefore be due to reduced expression of cyclin E and/or cyclin A. Therefore, we measured the kinase activity associated with cyclin E and cyclin A. As shown in the right panel of Fig 6, retinoic acid prevented activation of the kinase coimmunoprecipitated with either cyclin E or cyclin A, as compared with that of control cells stimulated for 32 hours in the absence of retinoic acid.
Retinoic acid decreases the kinase activity associated with cyclin E and cyclin A. Mitogenic stimulation and retinoic acid treatment of the B cells were performed as described in the legend to Fig 3B. Whole cell extracts were prepared, immunoprecipitated with antibodies against cyclin E (top) or cyclin A (bottom), and then the associated kinase activity of the immunoprecipitates was determined with histone H1 as substrate. IP, immunoprecipitating antibody; μ, F(ab′)2 fragment of rabbit antihuman IgM; SAC,Staphylococcus aureus crude cell suspension; RA, retinoic acid.
Retinoic acid decreases the kinase activity associated with cyclin E and cyclin A. Mitogenic stimulation and retinoic acid treatment of the B cells were performed as described in the legend to Fig 3B. Whole cell extracts were prepared, immunoprecipitated with antibodies against cyclin E (top) or cyclin A (bottom), and then the associated kinase activity of the immunoprecipitates was determined with histone H1 as substrate. IP, immunoprecipitating antibody; μ, F(ab′)2 fragment of rabbit antihuman IgM; SAC,Staphylococcus aureus crude cell suspension; RA, retinoic acid.
The effect of retinoic acid on the binding of p21Cip1 to CDK2.
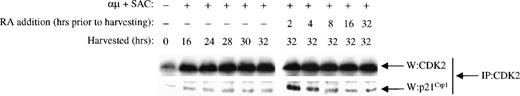
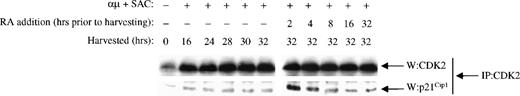
To examine whether the transient increased expression of p21Cip1 resulted in a transient increase in its binding to CDK2, we performed coimmunoprecipitation studies. CDK2 was immunoprecipitated from B cells stimulated in the presence or absence of retinoic acid. As shown in Fig 7, the expression of CDK2 was unchanged by retinoic acid in agreement with the Western blot analysis in Fig 5A. The level of p21Cip1coimmunoprecipitated with CDK2 did not, however, follow the expression of CDK2, but rather paralleled the expression of p21Cip1itself. The CDK2 present in unstimulated cells contained small amounts of p21Cip1. Entry into G1 was accompanied by a moderate increase in the amount of p21Cip1 associated with CDK2, and it remained constant throughout G1. Analysis of CDK2 immunoprecipitates from retinoic acid-treated cells showed an increase in the amount of p21Cip1 associated with the CDK2 complexes. At 2 hours after retinoic acid treatment of the cells, the amount of p21Cip1 that associated with CDK2 was at its peak level, and it then declined gradually upon longer exposure of the cells to retinoic acid. Thus, at 8 hours the level of p21Cip1associated with CDK2 had decreased to control levels. The converse experiment was also performed by immunoprecipitation with anti-p21Cip1 antibodies and then immunoblotting for CDK2. Whereas p21Cip1 immunoprecipitated from stimulated cells contained a constant amount of CDK2, there was a substantial increase in the amount of CDK2 associated with p21Cip1 at 2 hours after retinoic acid treatment (data not shown). Retinoic acid had no effect on the level of p27Kip1 associated with CDK2 (data not shown).
Retinoic acid induces a transient increase in the association of p21Cip1 with CDK2 complexes. Mitogenic stimulation and retinoic acid treatment of the B cells were performed as described in the legend to Fig 3B. Whole cell extracts were immunoprecipitated with antibody against CDK2. The immunoprecipitates were then examined for CDK2 (top) and p21Cip1 (bottom) levels by Western blot analysis. IP, immunoprecipitating antibody; W, Western blot antibody; μ, F(ab′)2 fragment of rabbit antihuman IgM; SAC, Staphylococcus aureus crude cell suspension; RA, retinoic acid.
Retinoic acid induces a transient increase in the association of p21Cip1 with CDK2 complexes. Mitogenic stimulation and retinoic acid treatment of the B cells were performed as described in the legend to Fig 3B. Whole cell extracts were immunoprecipitated with antibody against CDK2. The immunoprecipitates were then examined for CDK2 (top) and p21Cip1 (bottom) levels by Western blot analysis. IP, immunoprecipitating antibody; W, Western blot antibody; μ, F(ab′)2 fragment of rabbit antihuman IgM; SAC, Staphylococcus aureus crude cell suspension; RA, retinoic acid.
Effects of retinoic acid on mRNA levels of cyclin E, cyclin A, and p21Cip1.
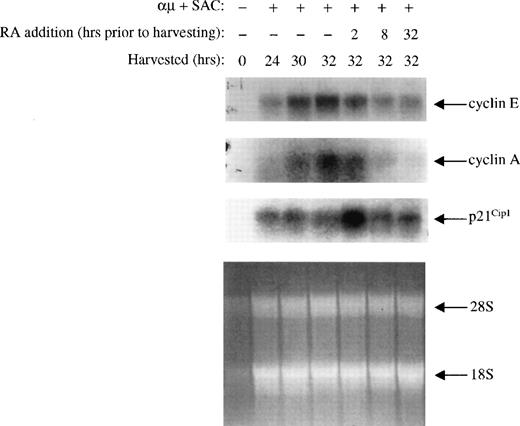
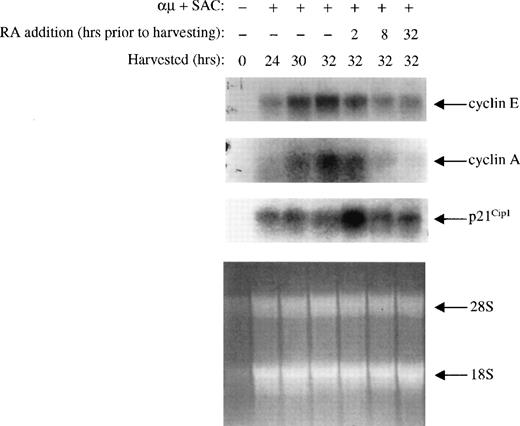
The changes noted in expression of the various cell cycle components were also measured at the mRNA level. By Northern blot analysis, we analyzed the steady state levels of cyclin E, cyclin A, and p21Cip1 mRNAs isolated from cells treated with or without retinoic acid. As shown in Fig 8, the mRNA levels of cyclin E and A were low in unstimulated cells, and both increased upon stimulation of the cells. Retinoic acid prevented the increase in mRNA expression of both cyclin E and A, and the kinetics was comparable to that of the protein levels.
Effect of retinoic acid on cyclin E, cyclin A, and p21Cip1 mRNA expression. Mitogenic stimulation and retinoic acid treatment of the B cells were performed as described in the legend to Fig 3B. mRNA from 25 × 106 cells was extracted and analyzed by Northern blotting using 32P-labeled cyclin E, cyclin A, and p21Cip1 probes as described in Materials and Methods. As a loading control we have shown the residual rRNA levels present in the different lanes. μ, F(ab′)2fragment of rabbit antihuman IgM; SAC, Staphylococcus aureuscrude cell suspension; RA, retinoic acid.
Effect of retinoic acid on cyclin E, cyclin A, and p21Cip1 mRNA expression. Mitogenic stimulation and retinoic acid treatment of the B cells were performed as described in the legend to Fig 3B. mRNA from 25 × 106 cells was extracted and analyzed by Northern blotting using 32P-labeled cyclin E, cyclin A, and p21Cip1 probes as described in Materials and Methods. As a loading control we have shown the residual rRNA levels present in the different lanes. μ, F(ab′)2fragment of rabbit antihuman IgM; SAC, Staphylococcus aureuscrude cell suspension; RA, retinoic acid.
The changes in the expression of p21Cip1 mRNA did also follow that of the protein expression. Thus, the level of p21Cip1 mRNA was low in unstimulated cells, and its level increased upon stimulation of the cells into G1. Furthermore, retinoic acid induced a rapid (within 2 hours) increase in the level of p21Cip1 mRNA, which declined to control levels within 8 hours of treatment.
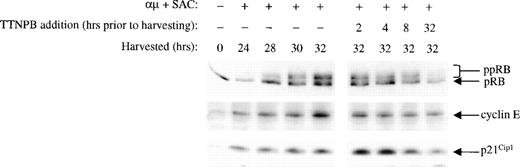
The RAR agonist TTNPB mimics and the RAR-selective antagonist Ro 41-5253 counteracts the effects of retinoic acid on the cell cycle machinery.
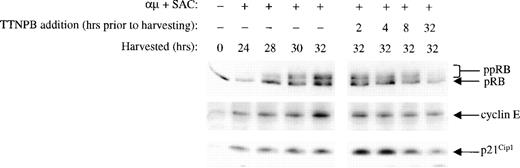
To exclude the possibility that the effects of retinoic acid on the cell cycle machinery were independent of the nuclear retinoic acid binding receptors, we analyzed the effect of the RAR-selective agonist TTNPB on some of the key events affected by retinoic acid. We have previously shown that an RAR agonist could inhibit proliferation of B lymphocytes,17 and in the present study we show that TTNPB grossly mimicked the effect of retinoic acid on phosphorylation of pRB, expression of cyclin E, and p21Cip1(Fig 9). Thus, TTNPB (100 nmol/L) inhibited both the pRB phosphorylation and cyclin E expression, whereas it induced a transient increase in the level of p21Cip1.
The RAR-specific agonist TTNPB mimics the effects of retinoic acid. Left panel: B cells (1.5 × 106/mL) were stimulated with μ (37 μg/mL) and SAC (0.005%) and harvested at the indicated times after stimulation. Right panel: Cells were stimulated with μ (37 μg/mL) and SAC (0.005%). TTNPB (100 nmol/L) was then added to the cell cultures at each indicated time before harvesting the cells at 32 hours. Total cell lysates were prepared and subjected to Western blot analysis for pRB (top), cyclin E (middle), and p21Cip1 (bottom). μ, F(ab′)2 fragment of rabbit antihuman IgM; SAC,Staphylococcus aureus crude cell suspension.
The RAR-specific agonist TTNPB mimics the effects of retinoic acid. Left panel: B cells (1.5 × 106/mL) were stimulated with μ (37 μg/mL) and SAC (0.005%) and harvested at the indicated times after stimulation. Right panel: Cells were stimulated with μ (37 μg/mL) and SAC (0.005%). TTNPB (100 nmol/L) was then added to the cell cultures at each indicated time before harvesting the cells at 32 hours. Total cell lysates were prepared and subjected to Western blot analysis for pRB (top), cyclin E (middle), and p21Cip1 (bottom). μ, F(ab′)2 fragment of rabbit antihuman IgM; SAC,Staphylococcus aureus crude cell suspension.
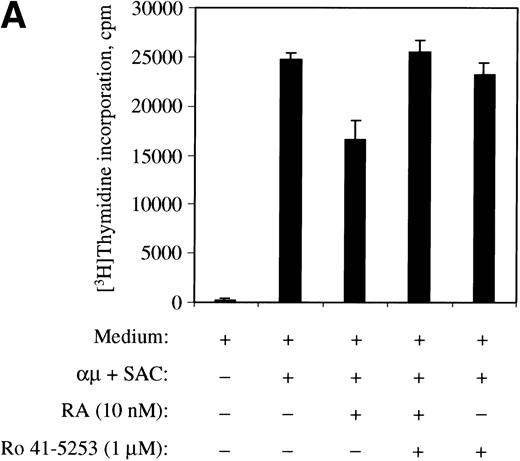
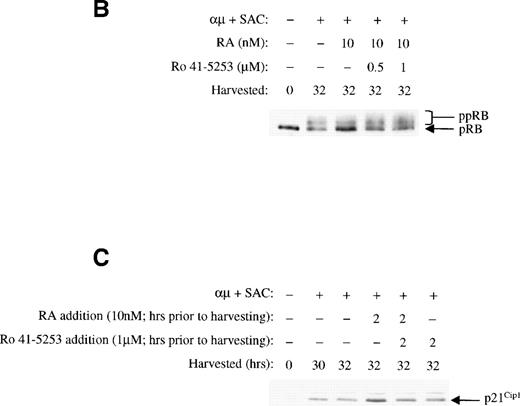
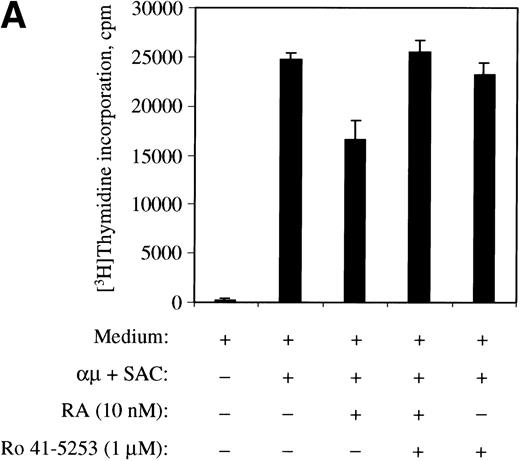
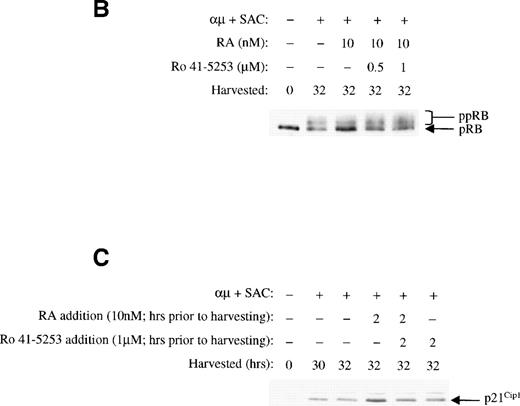
To further confirm the involvement of nuclear receptors in the actions of retinoic acid on the cell cycle machinery, we used the RARα antagonist Ro 41-5253. As shown in Fig10A, Ro 41-5253 (1 μmol/L) counteracted the inhibitory effect of retinoic acid on DNA synthesis. Furthermore, Ro 41-5253 prevented both the retinoic acid–mediated inhibition of pRB phosphorylation (Fig 10B) and the transient induction of p21Cip1 (Fig 10C). Note that we in the experiments presented in Fig 10 used retinoic acid at the concentration of 10 nmol/L, because the concentration of dimethyl sulfoxide (DMSO) in the solution of 1 μmol/L Ro 41-5253 was slightly toxic to the cells in the presence of higher concentrations of retinoic acid.
The RAR-selective antagonist Ro 41-5253 blocks the effects of retinoic acid. (A) The effect of Ro 41-5253 on DNA synthesis was measured as described in Materials and Methods, using Ro 41-5253 at a concentration of 1 μmol/L, and retinoic acid at the concentration of 10 nmol/L. (B) B cells (1.5 × 106/mL) were stimulated with μ (37 μg/mL) and SAC (0.005%) for 32 hours in the absence or presence of retinoic acid (10 nmol/L). For Ro 41-5253 treatment of the cells, Ro 41-5253 (0.5 or 1 μmol/L) was added to the cell cultures alone or together with retinoic acid and the cells were harvested at 32 hours. Total cell lysates were prepared and subjected to Western blot analysis for pRB as described in Material and Methods. (C) Ro 41-5253 (1 μmol/L) was added to the cell cultures alone or together with retinoic acid (10 nmol/L) at the indicated times before harvesting the cells at 32 hours. Total cell lysates were prepared and subjected to Western blot analysis for p21Cip1. μ, F(ab′)2 fragment of rabbit antihuman IgM; SAC,Staphylococcus aureus crude cell suspension.
The RAR-selective antagonist Ro 41-5253 blocks the effects of retinoic acid. (A) The effect of Ro 41-5253 on DNA synthesis was measured as described in Materials and Methods, using Ro 41-5253 at a concentration of 1 μmol/L, and retinoic acid at the concentration of 10 nmol/L. (B) B cells (1.5 × 106/mL) were stimulated with μ (37 μg/mL) and SAC (0.005%) for 32 hours in the absence or presence of retinoic acid (10 nmol/L). For Ro 41-5253 treatment of the cells, Ro 41-5253 (0.5 or 1 μmol/L) was added to the cell cultures alone or together with retinoic acid and the cells were harvested at 32 hours. Total cell lysates were prepared and subjected to Western blot analysis for pRB as described in Material and Methods. (C) Ro 41-5253 (1 μmol/L) was added to the cell cultures alone or together with retinoic acid (10 nmol/L) at the indicated times before harvesting the cells at 32 hours. Total cell lysates were prepared and subjected to Western blot analysis for p21Cip1. μ, F(ab′)2 fragment of rabbit antihuman IgM; SAC,Staphylococcus aureus crude cell suspension.
DISCUSSION
The present study is to our knowledge the first attempt to elucidate the effect of retinoic acid on the cell cycle machinery in normal cells, and in lymphocytes particularly. Our previous published data on the growth-inhibiting effect of retinoids on human B-lymphocytes8,10 are supported by several reports showing the inhibitory effect of retinoids on lymphoid growth.5,31,32 However, most reports regard retinoids as important activators of lymphoid proliferation.1-3Therefore, we found it important to examine the mechanisms whereby retinoic acid exerts its growth-inhibiting effects on normal B cells.
In our previous report, we showed that retinoids, including retinoic acid, blocked the cell cycle progression of human B lymphocytes in mid to late G1.8 We defined the point of inhibition to be somewhere between 24 hours into G1 and the restriction point, the restriction point being located 30 to 35 hours into G1 of human B lymphocytes.26 In the present study, we wished to define the part of the cell cycle machinery that was affected by retinoic acid. Therefore, we added retinoic acid to the B-lymphocyte cultures at different times during G1, and all the cells were harvested at 32 hours. Because phosphorylation of pRB is one of the critical events in the regulation of G1/S transition,12 we first studied the effect of retinoic acid on this component of the cell cycle machinery. Indeed we observed that concentrations of retinoic acid as low as 1 to 10 nmol/L, ie, physiological levels, were able to prevent pRB phosphorylation. Effects were noted even when retinoic acid was added as late as 2 hours before the restriction point in G1.
The key regulators of pRB phosphorylation in early to mid G1 are the cyclin-dependent kinases CDK4 and CDK6, whereas CDK2 has a role in late G1 and at the G1/S transition.12 These kinases are activated through their binding to specific cyclins and are inhibited by specific CKIs.11 We examined the effects of retinoic acid on the various G1 kinases, and we found that the activity of CDK2 was reduced. We did not detect CDK4 activity in the isolated B cells, and CDK6 activity was not affected by retinoic acid. The inhibition of CDK2 activity could be due to reduced levels of cyclin E or cyclin A, or it could be due to the increase in the level of one of the CKIs. We observed that retinoic acid prevented expression of both cyclin E and cyclin A, and we also noted a transient increase in the expression of the CKI p21Cip1 2 to 4 hours after addition of retinoic acid. The increased expression of p21Cip1 was associated with increased binding to CDK2. No effects were observed on the levels of cyclin D2 and cyclin D3, or on the level of p27Kip1. Importantly, both the transient induction of p21Cip1 and the reduced expression of cyclin E and A could also be detected when retinoic acid was added to the cells early in G1. Thus it appears that these events could be the cause rather than the result of the cell cycle arrest mediated by retinoic acid in mid to late G1.
Effects on the cell cycle machinery have also been noted in other cell systems that are growth-inhibited by retinoids. Zhou et al33 have reported an inhibitory effect of retinoids on the expression of D-type cyclins and CDKs, whereas, in agreement with our present results on B lymphocytes, retinoic acid has been shown to reduce the level of cyclin E in HL-60 cells34 and in bronchial epithelial cell lines.35 Effects of retinoic acid on CKIs have also been reported. Increased levels of p27Kip1 upon treatment with retinoic acid have been shown in astrocytoma cells36 as well as in neuroblastoma cells.37 Increased levels of p27Kip1 are, however, commonly observed in growth-inhibited cells.28 38This is usually a late event in growth inhibition that is observed several hours after the cells have received the growth- inhibitory signal. It is therefore possible that the increase in the levels of p27Kip1 is the result rather than the cause of growth inhibition. We did also note a small increase in p27kip1levels upon treatment of B cells with retinoic acid for 32 hours, but we regard this to be a secondary effect.
Retinoic acid has, in agreement with our present results, been reported to increase the level of p21Cip1. For instance, Liu et al39 showed that differentiation of U937 cells with retinoic acid was associated with induced expresssion of p21Cip1. However, in MCF-7 cells treated with retinoic acid the level of p21Cip1 was reduced.40 Thus, it appears that the effect of retinoic acid on p21Cip1 depends on the cell system. It is, however, puzzling that the increase in the expression of p21Cip1 and in its binding to CDK2 that we observed was only transient, whereas the activity of CDK2 was permanently reduced. A similar transient induction of p21Cip1 was observed at the mRNA level in HL60R cells that were growth inhibited by a novel synthetic retinoid, CD437,16 and by TGF-β in epithelial cells.41 It is, however, important to note that the relation between the level of p21Cip1 and its role as a CKI is not a simple one. In fact, it has recently been reported that p21Cip1 at low concentrations can function as an assembly factor between cyclins and CDKs, and thereby is required for activation of CDKs.42,43,44 This could explain the moderate increase in the expression of p21Cip1 that was noted upon stimulation of the B cells into G1. At higher concentrations, however, p21Cip1 acts as a CKI.11 Therefore, it appears that the ability of p21Cip1 to act as an inhibitor or activator of CDKs depends on the relative levels of cyclins, CDKs, and of p21Cip1 itself. It is reasonable to assume that the early retinoic acid-mediated increase in the expression of p21Cip1 could lead to the rapid inhibition of CDK2 activity that we observed. Upon longer (more than 4 hours) exposure to retinoic acid, the reduction in CDK2 activity could be due to the substantial decrease in cyclin E and cyclin A levels.
It has been an issue of debate whether or not nuclear receptors mediate all the effects of retinoids in vivo. In target cells, vitamin A is metabolized into retinoic acid, and retinoic acid mediates its effect via nuclear RARs.45,46 However, vitamin A can also be metabolized into retro-retinoids such as anhydroretinol (AR) and 14-hydroxy-4, 14 retro-retinol (14-HRR), which do not function as regulators of transcription.15 AR has been shown to act as a growth inhibitor for lymphocytes,47 whereas 14-HRR appears to be required for normal lymphocyte proliferation.15 Although retinoic acid does not appear to be metabolized into retro-retinoids in vivo, it was still important to examine whether or not nuclear retinoid-binding receptors mediate the effects of retinoic acid on the cell cycle machinery. We have previously shown that normal B cells express the nuclear receptors RARα, RΑRγ, and retinoid X receptor α (RXRα), and that the RAR agonist TTNBP could mimic the effect of retinoic acid on growth and apoptosis in normal human B lymphocytes.17 In the present study, TTNBP essentially functioned as retinoic acid in regulating the components of the cell cycle machinery. It prevented pRB phosphorylation and cyclin E expression, and transiently induced p21Cip1 expression.
To do the opposite experiments, ie, to block the effects of retinoic acid, we used the RAR-selective antagonist Ro 41-5253. Ro 41-5253 prevented the inhibition of both DNA synthesis and of pRB phosphorylation, and it prevented the transient increase in the expression of p21Cip1 induced by retinoic acid. It has been shown that the gene coding for p21Cip1 possesses a RAR/RXR-responsive element in its promoter,39 and in the present study we showed that retinoic acid altered the expressions of cyclin E, cyclin A, and p21Cip1 at the mRNA levels. Thus, the present findings support the notion that retinoic acid mediates its effects on the cell cycle machinery at the transcriptional level via nuclear RARs.
Taken together, the present study firmly establishes the inhibitory role of retinoic acid on the cell cycle progression of normal human B lymphocytes. The key event of pRB phosphorylation near the restriction point in G1 is prevented by retinoic acid, and it appears that this effect is primarily due to downregulation of cyclin E, cyclin A, and induced expression of p21Cip1. We are currently in the process of analyzing the effects of the various retro-retinoids on the cell cycle machinery in normal B lymphocytes, to further unravel the role of retinoids in lymphoid proliferation.
ACKNOWLEDGMENT
The authors thank Hilde R. Haug for excellent technical assistance.
Supported by The Norwegian Cancer Society, The Norwegian Research Council, Freia Research Foundation, and Jahre Research Foundation.
The publication costs of this article were defrayed in part by page charge payment. This article must therefore be hereby marked “advertisement” in accordance with 18 U.S.C. section 1734 solely to indicate this fact.

![Fig. 1. Effect of retinoic acid on DNA synthesis. Human B cells were isolated and stimulated with μ (37.5 μg/mL) and SAC (0.005%) as described in Material and Methods. Cells (0.75 × 106 cells/mL) were cultured in 96-well culture plates in triplicates. Retinoic acid (10 or 1000 nmol/L) was added at the indicated times (T) after stimulation. [3H]Thymidine incorporation (mean ± SD) was measured as described in Material and Methods. One representative experiment of three is shown. μ, F(ab′)2 fragment of rabbit antihuman IgM; SAC,Staphylococcus aureus crude cell suspension; RA, retinoic acid.](https://ash.silverchair-cdn.com/ash/content_public/journal/blood/94/4/10.1182_blood.v94.4.1348/4/m_blod41629001x.jpeg?Expires=1770288747&Signature=LHAnH0H412qNETZ~6czvD6ZrGnjzCZ4AT9A02C0erjB-SjNp7dxNBhpDfQdU1LGD91MOy8b9rE5ZgWJYLlSpToxOVPPDUlXF~zGcMD~uX4v35e2WMdgBTrMXrT1xaIbQDUVGrDzzEUqW07oa6BkfLnwR4Nh3SWkXfrUeE8FkmV5GNPCXBJVFE7KcboHh~gVP9FUVHJG9LUBFBf7Bpu3F2dLECiAi78M6s7RrBYnDlcYoAQ~zWhIXCxo9od3QtJQFox209EbNAYW1bA88xi0gJtVF5yLXh7QLQ94Fi0dVPqsVkwvbXS7QVdqC-8MDNU~TC6KN9jWMmXSMawvrRo4Wmw__&Key-Pair-Id=APKAIE5G5CRDK6RD3PGA)

![Fig. 1. Effect of retinoic acid on DNA synthesis. Human B cells were isolated and stimulated with μ (37.5 μg/mL) and SAC (0.005%) as described in Material and Methods. Cells (0.75 × 106 cells/mL) were cultured in 96-well culture plates in triplicates. Retinoic acid (10 or 1000 nmol/L) was added at the indicated times (T) after stimulation. [3H]Thymidine incorporation (mean ± SD) was measured as described in Material and Methods. One representative experiment of three is shown. μ, F(ab′)2 fragment of rabbit antihuman IgM; SAC,Staphylococcus aureus crude cell suspension; RA, retinoic acid.](https://ash.silverchair-cdn.com/ash/content_public/journal/blood/94/4/10.1182_blood.v94.4.1348/4/m_blod41629001x.jpeg?Expires=1771177768&Signature=2jSR4vXQcT89ooWnrr40PqkXfhu4PgKnXv5hfWKNe71Kqwz8PJfro-0QDBeeD5fY1REIdJiem5Bi-hFQbWYFbWvBJW9AA3kOHz4hZqvVmqnwgJUSMQWkdtVaVFUVmRkSy6Xxuh1hzPlnEBZhWWeALJsS1TZv~p-sVoV9hi7MBzHBXlvzNxByRw7-tc248TxRuYVDaB1FF4REz1uIAO4hjTVCBbGzE6V1OCih8X-i212nZLLV-AhFeHWLexHcv8SJiGp-vFZ-2pdSxDY6hRwj-drvu43SM0fRvfriQWfgXP1dz6S3NRQQcdmI7rFjEuK3nu2jaiMnbS3NpWIHO9jSvw__&Key-Pair-Id=APKAIE5G5CRDK6RD3PGA)