During chronic HIV infection, asymptomatic individuals demonstrate a strong CD8+ cell noncytotoxic antiviral response (CNAR). With the onset of symptoms or reduction in CD4+ cell counts, CNAR decreases. Presently, it is recommended that infected individuals receive antiretroviral therapy if CD4+ cell counts fall below 350 cells/μL. To determine whether CNAR lends support to this recommendation for initiation of antiretroviral treatment, we examined CNAR in 20 healthy, untreated, HIV-infected men exhibiting a range of CD4+ cell numbers. Our results indicate that the asymptomatic untreated HIV-infected individuals with less than 300 CD4+ cells/μL had a significantly lower CNAR than those with higher CD4+ cell counts. These data on CNAR in untreated, healthy, HIV-infected individuals support the current recommendation for when to initiate antiretroviral therapy.
Introduction
In HIV-infected individuals, antiretroviral therapy is recommended when CD4+ cell counts fall below 350 cells/μL, viral load rises above 30 000 copies/mL, or HIV-related symptoms appear.1 Although immunologic responses also decrease during chronic infection, this function has not been used to determine when antiretroviral therapy should be initiated. One of the immunologic responses reduced over time in HIV-infected individuals is the CD8+ cell noncytotoxic antiviral response (CNAR).
CNAR is first observed during the acute stage of HIV infection and then can be maintained for more than 10 years.2 In chronic HIV–infected individuals, the extent of CNAR correlates directly with the clinical state and the CD4+ cell counts.2-7 For example, chronic HIV–infected individuals who are asymptomatic and have more than 500 CD4+ cells/μL have a strong CNAR. Conversely, individuals who are symptomatic and have lower CD4+ cell counts have a significantly weaker CNAR.3,5,6,8 The clinical importance of CNAR is strongly suggested by studies of HIV and simian immunodeficiency virus infection in primates. Administration of anti–CD8 antibodies caused a return in virus production in these animals.9-11 The rapid rebound in viremia appears to reflect a noncytotoxic mechanism for control of virus replication.
Our goal was to determine whether CNAR in untreated asymptomatic individuals could provide an immunologic measure that supports the time now recommended to initiate antiretroviral therapy. To address this question, we compared the extent of CNAR in CD8+ cells from 20 randomly selected, untreated, asymptomatic individuals having a broad range of CD4+ cell counts.
Study design
Study subjects
After informed consent, peripheral blood was drawn from a cohort of 20 randomly selected men, chronically infected with HIV, who were clinically healthy and not on therapy. Of the 20 subjects, 2 completed antiretroviral treatment 3 years and 5 years before enrollment, whereas all others were therapy-naive. All subjects contracted HIV through sexual contact and with the exception of one individual (infected 2 years before enrollment), seroconverted more than 10 years before enrollment. Plasma viral loads were measured by the bDNA technique.12 This study received the approval of the Committee for Human Research, University of California, San Francisco.
Cell culture
Blood samples from clinically healthy seronegative and from HIV-1– seropositive subjects were collected by venipuncture. Complete blood counts and T-cell subset identification was assessed from ethylenediaminetetraacetic acid (EDTA)–treated blood. Peripheral blood mononuclear cells (PBMCs) were isolated from the heparinized blood by Ficoll gradient separation.13 Cells were cultured at 3 × 106 cells/mL in complete RPMI 1640 medium, containing 10% heat-inactivated (56°C, 30 minutes) fetal bovine serum (Gibco BRL, Rockville, MD), 1% penicillin/streptomycin, 2 mM glutamine, and 10% purified human interleukin 2 (IL-2) (Roche Diagnostics, Indianapolis, IN).
Acute infection assay
From the PBMCs of seronegative individuals, CD4+cells were obtained by immunomagnetic (IM) bead separation (Miltenyi Biotech, Auburn, CA), cultured at 3 × 106 cells/mL in complete medium, and supplemented with 10% purified human IL-2. CD4+ cells were then stimulated with phytohemaglutinin-leucogglutinin (PHA-L) (3 μg/mL) for 3 days at 37°C before being infected with HIV-1. For recovery of CD8+ cells, PBMCs from seropositive subjects were first stimulated with 3 μg/mL PHA-L (Sigma Chemicals, St Louis, MO) for 3 days.
After 3 days of PHA stimulation, CD8+ cells were isolated from the seropositive PBMC cultures by positive selection with IM beads. The PHA-stimulated CD4+ cells were infected with HIV-1SF33, a β-chemokine–insensitive, CXCR4-dependent viral isolate, at a titer in which peak reverse transcriptase (RT) activities14 were reached at 7 days in infected CD4+ cells cultured alone.8 CD8+cells and CD4+ cells were cocultured in duplicate at input CD8+:CD4+ cell ratios ranging from 0.25:1 to 2:1, in complete medium supplemented with 100 U/mL recombinant human IL-2 (generously provided by Glaxo IMB, Geneva, Switzerland). Culture supernatants were monitored for RT activity and percent suppression calculated as described previously.8
Statistical analysis
Exact Wilcoxon rank sum tests were used to evaluate differences in the distributions of markers of antiviral response for symmetry and the presence of outliers between individuals with CD4+ cell counts of more than or less than 300. Differences were deemed significant if exact P values were less than .05. For analysis purposes, a limit of detection (50 HIV RNA copies/mL) was set for undetectable viral load values.
Results and discussion
Study group characteristics
A comparison of 20 randomly selected subjects showed a distinct division in the range of CD4+ cell counts at more than 481 CD4+ cells/μL (> 300 CD4+ cells/μL) and at less than 300 CD4+ cells/μL (< 300 CD4+cells/μL) (Table 1).
CD4+ cell count, CD8+ cell count, and plasma viral loads differed significantly between the 2 groups. The mean CD4+ cell count for those individuals in the group with higher CD4+ cell counts was 629 cells/μL whereas the mean cell number for those individuals in the group with lower CD4+ cell counts was 220 cells/μL (P = .001). CD8+ cell numbers were higher (1605 CD8+ cells/μL) in individuals with higher CD4+ cell counts than in individuals with lower CD4+ cell counts (677 CD8+ cells/μL) (P = .01). Plasma viral load values were lower (4173 HIV RNA copies/mL) in individuals with higher CD4+ cell counts than in individuals with lower CD4+ cell counts (26 314 HIV RNA copies/mL) (P = .03). T-cell subsets and plasma viral load values, obtained at the time CNAR was assessed, are presented in Table 1 as median and range (in parentheses). Pvalues were determined by Wilcoxon rank sum test for paired samples.
Antiviral response
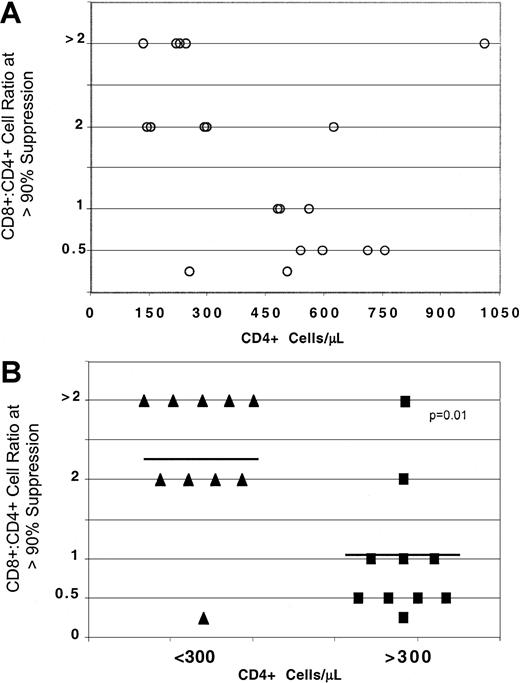
By analyzing suppression of HIV replication as a function of CD4+ cell number, a difference in the extent of suppression was observed in individuals with less than 300 CD4+cells/μL in comparison to those individuals with higher CD4+ cell counts (Figure 1A).
CD8+ cell noncytotoxic antiviral response in HIV-infected individuals with different CD4+ cell numbers.
CD8+ cells from HIV-seropositive individuals were isolated from PHA-stimulated PBMCs and cocultured at different input cell ratios with PHA-stimulated seronegative CD4+ cells, acutely infected with HIV-1SF33. The ratio of CD8+:CD4+ cells required to reach more than or equal to 90% suppression of HIV replication is indicated for each infected individual (A) within a range of CD4+ cells/μL (○) and (B) after dividing the subjects into 2 groups either with less than (▴) or greater than (▪) 300 CD4+ cells/μL. Percent suppression is calculated from the amount of RT activity14 in the supernatants of the cocultures of CD8+ and CD4+ cells compared to the RT activity in the supernatants from infected CD4+ cells cultured alone. Solid lines represent the average CD8:CD4+ cell ratio for more than or equal to 90% suppression.
CD8+ cell noncytotoxic antiviral response in HIV-infected individuals with different CD4+ cell numbers.
CD8+ cells from HIV-seropositive individuals were isolated from PHA-stimulated PBMCs and cocultured at different input cell ratios with PHA-stimulated seronegative CD4+ cells, acutely infected with HIV-1SF33. The ratio of CD8+:CD4+ cells required to reach more than or equal to 90% suppression of HIV replication is indicated for each infected individual (A) within a range of CD4+ cells/μL (○) and (B) after dividing the subjects into 2 groups either with less than (▴) or greater than (▪) 300 CD4+ cells/μL. Percent suppression is calculated from the amount of RT activity14 in the supernatants of the cocultures of CD8+ and CD4+ cells compared to the RT activity in the supernatants from infected CD4+ cells cultured alone. Solid lines represent the average CD8:CD4+ cell ratio for more than or equal to 90% suppression.
A statistically lower number of CD8+ cells (average cell ratio of 1.1:1) was required to reach 90% suppression of HIV replication in the group with higher CD4+ cell counts than in the group with less than 300 CD4+ cells (average cell ratio of 2.35:1) (P = .01) (Figure 1B). In some subjects with less than 300 CD4+ cells/μL, the CD8+:CD4+ cell input of more than 2:1 in the coculture (Figure 1B) indicated an inability of CNAR to reach 90% suppression at the ratio of 2:1.
There were 2 subjects in the present study who represented exceptions. One showed strong CNAR with CD4+ cell counts below 300 cells/μL. This individual did not have a substantially different CD4+ cell count, CD8+ cell count, or plasma viral load from the other individuals in this group. The other individual had poor CNAR but an exceptionally high CD4+cell count (1033 cells/μL) and an undetectable viral load. This individual had the highest CD4+ cell count of the cohort along with a higher percentage of CD4+ cells than CD8+ cells. Both individuals seroconverted more than 10 years before enrollment in the study and were antiretroviral therapy–naive.
The past and these present findings suggest that the HIV-infected individual with a strong CNAR, despite the low CD4+ cell count, would remain healthy for a longer time than the individual with a low CNAR and high CD4+ cell count.2 5-8Other subjects who show similar features need to be followed over time to assess this possibility.
Using an exploratory analysis of the extent of CNAR in association with viral load, we observed a trend between low CNAR and high viral load (data not shown). Nevertheless, the number of subjects in each CD4+ cell number group was too low to conclude statistically a relationship of CNAR to viral load.
The present study of untreated HIV-infected individuals, with no HIV-related symptoms, confirms that the extent of CNAR correlates with CD4+ cell counts. Asymptomatic individuals with CD4+ cell counts below 300 cells/μL showed less ability to suppress HIV replication than those with higher CD4+cells counts. The findings provide immunologically based evidence supporting the current recommendation for the time to initiate combination antiretroviral therapy.1 The results suggest that CNAR could be an additional marker to help determine the optimal time to begin antiretroviral treatment.
The authors thank Sue Fujimura for her technical assistance.
Prepublished online as Blood First Edition Paper, April 17, 2002; DOI 10.1182/blood-2001-11-0078.
Supported by a grant from the National Institutes of Health (RO1 AI49926-01). J.C.C. is a trainee under the National Institutes of Health Training Program (T32 AI07395).
The publication costs of this article were defrayed in part by page charge payment. Therefore, and solely to indicate this fact, this article is hereby marked “advertisement” in accordance with 18 U.S.C. section 1734.
References
Author notes
Jay A. Levy, UCSF, Department of Medicine, San Francisco, CA 94143-1270; e-mail: jalevy@itsa.ucsf.edu.