Abstract
We report 28 patients with advanced chronic graft-versus-host disease (cGVHD) treated with extracorporeal photopheresis (ECP). All had failed conventional immunosuppressive therapy. Of the patients, 27 had extensive cGVHD and 20 had more than 50% cutaneous surface area involvement. ECP was initiated approximately 2 years after onset of cGVHD and 3 years following allogeneic stem cell transplantation and administered fortnightly for 4 months and then monthly. Response was assessed using quantifiable disease measures, including skin score, liver function tests (LFTs), blood counts, and lung function tests. Regression analysis allowed assessment of any pretreatment clinical or laboratory parameters that predicted response. There were 25 patients who completed 3 months and 21 who completed 6 months of treatment. Systemic immunosuppression was stable or reduced in 86% of patients. There were 3 patients who died from cGVHD. After 6 months, median skin scores were 53% lower (P = .003) in sclerodermoid and lichenoid disease. Of 6 patients with mucosal ulceration, 3 improved. A nonsignificant improvement of LFTs occurred. We infer that ECP is effective even in patients with extensive cutaneous cGVHD of 2 years duration that is resistant to conventional therapy. Furthermore, both sclerodermoid and lichenoid subtypes responded. However, no baseline parameters predicted a favorable response to ECP, so patient selection must continue to be made on clinical grounds.
Introduction
Chronic graft-versus-host disease (cGVHD) affects more than 50% of long-term survivors of allogeneic stem cell transplantation.1,2 The incidence of cGVHD is likely to rise further with the greater availability and use of HLA-matched unrelated donors and the increasing use of allogeneic peripheral blood as a source of stem cells. cGVHD is an important cause of morbidity and mortality in such patients and accounts for one quarter and two thirds of deaths following transplantations performed for leukemia and severe aplastic anemia, respectively.3,4 Recognized treatments for cGVHD utilize agents used in the treatment of autoimmune disorders and are associated with response rates of 20% to 80%, but may cause significant toxicity.5,6
Phototherapy has also been used experimentally and clinically to modify GVHD. Antigen-presenting cells and T lymphocytes are susceptible to photoinactivation with either ultraviolet B irradiation or ultraviolet A irradiation (UVA) in the presence of a photosensitizing agent such as 8-methoxypsoralen (8-MOP). These effects have been used experimentally to prevent the development of GVHD in murine models.7,8 Clinically, UVA phototherapy may be administered to the skin surface as PUVA (ie, psoralen and UVA irradiation) or directly to peripheral blood leukocytes obtained from the patient by apheresis. Concentrated leukocytes are incubated with 8-MOP, irradiated, and then reinfused, a technique known as extracorporal photopheresis (ECP). ECP was initially developed in the 1980s for the treatment of clonal T-cell disorders such as cutaneous T-cell lymphoma and Sezary syndrome.9,10 In randomized studies, it has proved effective in preventing posttransplantation organ rejection and reversing episodes of cardiac rejection.11,12 Since 1994, several uncontrolled case series have reported successful treatment of both acute and chronic GVHD with ECP. To date there are published data on 67 adult patients and 20 pediatric patients with cGVHD that suggest that skin and mucosal disease are more responsive to ECP than systemic manifestations such as hepatic, gastrointestinal, and pulmonary disease.13-24
In this study we describe the clinical effects of ECP in 28 patients with refractory cGVHD and correlate changes over time with quantitative estimates of CD4, CD8, and natural killer (NK) cell populations. Because this is the largest series reported to date, we were able to perform regression analysis in an attempt to identify baseline clinical and laboratory parameters that were predictive of outcome.
Patients and methods
Patients
Entering the study were 28 adult patients of a median age of 34 years (range, 18-51 years). Preliminary data on 5 of these patients have been previously reported.19 Patients were referred from hospitals in southern England and were treated with ECP in the Skin Tumour Unit, St John's Institute of Dermatology, St Thomas' Hospital in London, England. Enrolled patients were adults who had developed cGVHD following treatment of hematologic malignancies with HLA-matched bone marrow or peripheral blood stem cell allografts and who were refractory to conventional immunosuppressive treatment. To assess the severity of cGVHD in our patient cohort, disease was classified at baseline as either extensive or limited using the criteria of Shulman et al.25 In addition, the extent of cGVHD was assessed using prognostic criteria that recently have been identified as important predictors of disease-specific survival in cGVHD; these criteria are cutaneous involvement of more than 50%, platelet count of less than 100 × 109/L, and the presence of progressive cGVHD.26 Of the patients, 27 had received 2 or more immunosuppressive/immunomodulatory systemic treatments and 5 had received 4 or more treatments. Previously, 5 patients had received at least one course of PUVA therapy, although treatment had been discontinued at an early stage in 1 patient who did not tolerate oral psoralen administration. ECP was undertaken in 3 of these patients who had gone on to develop systemic cGVHD. The other patient had cutaneous cGVHD with sclerodermoid features that had not responded to PUVA therapy. ECP treatment was considered only if the following criteria were met: histologically established cGVHD, adequate hemodynamic and cardiac function, body weight more than 40 kg, hemoglobin count higher than 90 g/L (9 g/dL), platelet count higher than 20 × 109/L, and neutrophil count higher than 1 × 109/L. Approval for the study was obtained from the Local Research Ethics Committee of Guy's and St. Thomas' Hospital National Health Service Trust for these studies according to the Declaration of Helsinki.
The pretreatment characteristics of the group are illustrated in Table 1.
Characteristics of study population (N = 28)
Characteristic . | No. . |
|---|---|
| Male | 20 |
| Female | 8 |
| Hematologic malignancy | |
| Acute lymphoblastic leukemia | 5 |
| Chronic myeloid leukemia | 14 |
| Acute myelogenous leukemia | 6 |
| Myeloma | 3 |
| Donor | |
| Sibling | 19 |
| Matched unrelated | 9 |
| Donor lymphocyte infusion | 11 |
| cGVHD disease type | |
| Progressive | 9 |
| De novo | 14 |
| Relapsed | 5 |
| Predictive factors | |
| More than 50% cutaneous involvement | 20 |
| Progressive cGVHD | 9 |
| Platelet count less than 100 × 109/L | 1 |
| No predictive factors | 4 |
| 1 of 3 predictive factors | 17 |
| 2 of 3 predictive factors | 7 |
| Systemic therapy | |
| Prednisolone/methylprednisolone | 26 |
| Cyclosporin | 24 |
| Mycophenolate mofetil | 7 |
| Thalidomide | 5 |
| PUVA | 5 |
| Azathioprine | 4 |
| Tacrolimus (FK 506) | 2 |
| Methotrexate | 1 |
| Cyclophosphamide | 1 |
| Median delay before starting ECP, mo (range) | |
| From diagnosis | 45 (15-193) |
| From allograft | 34 (10-167) |
| From onset of cGVHD | 23 (2-164) |
Characteristic . | No. . |
|---|---|
| Male | 20 |
| Female | 8 |
| Hematologic malignancy | |
| Acute lymphoblastic leukemia | 5 |
| Chronic myeloid leukemia | 14 |
| Acute myelogenous leukemia | 6 |
| Myeloma | 3 |
| Donor | |
| Sibling | 19 |
| Matched unrelated | 9 |
| Donor lymphocyte infusion | 11 |
| cGVHD disease type | |
| Progressive | 9 |
| De novo | 14 |
| Relapsed | 5 |
| Predictive factors | |
| More than 50% cutaneous involvement | 20 |
| Progressive cGVHD | 9 |
| Platelet count less than 100 × 109/L | 1 |
| No predictive factors | 4 |
| 1 of 3 predictive factors | 17 |
| 2 of 3 predictive factors | 7 |
| Systemic therapy | |
| Prednisolone/methylprednisolone | 26 |
| Cyclosporin | 24 |
| Mycophenolate mofetil | 7 |
| Thalidomide | 5 |
| PUVA | 5 |
| Azathioprine | 4 |
| Tacrolimus (FK 506) | 2 |
| Methotrexate | 1 |
| Cyclophosphamide | 1 |
| Median delay before starting ECP, mo (range) | |
| From diagnosis | 45 (15-193) |
| From allograft | 34 (10-167) |
| From onset of cGVHD | 23 (2-164) |
ECP procedure
ECP was performed using intravenous Uvadex (liquid methoxsalen) with either the UVARS photopheresis system (from 1994-1999) or the UVAR XTS photopheresis system (1999-present) (Therakos, Ascot, United Kingdom) as previously described.19 Venous access was obtained peripherally (22 patients) or via a Hickman line (6 patients). Each photopheresis procedure lasted 3 to 4 hours in total.
Treatment protocol
Only patients giving fully informed consent received treatment. Subjects received ECP on 2 consecutive days, fortnightly for the first 4 months, and then monthly. At 6 months a decision was made whether to continue treatment depending upon the clinical response, the inconvenience of traveling long distances, and the patients' own preferences. Whenever possible, changes in systemic immunosuppressive medication during treatment were avoided and any alterations were noted. Adverse events and reasons for discontinuation of treatment were recorded.
Assessment of extent of disease
Cutaneous cGVHD. All patients were fully examined. To allow assessment of clinical response to treatment of skin involvement, a quantitative measure of cutaneous cGVHD was obtained at each patient attendance by a member of the staff trained in an appropriate method of skin scoring. All staff received common training and individual subjects were assessed whenever possible by the same observer on each attendance to reduce the effect of interobserver variation. Extent of cutaneous cGVHD at diagnosis has been validated as the most important predictor of survival.26 We used a technique to provide a quantitative assessment of cutaneous cGVHD disease activity during treatment by combining surface area involvement and severity of disease in each area of skin assessed. The measuring technique was adapted from the modified Rodman scleroderma skin scoring method, which has been validated and extensively used in the assessment of sclerodermatous changes in systemic sclerosis.27 Clinical and histopathologic features of cutaneous sclerodermoid-type cGVHD and diffuse systemic sclerosis are virtually indistinguishable. Areas of inflamed lichenoid cGVHD were assessed using a similarly adapted method that combined surface area involvement and extent of inflammation. This technique classifies the presence of both erythema and lichenoid lesions in an area of skin in a higher group than either feature alone, because their combination is associated with increased histologic inflammation and keratinocyte apoptosis and therefore is consistent with more active disease. The skin scores were calculated using the following formula: total skin score = [(percentage of body surface area containing erythematous or lichenoid lesions × grade of erythema)] + [percentage body surface area containing sclerodermoid lesions × grade of sclerodermoid change] (Table 2).
Grading system for cutaneous cGVHD
. | Characteristic . |
|---|---|
| Grading of erythema/lichenoid disease | |
| Grade 0 | No lesions |
| Grade 1 | Erythema or lichenoid lesions |
| Grade 2 | Erythema and lichenoid lesions |
| Grading of sclerodermoid disease | |
| Grade 0 | Normal skin thickness |
| Grade 1 | Thickened skin |
| Grade 2 | Thickened and fixed skin |
| Grade 3 | Hidebound |
. | Characteristic . |
|---|---|
| Grading of erythema/lichenoid disease | |
| Grade 0 | No lesions |
| Grade 1 | Erythema or lichenoid lesions |
| Grade 2 | Erythema and lichenoid lesions |
| Grading of sclerodermoid disease | |
| Grade 0 | Normal skin thickness |
| Grade 1 | Thickened skin |
| Grade 2 | Thickened and fixed skin |
| Grade 3 | Hidebound |
Cutaneous biopsies were performed from the clinically most representative area of skin prior to starting ECP to confirm the diagnosis of cGVHD and to establish whether the changes were predominantly lichenoid, predominantly sclerodermoid, or mixed. Serial clinical photographs were also used to record extent of mucocutaneous disease.
Oral mucosal surfaces were examined, photographed, and classified as normal, pigmentary changes only, inflammatory changes, or mild/severe ulceration.
Hepatic cGVHD. Diagnoses of hepatic cGVHD had been made before the study on clinical grounds by the referring clinician on the basis of biochemical abnormalities of liver function in patients for whom other causes of hepatic dysfunction had been excluded and who had histologically confirmed cGVHD at other sites. It was not considered ethically justifiable to perform pretreatment and posttreatment liver biopsies to assess treatment response in view of the potential complications of this procedure. Hepatic cGVHD is characterized by mixed cholestatic and hepatocellular abnormalities with elevation of both alkaline phosphatase and transaminases.25 Typically, response to hepatic cGVHD to treatment is associated with reduction of liver function enzymes within 2 to 4 weeks, although values may not normalize completely without resolution of cGVHD in other organ systems.28 In this study, the response of hepatic cGVHD was assessed using serial laboratory investigations; total bilirubin, alkaline phosphatase, alanine transaminase (ALT), and γ-glutaryl transferase (γGT) levels were obtained from serum samples before each treatment cycle.
Pulmonary cGVHD. Measurements of pulmonary function tests included vital capacity, forced expiratory volume in one second, total carbon monoxide gas transfer, and coefficient of carbon monoxide diffusion. Repeated measurements were performed for patients with abnormal baseline values.
Neuromuscular cGVHD. One patient who developed symptoms consistent with mixed sensorimotor peripheral neuropathy underwent electromyography (EMG) before and during ECP treatment.
Laboratory analyses
A complete blood count, differential white count, and biochemical profile were performed at each visit. Lymphocyte subsets were analyzed at baseline and then monthly for CD4+, CD8+, and CD16+/CD56+ NK cells using flow cytometric analysis (Beckman Coulter color flow cytometry analyzer; Beckman Coulter, High Wycombe, United Kingdom).
Statistical methodology
Non-normally distributed parametric data are summarized using median values and ranges. Statistical analysis of trends in measured parameters during treatment is analyzed using paired Student t test, comparing values during treatment with baseline values. Unpaired t tests were used to compare baseline values of subjects who responded to treatment with those who did not. Backwards step-wise logistic regression analysis was used to assess whether baseline parameters predicted outcome of cutaneous or hepatic responder status at 3 or 6 months. Spearman rank correlations were performed to assess association between skin scores, liver function tests (LFTs), and hematologic parameters with time.
Results
Entered into the study were 28 patients with treatment-resistant cGVHD; they received at least one cycle of ECP. Using the criteria of Shulman et al, 27 patients had extensive cGVHD and a single patient with isolated mucosal disease had limited cGVHD.25 Of the patients, 24 had at least one feature that previously has been associated with reduced disease-specific survival, including 20 who had greater than 50% cutaneous involvement, 9 with progressive cGVHD, and 1 with thrombocytopenia (platelet count, < 100 000 μL). Of the patients, 17 had 1 of these features, 7 had 2 features, and 4 had none.26 There were 14 patients who had previously developed acute GVHD (aGVHD); 9 of these had steadily progressed to cGVHD, while 5 had completely responded to initial treatments for aGVHD, but then relapsed. Another 14 patients had de novo cGVHD. Histologically, 8 patients had findings consistent with lichenoid cGVHD, 6 patients had sclerodermoid features, and 12 patients had both lichenoid and sclerodermoid features. One patient with clinical follicular hyperkeratosis had an unusual histologic pattern of GVHD with a perifollicular infiltrate but no epidermal involvement. This patient had previously had esophageal and liver biopsies consistent with cGVHD. A further patient with biopsy-proven lung disease had no clinical involvement of the skin.
Of the patients, 25 completed 3 months of treatment, 21 completed 6 months of treatment, and 6 completed 12 months of treatment. Median duration of treatment was 6 months (range, 1-58 months).
Adverse events
During ECP treatment, 5 patients developed severe complications, including 4 patients who died. After 0.5, 3, and 20 months of treatment 3 patients died from advanced cGVHD; these deaths were not clearly treatment related. One patient died from renal failure and sepsis after 4 months of treatment, having developed acute invasive pulmonary aspergillosis. Another patient completed only one cycle of ECP, having developed acute dyspnea and hypoxia several hours after the first treatment. This patient subsequently developed acute respiratory distress syndrome (ARDS) and required transfer to an intensive care unit. Investigations failed to identify an infective cause and the patient recovered fully.
No other patients experienced adverse events that could be attributed to treatment. ECP was discontinued in 2 patients who had become unwell between treatments. One of these patients had pulmonary cGVHD and developed a pneumothorax in association with pleural effusions after receiving 3 cycles of treatment through peripheral cannulae. Another patient developed symptomatic exercise-induced ischemic heart disease that precluded further ECP.
Having improved and stabilized, 4 patients discontinued treatment after lengthy courses. Of the remaining patients, the reasons for discontinued treatment were as follows: deterioration of cGVHD (n = 2); poor response to treatment (n = 2); severe difficulties with venous access (n = 1); inconvenience of treatment schedules (n = 2); external funding difficulties (n = 1); and lung transplantation (n = 1). At present, 8 patients are continuing to receive treatment.
All patients were receiving systemic immunosuppressive medication throughout the ECP treatment course. During ECP treatment, systemic immunosuppression was stable in 15 patients, reduced in 9 patients, and increased in 4 patients.
Effect of ECP on clinical parameters
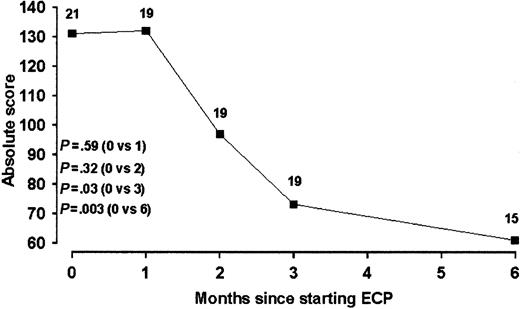
Skin disease. At the initiation of treatment, 25 patients had histologically confirmed cutaneous cGVHD. Of the remaining 3 patients, 1 had no clinical mucocutaneous disease and 2 had mucosal disease only. Evaluation of treatment response was possible in 21 patients who had baseline scores available and who completed at least 2 cycles of treatment. Baseline scores were unavailable in 2 patients. Clinical response was defined as a more than 25% reduction in initial skin score for patients who had stable or reduced levels of systemic immunosuppression. Any patient requiring increased immunosuppression was deemed to be a nonresponder, notwithstanding any clinical improvement in skin disease. Using these criteria, 38% (8/21 patients) were responders at 3 months and 48% (10/21 patients) were responders at 6 months. Median skin scores reduced during treatment and were as follows: 131, 132 (P = .59), 96 (P = .32), 71 (P = .03), and 61 (P = .003) at 0, 1, 2, 3 and 6 months, respectively (Figure 1). In contrast to a previous study, which reported an 80% complete remission rate of skin disease to ECP, complete remission of cutaneous cGVHD was seen only in a single subject in whom oral disease was persistent.17 No baseline clinical or laboratory parameters were predictive of responder status for cutaneous cGVHD.
Serial skin scores during ECP in patients with cutaneous cGVHD. Data are expressed as population medians. The number of patients at each time point is indicated.
Serial skin scores during ECP in patients with cutaneous cGVHD. Data are expressed as population medians. The number of patients at each time point is indicated.
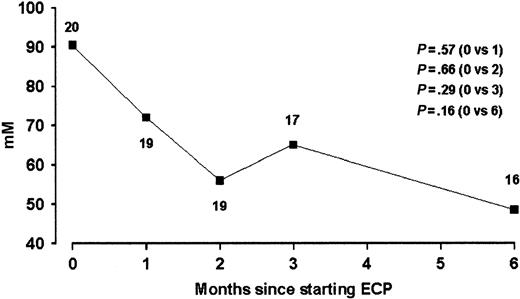
Hepatic disease. Of the patients, 25 (89%) had abnormalities of liver function and had been diagnosed with hepatic GVHD by the referring hematologic centers when other possible causes of abnormal liver function had been excluded. The majority of patients continued currently administered immunosuppressive therapies during the study so that the effect of ECP treatment on hepatic cGVHD could be assessed. During the study, any changes in medications known to be associated with hepatic dysfunction were recorded. At the start of ECP treatment 14 patients had abnormal alkaline phosphatase, 20 abnormal ALT, and 22 abnormal γGT. At the start of the study 3 patients had elevated bilirubin levels, although none was grossly elevated. After 3 months of ECP treatment, improvement of liver function by at least 25% occurred in 8 patients who had abnormal baseline values (Table 3). In 7 of these patients, immunosuppressive medications were stable and ECP was considered to be the cause of improvement. Using a definition of clinical response as reduction of an abnormal liver function enzyme value from baseline by at least 25%, then after 3 and 6 months clinical response was observed in, respectively, 3 and 1 patients with abnormal alkaline phosphatase (n = 14), 6 and 7 patients with abnormal ALT (n = 20), and 7 and 8 patients with abnormal GT (n = 22). No baseline clinical and laboratory parameters predicted responder status for hepatic cGVHD. Overall median baseline liver function test values and percentage changes in values at 3 and 6 months are illustrated for our population (Table 4). A trend of improvement of ALT was observed during ECP treatment (Figure 2), however, median population alkaline phosphatase and γGT values did not significantly change during treatment. Alteration in liver function did not significantly correlate with changes in skin scores in this population.
Alteration of immunosuppression in patients achieving at least 25%reduction in liver function test values after 3 months of ECP
Patient no. . | Liver function tests achieving 25% or greater reduction . | Immunosuppressive medications . | . | Change in immunosuppression . | |
|---|---|---|---|---|---|
| . | . | CyA . | MMF . | . | |
| 2 | γGT ALT | — | ↔ | Stable | |
| 3 | γGT ALT AP | ↔ | — | Stable | |
| 7 | γGT ALT AP B | — | ↑ | Increased | |
| 9 | γGT ALT | ↔ | — | Stable | |
| 10 | γGT ALT AP | ↔ | — | Stable | |
| 13 | γGT | ↔ | — | Stable | |
| 20 | γGT ALT | ↔ | — | Stable | |
| 27 | γGT ALT AP | — | — | Stable | |
Patient no. . | Liver function tests achieving 25% or greater reduction . | Immunosuppressive medications . | . | Change in immunosuppression . | |
|---|---|---|---|---|---|
| . | . | CyA . | MMF . | . | |
| 2 | γGT ALT | — | ↔ | Stable | |
| 3 | γGT ALT AP | ↔ | — | Stable | |
| 7 | γGT ALT AP B | — | ↑ | Increased | |
| 9 | γGT ALT | ↔ | — | Stable | |
| 10 | γGT ALT AP | ↔ | — | Stable | |
| 13 | γGT | ↔ | — | Stable | |
| 20 | γGT ALT | ↔ | — | Stable | |
| 27 | γGT ALT AP | — | — | Stable | |
CyA indicates cyclosporin; —, not administered; ↔, dosage stable; AP, alkaline phosphatase; B, total bilirubin; and ↑, dosage increased.
Changes in skin scores and abnormal liver function values during ECP
. | Baseline . | . | 3 mo . | . | 6 mo . | . | |||
|---|---|---|---|---|---|---|---|---|---|
. | n, abnormal at baseline . | Median (range) . | n . | % change of median (P) . | n . | % change of median (P) . | |||
| Skin score | 21 | 131 (10-349) | 19 | -46 (.03) | 15 | -53 (.003) | |||
| Alkaline phosphatase | 14 | 384 (141-1009) | 11 | -7 (.37) | 11 | +14 (1.0) | |||
| ALT | 20 | 91 (40-607) | 17 | -29 (.29) | 16 | -47 (.15) | |||
| γGT | 22 | 255 (5-2209) | 18 | +101 (.61) | 17 | +23 (.14) | |||
. | Baseline . | . | 3 mo . | . | 6 mo . | . | |||
|---|---|---|---|---|---|---|---|---|---|
. | n, abnormal at baseline . | Median (range) . | n . | % change of median (P) . | n . | % change of median (P) . | |||
| Skin score | 21 | 131 (10-349) | 19 | -46 (.03) | 15 | -53 (.003) | |||
| Alkaline phosphatase | 14 | 384 (141-1009) | 11 | -7 (.37) | 11 | +14 (1.0) | |||
| ALT | 20 | 91 (40-607) | 17 | -29 (.29) | 16 | -47 (.15) | |||
| γGT | 22 | 255 (5-2209) | 18 | +101 (.61) | 17 | +23 (.14) | |||
Serial serum alanine transaminase during ECP treatment in patientswith cGVHD and abnormal baseline ALT. Data are expressed as population medians. The number of patients at each time point is indicated.
Serial serum alanine transaminase during ECP treatment in patientswith cGVHD and abnormal baseline ALT. Data are expressed as population medians. The number of patients at each time point is indicated.
Pulmonary disease. During the study, 17 patients underwent pulmonary function measurement. Baseline spirometry was normal in 7 of these patients. Of the remainder, 7 patients had restrictive defects and 3 patients had mixed obstructive/restrictive defects. Repeat pulmonary function tests were performed in 8 patients with abnormal baseline spirometry 5 to 14 months after starting ECP. Median vital capacity reduced by 2% during the period of study. Vital capacity was stable in 4 patients (< 10% change), decreased in 3 patients (–17%, –16%, and –14%), and increased in one patient (+17%). At baseline 4 patients had reduced coefficients of gas transfer (< 80% predicted). On repeated measurement in one of these patients after 10 months, there was no significant change in gas transfer.
Mucosal disease. At the start of treatment, 14 patients had mucosal GVHD. Of these, 6 were classified as severe ulceration of which 3 improved during ECP. No patient with mucosal disease deteriorated during treatment.
Neuromuscular disease. One patient had been diagnosed with mixed sensorimotor polyneuropathy following electromyelographic studies at the start of treatment. There were no significant changes in symptoms or measured variables after 18 months of treatment.
Effect of ECP on hematologic parameters
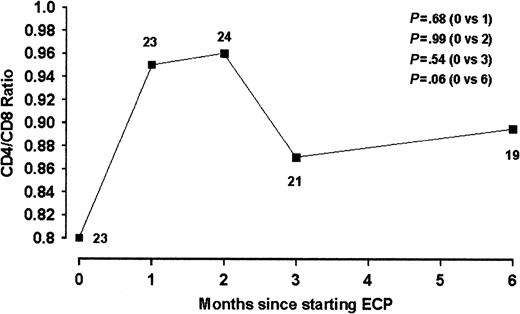
Statistically significant changes in platelet counts, overall white cell counts, and total hemoglobin levels were not observed in the studied population over the treatment period. Natural killer cells have been observed to rise by 2- to 10-fold in a previous study over 6 months of treatment.24 In our study, NK values did not change significantly and the median values actually decreased (Table 5). Similarly, normalization of inverted CD4/CD8 ratios has been observed previously in 3 patients undergoing ECP, largely relating to a reduction in CD8 counts. In our series, median CD4/CD8 ratios showed an upward trend after 6 months of treatment (Figure 3), with an increase in median CD4 and decrease in median CD8 counts. However, these changes were not statistically significant.
Changes in hematologic parameters during ECP
. | Baseline . | . | 3 mo . | . | 6 mo . | . | |||
|---|---|---|---|---|---|---|---|---|---|
. | n . | Median (range) . | n . | % change of median (P) . | n . | % change of median (P) . | |||
| NK cells | 18 | 0.14 (0.02-0.78) | 18 | -3 (.62) | 18 | -31 (.52) | |||
| Total lymphocytes | 23 | 0.89 (0.16-2.88) | 23 | +12 (.97) | 20 | +1 (.67) | |||
| CD4+ lymphocytes | 24 | 0.24 (0.03-0.77) | 22 | +21 (.34) | 19 | +19 (.72) | |||
| CD8+ lymphocytes | 24 | 0.28 (0.06-1.57) | 22 | -16 (.63) | 19 | -25 (.24) | |||
| CD4/CD8 ratio | 23 | 0.80 (0.11-4.83) | 21 | +9 (.54) | 19 | +11 (.06) | |||
. | Baseline . | . | 3 mo . | . | 6 mo . | . | |||
|---|---|---|---|---|---|---|---|---|---|
. | n . | Median (range) . | n . | % change of median (P) . | n . | % change of median (P) . | |||
| NK cells | 18 | 0.14 (0.02-0.78) | 18 | -3 (.62) | 18 | -31 (.52) | |||
| Total lymphocytes | 23 | 0.89 (0.16-2.88) | 23 | +12 (.97) | 20 | +1 (.67) | |||
| CD4+ lymphocytes | 24 | 0.24 (0.03-0.77) | 22 | +21 (.34) | 19 | +19 (.72) | |||
| CD8+ lymphocytes | 24 | 0.28 (0.06-1.57) | 22 | -16 (.63) | 19 | -25 (.24) | |||
| CD4/CD8 ratio | 23 | 0.80 (0.11-4.83) | 21 | +9 (.54) | 19 | +11 (.06) | |||
CD4/CD8 ratio during ECP treatment in patients with cGVHD. Data are expressed as population medians. The numbers of patients at each time point is indicated.
CD4/CD8 ratio during ECP treatment in patients with cGVHD. Data are expressed as population medians. The numbers of patients at each time point is indicated.
Discussion
We have presented data from 28 patients with advanced cGVHD treated with ECP. All patients had histologically confirmed disease and were refractory to conventional treatment. Systemic immunosuppressive medications were stable or reduced in the majority of patients during treatment. Cutaneous cGVHD was the most responsive to ECP, with a significant reduction in quantitative skin scores at 3 and 6 months. Median change from baseline was 46% at 3 months and 53% at 6 months, indicating that most of the improvement occurs in the first 3 months of therapy. Partial responses of hepatic cGVHD were seen in 7 patients after 3 months of treatment and 8 patients after 6 months of treatment. Mucosal cGVHD did not deteriorate in any patient during treatment and improved in 3 of 6 subjects with severe ulcerative disease. Pulmonary function and neuromuscular conduction did not respond to therapy.
Since the first report of the use of ECP in cGVHD in 1994, several nonrandomized small case series have been published, with the largest series comprising 18 patients.13-24 In contrast to previously reported studies, the majority of our patients had advanced chronic cGVHD. Of our patients, 27 were classified as having extensive cGVHD using the criteria of Shulman et al.25 A more recent prognostic model grading system identified 3 factors associated with reduced disease-specific survival in cGVHD.26 In our series, 24 patients had at least one of these factors at the start of the study, and 20 patients had cutaneous cGVHD affecting more than 50% of total surface area. This is a highly significant predictive factor of cGVHD-specific survival associated with a hazard ratio of 7.0. Our experience is consistent with previously reported findings, suggesting that mucocutaneous disease is most responsive to ECP treatment. Thus, our response rate at 6 months (10 of 21 patients) compares with a reported response rate in the literature of 57 of 73 cases with skin involvement (Table 6). In previously reported cases, response of skin disease was assessed subjectively, whereas more formal methods, with grading of disease severity and surface area involvement, were rarely used. In our series, complete resolution of cutaneous disease was unusual, occurring in only 1 of our 21 patients, although more than half of the subjects had responded by 6 months of treatment. In comparison, the largest studies to date report markedly different complete and partial response rates (Table 6). Differences in patient population might explain response variability between series, with a possible explanation for this variability being the delay from allograft to initiation of treatment: response rates are higher in series where median delay to treatment is fewer than 17 months compared with those longer than 34 months. However, analysis of our own data failed to identify a statistically significant correlation between delay to treatment and responder status. Differences in treatment methodology might also account for response variability. In the majority of series, treatment was given on 2 consecutive days every 2 to 3 weeks for the first 3 to 6 months, with frequency of administration then reduced depending on response. However, we used a similar treatment schedule with fortnightly cycles for 4 months and then monthly. It is not clear that outcomes improve with more frequent ECP administration protocols. The data available do not suggest that the route of psoralen administration markedly alters response.
ECP treatment of cGVHD: previously reported response rates
. | . | Time since transplantation medians, mo . | . | . | Clinical response . | . | . | ||
|---|---|---|---|---|---|---|---|---|---|
| Authors . | n . | . | Psoralen type . | Treatment frequency . | Skin . | Liver . | Lung . | ||
| Present study | 28 | 34 | Uvadex to Buffy coat | 2 × 2 weekly for 4 months then 2 × monthly | n = 21 | n = 25 | n = 9 | ||
| 1 CR | 0 CR | 0 CR | |||||||
| 9 PR | 8 PR | 0 PR | |||||||
| Smith et al16 | 18 | 36 | Oral 8-MOP | Variable from 2-3 × weekly to 2 × 3 weekly | n = 11 | n = 13 | n = 2 | ||
| Intervals increased if response | 1 CR | 2 CR | 0 CR | ||||||
| 2 PR | 1 PR | 0 PR | |||||||
| Greinix et al17 | 15 | 15 | Uvadex to Buffy coat | 2 × 3 weekly for 3 months then 2 × monthly | n = 15 | n = 9 | — | ||
| 12 CR | 7 CR | ||||||||
| 3 PR | 2 PR | ||||||||
| Salvaneschi et al20 | 14 (pd) | 17 | 8-MOP to Buffy coat | 2 × 2 weekly for 3 months then 2 × 3 weekly | n = 12 | n = 9 | — | ||
| 5 CR | 3 CR | ||||||||
| 5 PR | 3 PR | ||||||||
| Alcindor et al24 | 10 | 18 | Oral 8-MOP | 2 × 2 weekly | n = 10 | n = 3 | — | ||
| 7 PR | 2 PR | ||||||||
| Rossetti et al14 | 8 | 20 | Uvadex to Buffy coat | 2 × 3 weekly for first 6 months | n = 7 | n = 2 | n = 5 | ||
| Intervals increased if response | 2 CR | 1 PR | 1 CR | ||||||
| 2 PR | 1 PR | ||||||||
| Besnier et al21 | 5 | > 36 | 8-MOP to Buffy coat | 3 × weekly for 3 weeks | n = 2 | n = 2 | n = 1 | ||
| Intervals increased if response | 2 PR | 1 PR | 1 PR | ||||||
| Child et al19* | 6 | 7 | Uvadex to Buffy coat | 2 × 2 weekly for 4 months then 2 × monthly | n = 6 | n = 3 | n = 4 | ||
| 1 CR | 1 PR | 2 PR | |||||||
| 6 PR | |||||||||
| Dippel et al18 | 4 | 34 | Oral 8-MOP | 2 × 4 weekly | n = 4 | — | — | ||
| 1 CR | |||||||||
| 2 PR | |||||||||
| Dall' Amico et al15 | 4 (pd) | 16 | Uvadex to Buffy coat | 2 × 3 weekly for 6 months | n = 3 | n = 1 | n = 2 | ||
| Intervals increased if response | 1 CR | 1 PR | 1 CR | ||||||
| 2 PR | 1 PR | ||||||||
| Rossetti et al22 | 1 (pd) | > 9 | Uvadex to Buffy coat | 2 × 4 weekly for 15 months | n = 1 | — | n = 1 | ||
| 1 PR | 1 PR | ||||||||
| Konstantinow et al23 | 1 (pd) | 24 | 8-MOP to Buffy coat | 2 × 2 weekly for 4 months then 2 × 4-7 weekly | n = 1 | n = 1 | — | ||
| 1 CR | 1 CR | ||||||||
| Owsianowski et al13 | 1 | 60 | Oral 8-MOP | 2 × monthly | n = 1 | n = 1 | — | ||
| 1 PR | 1 PR | ||||||||
| Total (%) | 115; 95 adults, 20 pd | n = 94 | n = 69 | n = 24 | |||||
| 25 (27) CR | 13 (19) CR | 2 (8) CR | |||||||
| 42 (45) PR | 21 (30) PR | 6 (25) PR | |||||||
. | . | Time since transplantation medians, mo . | . | . | Clinical response . | . | . | ||
|---|---|---|---|---|---|---|---|---|---|
| Authors . | n . | . | Psoralen type . | Treatment frequency . | Skin . | Liver . | Lung . | ||
| Present study | 28 | 34 | Uvadex to Buffy coat | 2 × 2 weekly for 4 months then 2 × monthly | n = 21 | n = 25 | n = 9 | ||
| 1 CR | 0 CR | 0 CR | |||||||
| 9 PR | 8 PR | 0 PR | |||||||
| Smith et al16 | 18 | 36 | Oral 8-MOP | Variable from 2-3 × weekly to 2 × 3 weekly | n = 11 | n = 13 | n = 2 | ||
| Intervals increased if response | 1 CR | 2 CR | 0 CR | ||||||
| 2 PR | 1 PR | 0 PR | |||||||
| Greinix et al17 | 15 | 15 | Uvadex to Buffy coat | 2 × 3 weekly for 3 months then 2 × monthly | n = 15 | n = 9 | — | ||
| 12 CR | 7 CR | ||||||||
| 3 PR | 2 PR | ||||||||
| Salvaneschi et al20 | 14 (pd) | 17 | 8-MOP to Buffy coat | 2 × 2 weekly for 3 months then 2 × 3 weekly | n = 12 | n = 9 | — | ||
| 5 CR | 3 CR | ||||||||
| 5 PR | 3 PR | ||||||||
| Alcindor et al24 | 10 | 18 | Oral 8-MOP | 2 × 2 weekly | n = 10 | n = 3 | — | ||
| 7 PR | 2 PR | ||||||||
| Rossetti et al14 | 8 | 20 | Uvadex to Buffy coat | 2 × 3 weekly for first 6 months | n = 7 | n = 2 | n = 5 | ||
| Intervals increased if response | 2 CR | 1 PR | 1 CR | ||||||
| 2 PR | 1 PR | ||||||||
| Besnier et al21 | 5 | > 36 | 8-MOP to Buffy coat | 3 × weekly for 3 weeks | n = 2 | n = 2 | n = 1 | ||
| Intervals increased if response | 2 PR | 1 PR | 1 PR | ||||||
| Child et al19* | 6 | 7 | Uvadex to Buffy coat | 2 × 2 weekly for 4 months then 2 × monthly | n = 6 | n = 3 | n = 4 | ||
| 1 CR | 1 PR | 2 PR | |||||||
| 6 PR | |||||||||
| Dippel et al18 | 4 | 34 | Oral 8-MOP | 2 × 4 weekly | n = 4 | — | — | ||
| 1 CR | |||||||||
| 2 PR | |||||||||
| Dall' Amico et al15 | 4 (pd) | 16 | Uvadex to Buffy coat | 2 × 3 weekly for 6 months | n = 3 | n = 1 | n = 2 | ||
| Intervals increased if response | 1 CR | 1 PR | 1 CR | ||||||
| 2 PR | 1 PR | ||||||||
| Rossetti et al22 | 1 (pd) | > 9 | Uvadex to Buffy coat | 2 × 4 weekly for 15 months | n = 1 | — | n = 1 | ||
| 1 PR | 1 PR | ||||||||
| Konstantinow et al23 | 1 (pd) | 24 | 8-MOP to Buffy coat | 2 × 2 weekly for 4 months then 2 × 4-7 weekly | n = 1 | n = 1 | — | ||
| 1 CR | 1 CR | ||||||||
| Owsianowski et al13 | 1 | 60 | Oral 8-MOP | 2 × monthly | n = 1 | n = 1 | — | ||
| 1 PR | 1 PR | ||||||||
| Total (%) | 115; 95 adults, 20 pd | n = 94 | n = 69 | n = 24 | |||||
| 25 (27) CR | 13 (19) CR | 2 (8) CR | |||||||
| 42 (45) PR | 21 (30) PR | 6 (25) PR | |||||||
CR indicates complete response; PR, partial response; —, no patients reported; and pd, pediatric cases.
Excludes preliminary data from 5 patients to avoid duplication with present study.
Hepatic cGVHD improved in 24 of 44 patients in previous series including 13 complete responses and 13 partial responses. Our analysis of 4 variables in 25 patients with abnormal baseline liver function identified only partial responses of elevated ALT, GGT, and alkaline phosphatase in only 7, 8, and 1 patients, respectively, after 6 months of treatment. In no patient with abnormal liver function did ECP produce complete normalization of LFT values at any stage. Median values did not show statistically significant change during treatment, although a trend toward improvement of ALT was observed. These data suggest that in patients with long-standing cGVHD, only a relatively modest response of hepatic disease to ECP treatment can be expected.
Accurate quantitative assessment of the extent of active pulmonary cGVHD and its response to treatment is difficult using standard pulmonary function tests, because chronic changes from total body irradiation and advanced disease may be poorly reversible.29 In previous series, pulmonary function improved in 8 of 15 cases, but in our series only 1 patient of 9 showed a minor improvement during ECP.
Of the patients in our series, 2 developed severe complications during treatment, including 1 patient with sepsis who died from acute invasive pulmonary aspergillosis and 1 who developed ARDS. The etiology of ADRS is multifactorial, but is secondary to infection in approximately 40% of cases.30 To our knowledge, ARDS has not been reported previously in association with ECP, although rarely it may be provoked by blood product transfusions. Despite the negative cultures from this patient, underlying infection, rather than psoralen-related or hemodynamic effects of treatment, would seem the most likely etiology in this patient.
The mechanism of action of ECP in cGVHD is unclear. Apoptotic markers have been identified on lymphocytes immediately after ECP in patients with cutaneous T-cell lymphoma, GVHD, and scleroderma.31,32 Upon reinfusion, these cells are rapidly phagocytosed and may modulate the immune responses by release of cytokines.33,34 A recent study in 10 subjects with cGVHD suggested that effects of ECP include normalization of abnormal CD4/CD8 lymphocyte populations, increase in NK cell populations, and reduced CD80+ and CD123+ circulating dendritic cells.24 This group proposed 2 possible mechanisms of action of ECP in GVHD. First, a reduction in the number and function of dendritic cells might be expected to decrease their ability to present alloantigens and stimulate immune effector mechanisms. This may be important, as the initial target for CD8 lymphocytes in GVHD is thought to be proteins expressed by host antigen-presenting cells.35 Second, ECP may have a direct effect on populations of alloreactive T-cell populations such as CD8 lymphocytes. Adoptive transfer of NK cells appears to be associated with a GVHD preventative effect in animal models, an effect partly related to secretion of transforming growth factor beta.36,37 In our series of 28 subjects, we did not observe significant changes in NK cell populations. This suggests that previously observed increases in NK cell numbers following ECP are insufficient alone to explain the mechanism of action. A median overall trend of elevation in CD4/CD8 ratios was nevertheless observed, attributable to both a decrease in CD8 and an increase in CD4 populations. However, this effect did not clearly correlate with clinical response. It is likely that assessment of lymphocyte subsets is crude and may be missing a more selective effect of ECP on pathogenic T-cell clones that mediate cGVHD.
The rising incidence of cGVHD and poor response of many patients to conventional immunosuppressive treatments have led to the increasing use of ECP as a treatment for refractory disease. This series illustrates that ECP can produce clinical improvements in patients with advanced disease and features that are associated with an adverse prognosis. Nevertheless, ECP is a time-consuming and relatively expensive treatment that requires specialized equipment and staff expertise. Improved criteria for patient selection would be useful to improve direction of this treatment resource. Our analysis of pretreatment patient characteristics and laboratory parameters did not identify any variables that were predictive of a favorable response to treatment. However, comparison of our data with previous smaller series suggests that the initiation of ECP at an earlier stage is associated with more favorable response to treatment. In our study of patients with advanced cGVHD, ECP was initiated approximately 3 years after allogeneic transplantation and 2 years after onset of cGVHD. These data provide new evidence that ECP can be effective in extensive, long-standing cGVHD when treatment is initiated at an advanced stage after conventional immunosuppressive and corticosteroid therapy has failed. ECP should be considered most beneficial for patients with predominantly mucocutaneous cGVHD. However, in the absence of baseline criteria that accurately predict response, selection of these patients must continue to be made on clinical grounds.
Prepublished online as Blood First Edition Paper, April 24, 2003; DOI 10.1182/blood-2002-11-3351.
The publication costs of this article were defrayed in part by page charge payment. Therefore, and solely to indicate this fact, this article is hereby marked “advertisement” in accordance with 18 U.S.C. section 1734.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal