Abstract
Sickle cell disease (SCD) results in chronic hypoxia and secondarily increased erythropoietin concentrations. Leukocytosis and activated monocytes are also observed in SCD in absence of infection or vaso-occlusion (steady state), the reasons for which are unknown. We found that erythroid cells produced placenta growth factor (PlGF), an angiogenic growth factor belonging to the vascular endothelial growth factor (VEGF) family, and its expression was induced in bone marrow CD34+ progenitor cells in the presence of erythropoietin. Furthermore, the steady state circulating PlGF levels in subjects with severe SCD (at least 3 vaso-occlusive crises [VOCs] per year) were 18.5 ± 1.2 pg/mL (n = 9) compared with 15.5 ± 1.2 pg/mL (n = 13) in those with mild SCD (fewer than 3 VOCs per year) and 11.3 ± 0.7 pg/mL (n = 9) in healthy controls (P < .05), suggesting a correlation between PlGF levels and SCD severity. In addition, PlGF significantly increased mRNA levels of the proinflammatory cytochemokines interleukin-1β, interleukin-8, monocyte chemoattractant protein-1, and VEGF in peripheral blood mononuclear cells (MNCs) of healthy subjects (n = 4; P < .05). Expression of these same cytochemokines was significantly increased in MNCs from subjects with SCD at steady state (n = 14), compared with healthy controls. Of the leukocyte subfractions, PlGF stimulated monocyte chemotaxis (P < .05, n = 3). Taken together, these data show for the first time that erythroid cells intrinsically release a factor that can directly activate monocytes to increase inflammation. The baseline inflammation seen in SCD has always been attributed to sequelae secondary to the sickling phenomenon. We show that PlGF contributes to the inflammation observed in SCD and increases the incidence of vaso-occlusive events.
Introduction
Sickle cell disease (SCD) is characterized by sickling of red blood cells (RBCs) due to polymerization of sickle hemoglobin upon deoxygenation, resulting in vascular occlusion. Repeated sickling and oxygenation-deoxygenation damages RBC membranes and shortens their life span, causing hemolysis and anemia. Chronic hemolytic anemia, shortened life span of RBCs, and vascular occlusions are the hallmarks of the disease.1,2 The bone marrow responds to hypoxia and anemia with an erythropoietic response, reflected by high circulating reticulocytes.3,4
Another salient feature of SCD is leukocytosis that occurs despite absence of acute infection or inflammation. Activated monocytes,5-7 endothelial cells,5,7,8 neutrophils,9,10 and the coagulation cascade11,12 observed in SCD at steady state have been presumed to be secondary sequelae to the sickling phenomenon.2 While activated leukocytes13,14 have been shown to initiate and/or promote vaso-occlusion,15 the reasons for leukocytosis, activation of monocytes and neutrophils, and increased adhesivity of endothelium at steady state are largely unknown.
Activation of endothelial cells in SCD contributes to vascular occlusions.5,7,8,16 Vascular endothelial growth factor (VEGF), one of the most potent activators of endothelial cells that increases expression of intercellular adhesion molecule-1 (ICAM-1) on endothelial cells,8,17 is elevated in the plasma of patients with SCD. It therefore could increase adhesion of sickle RBCs to endothelial cells.
Placenta growth factor (PlGF) is another angiogenic growth factor belonging to the VEGF family.18-21 In contrast to widespread distribution of VEGF, PlGF production was initially considered to be restricted to placental trophoblasts and umbilical vein endothelial cells.21 However, recent studies show that PlGF is expressed in nonplacental tissues,22,23 including erythroid cells, but not by other mature hematopoietic cells.24
In addition, PlGF acts as a potent stimulant for VEGF secretion by monocytes25 and is coexpressed with VEGF in synovial fluid where high levels of PlGF homodimers and PlGF/VEGF heterodimers are found, suggesting that it plays a role in the inflammatory process.25 Because VEGF concentrations are elevated and erythropoiesis is expanded in individuals with SCD,26 we hypothesized that PlGF will be increased in the plasma of SCD patients and that PlGF may be the intrinsic red cell factor that mediates the activation of leukocytes in SCD, resulting in vaso-occlusive events.
In this report, we show that PlGF production was increased in the bone marrow from subjects with SCD reflected by an increase in PlGF plasma levels. Furthermore, PlGF activated peripheral blood mononuclear cells, specifically monocytes, and increased the expression of VEGF, proinflammatory cytokines, and chemokines by these cells. Finally, high PlGF concentrations correlated with increased incidence of vaso-occlusive events. Taken together, these data suggest that PlGF, an erythroid cell–derived factor, contributes to vascular occlusion and SCD severity.
Patients, materials, and methods
Subjects and samples
Heparinized blood was obtained from 22 subjects with SCD and 9 healthy controls according to a protocol approved by the Committee on Clinical Investigation (CCI, the institutional review board [IRB]) at Childrens Hospital Los Angeles, University of Southern California. Plasma was separated within 2 hours of sample collection. All subjects studied were under baseline conditions or at steady state, defined as the absence of vaso-occlusive or infectious episodes within the 3 weeks before or 3 weeks after phlebotomy. Subjects with SCD were patients followed at the Childrens Hospital Los Angeles and were diagnosed by hemoglobin electrophoresis. Controls were healthy adult volunteers who did not have SCD. Disease severity classification was based upon the Pediatric Hydroxyurea Group Multicenter Trial27 with minor changes: Subjects were classified as having severe disease if they had 3 or more vaso-occlusive episodes per year requiring inpatient admission or emergency room visits. Subjects were considered having mild disease if they had 2 or fewer such vaso-occlusive crises (VOCs) per year.
The blood samples obtained were centrifuged at 0°C to 4°C and 1000g for 15 minutes and plasma separated and stored at –80°C until it was assayed. Patients on chronic transfusion were excluded from the study. In 14 SCD subjects and 6 healthy controls, EDTA (ethylenediaminetetraacetic acid)–anticoagulated blood samples were collected to separate leukocytes and leukocyte fractions. Normal bone marrow samples were obtained from voluntary donations. Bone marrow samples from SCD subjects were obtained from individuals who had voluntarily donated 5 to 10 mL bone marrow when under anesthesia for another clinically indicated procedure. All blood and marrow donations were voluntary, obtained after informed consent, using protocols approved by the institutional IRB.
Separation of cell fractions from blood
Total leukocytes. Whole blood was subjected to hypotonic lysis by suspension in 3 volumes of sterile water for 30 seconds followed by rapid addition of one-tenth volume 10 × phosphate-buffered saline (PBS) to return the hemolysate isotonicity.28 The hemolysate was then centrifuged at 800g for 5 minutes to remove the red cell lysate. The pellet of white cells was washed once in PBS and resuspended in serum-free medium (X-vivo 15; BioWhittaker, Walkersville, MD) and used for studies on chemotaxis.
Peripheral blood mononuclear cells. Peripheral blood mononuclear cells (MNCs), composed mainly of lymphocytes and monocytes, were separated by layering freshly obtained peripheral blood on Ficoll-Hypaque (Pharmacia, Piscataway, NJ) and centrifugation at 1000g for 30 minutes at room temperature. The light density cells above the Ficoll were removed, washed once in PBS, and were either directly used for RNA analyses or resuspended in RPMI, with and without PlGF (250 ng/mL) for 1 hour before being used for RNA analysis.
Bone marrow CD34+cells. Bone marrow CD34+ cells were obtained as described previously.29,30 Briefly, fresh bone marrow samples were layered on Ficoll-Hypaque to obtain light density mononuclear cells (LD-MNCs), which were then enriched for CD34+ cells via magnetic-associated cell sorting (MACS), with 2 cycles of positive selection using anti-CD34 antibody and immunomagnetic beads and the Midi-MACS columns (Miltenyi Biotech, Auburn, CA). This typically results in a 90% to 95% pure population of CD34+ cells. CD34+ cells were placed in basal bone marrow medium,30 an Iscove modified Dulbecco medium (IMDM)–based medium containing 20% fetal bovine serum, in different cytokine combinations as described.
Erythroid (glycophorin A+) and nonerythroid (glycophorin A–) cells. Light density mononuclear cells, collected after density separation on Ficoll-Hypaque, were washed once with PBS, counted, and resuspended in Miltenyi buffer (1 × PBS, 0.5% bovine serum albumin [BSA], 5 mM EDTA; Miltenyi Biotech) at a concentration of 108 cells per 300 μL. To prevent nonspecific binding, human immunoglobulin G (IgG) (50 μg/100 μL; Baxter, Glendale, CA) was used as a blocking agent for 15 minutes at 6°C. Thereafter, cells were labeled with antiglycophorin A fluorescein isothiocyanate (FITC)–labeled antibody (Immunotech, Westbrook, ME), washed, and labeled with anti-FITC microbeads (Miltenyi Biotech) as per manufacturer's instructions. Cells were then passed through Midi-MACS columns and the eluted population kept aside as glycophorin A– cells. The glycophorin A+ fraction was passed through a second column to improve purity. Typically, a double-column sort results in more than 90% to 95% purity of the isolated fraction.31
RNA analyses
RNA was extracted from bone marrow cells or from peripheral blood MNCs using the RNA-Stat (Tel-Test, Friendswood, TX) RNA isolation kit using the manufacturer's instructions.
RT-PCR. RNA was quantified, and 1 μg total RNA per sample was reverse transcribed to cDNA in a 10 μL volume using the Superscript reverse transcriptase–polymerase chain reaction (RT-PCR) kit (PE Applied Biosystems, Foster City, CA). PCR was performed by amplifying 1 μL cDNA at 94°C for 1 minute, annealing for 1 minute, and 72°C for 1 minute for 40 cycles on a Perkin Elmer 9600 thermocycler (PE Applied Biosystems) using the following PlGF primer pairs: forward primer 5′-ACAAGCTTCCTACGTGGAGCTGACGTTCT-3′ and reverse primer 5′-AATCTAGATCCTTTCCGGCTTCATCTTCT-3′. PCR conditions were 55°C for 5 minutes and 95°C for 10 minutes for 1 cycle followed by 60°C for 1 minute and 95°C for 5 seconds for 40 cycles.
Ribonuclease (RNase) protection assay. RNase protection assays (RPAs) were performed on total RNA extracted from cells using the Riboquant In Vitro Transcription Kit (Pharmingen, San Diego, CA) as per the manufacturer's protocol. Briefly, 32P-labeled antisense RNA was synthesized in vitro using linearized templates for cytokines tumor necrosis factor-α (TNF-α) and interleukin-1β (IL-1β); chemokines macrophage inflammatory protein-1β (MIP-1β), monocyte chemoattractant protein-1 (MCP-1), and IL-8; the angiogenic growth factor VEGF; and housekeeping genes glyceraldehyde-3-phosphate dehydrogenase (GAPDH) and L32. Excess in vitro synthesized radiolabeled RNA was hybridized overnight to 2 μg of total cellular RNA. The mixture was digested with RNase, and protected fragments (double strands of cellular and in vitro–synthesized RNA) were resolved on a 6.5% polyacrylamide gel and quantified by phosphoimager analysis (Bio-Rad, Hercules, CA).
Western blot analyses
Cell pellets were lysed with 20 μL lysis buffer (50 mM Tris [tris(hydroxymethyl)aminomethane] HCl, 150 mM NaCl, 2 mM EDTA, 2 mM EGTA [ethylene glycol tetraacetic acid], 25 mM NaF, 25 mM β-glycerophosphate, 0.3% Nonidet P-40 [NP-40], 100 mM phenylmethylsulfonyl fluoride [PMSF], 1 μg/mL leupeptin, and 0.5 μg/mL aprotinin) per 2 million cells. Whole cell lysates were left on ice for 30 minutes and centrifuged at 4°C at 10 000g for 5 minutes. Lysate supernatants were then mixed with a 4 × NuPage sample buffer (Invitrogen, Carlsbad, CA) and boiled for 5 minutes at 90°C. The proteins were separated on a 10% polyacrylamide gel, transferred to nitrocellulose membrane, and blocked in 5% nonfat milk. The membranes were probed with monoclonal antihuman PlGF antibody (R&D Systems, Minneapolis, MN). The secondary antibody was a peroxidase-labeled antimouse antibody linked to an ECL-plus detection system (Amersham Biosciences, Piscataway, NJ). The membranes were probed with the secondary antibody and subjected to enhanced chemiluminescence (ECL), as per manufacturer's protocol, and then stripped and reprobed with antibody to human β-actin (Santa Cruz Biotechnology, Santa Cruz, CA) as a loading control.
Enzyme-linked immunosorbent assay (ELISA)
Blood was centrifuged at 4°C at 1000g for 15 minutes and the resulting plasma stored at –80°C until it was assayed. Plasma samples were assayed in duplicate for PlGF using a Quantikine human PlGF ELISA kit (R&D Systems), according to the manufacturer's directions. Optical density was measured at 450 nm and corrected for optical imperfections by subtracting the optical density at 550 nm in a Microplate Reader 3550-UV (Bio-Rad).
Electrophoretic mobility shift assays (EMSAs)
Nuclear extracts were prepared from cells treated with erythropoietin-containing medium at the time intervals indicated and from untreated control cells. Cells were lysed in 50 mM KCl, 25 mM HEPES (N-2-hydroxyethylpiperazine-N′-2-ethanesulfonic acid) (pH 8.0), 100 mM dithiothreitol (DTT), 1% NP-40, and protease inhibitor cocktail (Sigma) for 5 minutes on ice. The nuclei were washed, pelleted, and resuspended in extraction buffer (500 mM KCl, 25 mM HEPES [pH 8.0], 100 mM DTT, 10% glycerol, and protease inhibitor cocktail) at 4°C for 30 minutes with constant shaking, and the samples were spun at 13 000g for 5 minutes. Supernatant composed of nuclear extracts (5 μg protein per sample) were incubated in 25 μL 10 mM Tris-HCl (pH 7.6), 0.1 mM EDTA, 5 mM MgCl, 1 mM DTT, 10% glycerol, 40 mg/mL poly(dIdC), 50 mM KCl, and 32P-labeled oligonucleotide probe carrying metal transcription factor-1 (MTF-1) binding sites at room temperature for the indicated times. A 100-fold molar excess of unlabeled oligonucleotides was used as a competitor. The reaction mixtures were resolved on 4% acrylamide gel with 0.5% Tris-borate-EDTA (TBE) buffer. The sequence of the MTF-1 oligonucleotide probe used for the EMSA was 5′-CACGCACTGCGGGCTCCGGCGCTGCGGGCTGGCCGGGGCGCTGCGGGCTGACCG-3′.
Leukocyte chemotaxis assay
Leukocytes were obtained from whole blood using hypotonic lysis. Leukocyte migration assay was performed with minor modification of a previously described technique33 in a modified Boyden chemotactic apparatus (Costar Transwell Migration Plate; Corning, NY). Serum-free X-vivo 15 medium (BioWhittaker) containing 0, 1, or 5 nM concentrations of PlGF was placed in the lower chamber, and 50 000 leukocytes in 100 μL X-vivo 15 were placed in the upper chamber. Filters were removed, fixed with methanol, and stained with Diff-Quick (Baxter, Miami, FL). The number of cells that had transmigrated overnight were counted by an independent blinded observer and a differential count performed in 9 randomly chosen fields at × 100 magnification, and the counts were averaged. All experiments were performed in triplicate.
Statistical methods
For comparative analyses, P values were calculated using paired or unpaired t tests using the Excel 5.0 software (Microsoft, Seattle, WA). P values less than .05 were considered significant.
Results
PlGF is produced by erythroid cells and is expressed in bone marrow of patients with SCD
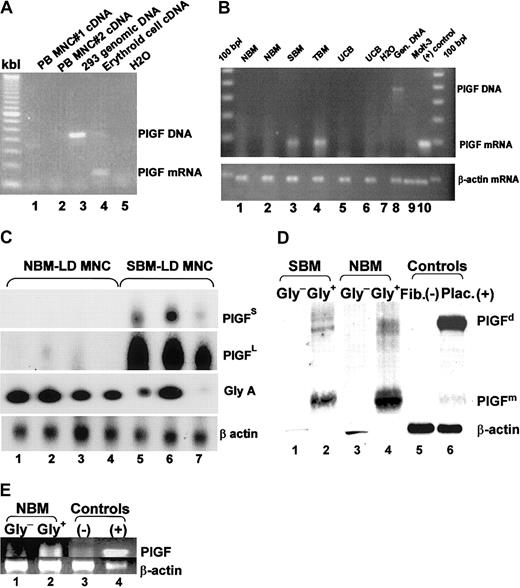
Normal bone marrow CD34+ progenitor cells were cultured in erythroid differentiation conditions for 2 weeks, as described previously,31 to obtain RNA from a relatively pure population of erythroblasts. Control nonerythroid cell RNA was extracted from peripheral blood MNCs (composed of lymphocytes and monocytes) from 2 healthy subjects. RT-PCR analyses were performed using PCR primers that spanned an intron to ensure that any contaminating genomic DNA would result in a different and larger-sized band. PlGF mRNA expression was only observed in the erythroid cultures (Figure 1A).
PlGF is produced by erythroid cells and is expressed in SCD bone marrow. (A) RNA was extracted from normal peripheral blood mononuclear cells (composed mainly of monocytes and lymphocytes, lanes 1 and 2) and an erythroid culture derived from normal bone marrow (lane 4) and subjected to RT-PCR analyses. PlGF primers used spanned 2 exons, giving a larger intron-containing band for genomic DNA (lane 3). (B) RT-PCR analysis on normal bone marrow cells (lanes 1 and 2), SCD bone marrow (SBM, lane 3), β-thalassemia major bone marrow (TBM, lane 4), and 2 normal umbilical cord blood mononuclear cells (UCB, lanes 5 and 6) using PlGF (top panel) and β-actin (bottom panel) primers. Lanes 7-10 are control lanes: H20 (lane 7) and the Molt T-cell line (lane 8) as negative controls, 293 cell genomic DNA as a DNA control (lane 9), and endothelial cells (lane 10) as a positive control. (C) RT-PCR analyses for PlGF, glycophorin A (an erythroid cell–specific gene), and β-actin followed by transfer and probing, showing the proportion of PlGF transcription in normal versus SCD light density mononuclear cells. The short (PlGFS) and long (PlGFL) exposure of the PlGF blot is shown to depict the small amount of PlGF mRNA in normal bone marrow and relatively high amount in sickle bone marrow. (D) Western blot analyses of glycophorin A+ (Gly+) and glycophorin A– (Gly–) light density mononuclear cells (LD-MNCs). Lanes 1 and 2 represent Gly+ and Gly– cells from SCD LD-MNCs (SBM), respectively; lanes 3 and 4 represent Gly+ and Gly– cells from normal bone marrow LD-MNCs (NBM), respectively; lanes 5 and 6 represent fibroblasts and placenta, as negative and positive controls, respectively. (E) RT-PCR analysis for PlGF on the NBM Gly+ and Gly– cells, shown in panel D (lanes 1 and 2). Lanes 3 and 4 represent controls.
PlGF is produced by erythroid cells and is expressed in SCD bone marrow. (A) RNA was extracted from normal peripheral blood mononuclear cells (composed mainly of monocytes and lymphocytes, lanes 1 and 2) and an erythroid culture derived from normal bone marrow (lane 4) and subjected to RT-PCR analyses. PlGF primers used spanned 2 exons, giving a larger intron-containing band for genomic DNA (lane 3). (B) RT-PCR analysis on normal bone marrow cells (lanes 1 and 2), SCD bone marrow (SBM, lane 3), β-thalassemia major bone marrow (TBM, lane 4), and 2 normal umbilical cord blood mononuclear cells (UCB, lanes 5 and 6) using PlGF (top panel) and β-actin (bottom panel) primers. Lanes 7-10 are control lanes: H20 (lane 7) and the Molt T-cell line (lane 8) as negative controls, 293 cell genomic DNA as a DNA control (lane 9), and endothelial cells (lane 10) as a positive control. (C) RT-PCR analyses for PlGF, glycophorin A (an erythroid cell–specific gene), and β-actin followed by transfer and probing, showing the proportion of PlGF transcription in normal versus SCD light density mononuclear cells. The short (PlGFS) and long (PlGFL) exposure of the PlGF blot is shown to depict the small amount of PlGF mRNA in normal bone marrow and relatively high amount in sickle bone marrow. (D) Western blot analyses of glycophorin A+ (Gly+) and glycophorin A– (Gly–) light density mononuclear cells (LD-MNCs). Lanes 1 and 2 represent Gly+ and Gly– cells from SCD LD-MNCs (SBM), respectively; lanes 3 and 4 represent Gly+ and Gly– cells from normal bone marrow LD-MNCs (NBM), respectively; lanes 5 and 6 represent fibroblasts and placenta, as negative and positive controls, respectively. (E) RT-PCR analysis for PlGF on the NBM Gly+ and Gly– cells, shown in panel D (lanes 1 and 2). Lanes 3 and 4 represent controls.
The chronic anemia in SCD results in erythroid cell hyperplasia in the bone marrow. Because we found expression of PlGF in erythroid cells, it was therefore likely that PlGF production would be increased in SCD. We isolated RNA from normal and SCD bone marrow light density mononuclear cells. Bone marrow samples from subjects with β-thalassemia, another hemoglobinopathy with increased erythropoiesis, were also included. An RT-PCR analysis showed that PlGF transcripts were present in the thalassemia and SCD bone marrow light density mononuclear cells (LD-MNCs), when visualized with ethidium bromide staining of the agarose gels, but not in normal bone marrow LD-MNCs (Figure 1B). This finding was intriguing because we detected PlGF transcripts in the erythroid cultures and a significant pool of erythroid cells is present in normal bone marrow. One possible explanation is that the level of PlGF expression in normal bone marrow was below the detection limit of ethidium bromide–visualized RT-PCR product but was detectable in SCD and thalassemia bone marrow due to the increased erythroid cell mass. Conversely, there could be factors in SCD and thalassemia subjects that could enhance production of PlGF.
Therefore, we improved the sensitivity of the assay and controlled for the number of erythroid cells by performing an RT-PCR for glycophorin A: Light density mononuclear cells were isolated from 4 normal and 3 SCD bone marrow cells, and a semiquantitative RT-PCR analysis was performed for PlGF, glycophorin A, and β-actin. The blot was transferred and probed with a corresponding 32P-labeled cDNA probes. The main observations were as follows: (1) There were fewer glycophorin A–expressing cells in the light density fraction in SCD bone marrow, suggesting that sickle erythroid cells are denser than normal. (2) Short exposure of the blot allowed the detection of PlGF transcripts only in SCD light density mononuclear fraction (Figure 1C, PlGFS), while the prolonged exposure of the same blot (Figure 1C, PlGFL) showed small amounts of PlGF mRNA detectable in normal bone marrow as well. (3) Most importantly, the PlGF mRNA expression, when normalized for glycophorin A expression, was very high in SCD bone marrow. Taken together, these data suggest that erythroid cells in SCD express a disproportionately high concentration of PlGF.
It was intriguing, however, that despite a high glycophorin A signal, there was a very faint band for PlGF in normal bone marrow. We reasoned that (1) the RT-PCR was performed on relatively impure erythroid populations, because normal light density mononuclear cells contain few erythroblasts and are predominantly composed of lymphocytes and monocytes, and (2) the glycophorin A RT-PCR was only semiquantitative. We therefore purified erythroid and nonerythroid cells from light density fraction of normal and SCD bone marrow cells using human glycophorin A–FITC–labeled antibody followed by an immunomagnetic selection with anti-FITC microbeads and performed Western blot analyses on equal numbers of glycophorin A+ and glycophorin A– cells from each of the bone marrow samples (Figure 1D). Due to limited volume of SCD bone marrow, comparison of relative PlGF concentrations between normal and SCD glycophorin A+ cells were not possible. However, PlGF monomers and dimers were detected only in the glycophorin A+ erythroid cell populations. Of note, PlGF was predominantly present in monomeric form in hematopoietic cells, while the converse was seen in placental tissue. We are currently investigating this aspect. Fibroblast lysates were used as a negative control and show a faint PlGF band. Recently, PlGF expression has been reported to be inducible in fibroblasts.23
A portion of the glycophorin A+ and glycophorin A– cells from the normal bone marrow were also subjected to RT-PCR analyses (Figure 1E). Due to limited cell numbers, cells from SCD bone marrow were only subjected to Western blot analyses (Figure 1D). Figure 1E reaffirms that PlGF transcripts are predominant in the glycophorin A+ erythroid cells.
PlGF expression is inducible by erythropoietin in bone marrow progenitor cells
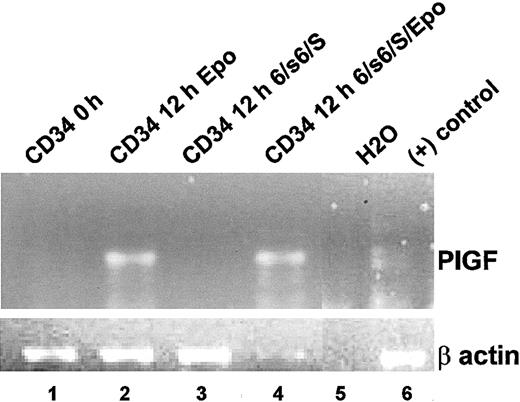
The anemia in SCD (and β-thalassemia) causes tissue hypoxia that results in increased erythropoietin levels.33 CD34+ progenitor cells from a normal bone marrow were analyzed for PlGF mRNA by RT-PCR analyses soon after isolation (Figure 2, lane 1) or after a 12-hour exposure to 3 U/mL erythropoietin (Figure 2, lane 2). We also wanted to determine if PlGF production in erythroid cells was a result of erythropoietin or was generated by erythroid cells in an erythropoietin-independent manner. Erythropoietin is essential for erythroid differentiation. We therefore used an erythropoietin-independent erythroid differentiation protocol previously shown to result in erythroid commitment and differentiation.35 CD34+ progenitor cells were cultured in an erythropoietin-independent erythroid medium (IL-6, soluble IL-6 [sIL-6] receptor, and stem cell factor34 [SCF]; Figure 2, lane 3) or in erythropoietin-independent erythroid medium with additional erythropoietin (3 U/mL; Figure 2, lane 4). No PlGF expression was observed in erythropoietin-independent erythroid differentiation conditions (Figure 2, lane 3). However, with addition of erythropoietin to these conditions (Figure 2, lane 4), PlGF transcripts became evident, demonstrating that erythropoietin induced transcription of PlGF.
Erythropoietin induces PlGF expression in bone marrow progenitor cells. CD34+ progenitor cells from a normal bone marrow were analyzed for PlGF mRNA by RT-PCR analyses soon after isolation (lane 1) or after a 12-hour exposure either to 3 U/mL erythropoietin (lane 2) or to erythropoietin-independent erythroid differentiation medium (IL-6, sIL-6 receptor, and SCF; lane 3) or erythropoietin-independent erythroid differentiation medium with additional erythropoietin (3 U/mL; lane 4). The bottom panel shows an RT-PCR for β-actin for the corresponding samples. Lanes 5 and 6 consist of negative (H2O) and positive controls (human pulmonary microvascular endothelial cells), respectively.
Erythropoietin induces PlGF expression in bone marrow progenitor cells. CD34+ progenitor cells from a normal bone marrow were analyzed for PlGF mRNA by RT-PCR analyses soon after isolation (lane 1) or after a 12-hour exposure either to 3 U/mL erythropoietin (lane 2) or to erythropoietin-independent erythroid differentiation medium (IL-6, sIL-6 receptor, and SCF; lane 3) or erythropoietin-independent erythroid differentiation medium with additional erythropoietin (3 U/mL; lane 4). The bottom panel shows an RT-PCR for β-actin for the corresponding samples. Lanes 5 and 6 consist of negative (H2O) and positive controls (human pulmonary microvascular endothelial cells), respectively.
Erythropoietin increases the nuclear DNA binding activity of metal response element-binding transcription factor-1
The VEGF promoter contains several hypoxia inducible factor (HIF-1α) response elements.35 In contrast, the PlGF promoter does not have HIF-1 response elements but has 9 consensus binding motifs for MTF-1.24 We therefore examined whether erythropoietin induced nuclear translocation and DNA binding of MTF-1. Due to limitations in the number of CD34+ progenitor cells that can be obtained from normal bone marrow, we utilized a Ewing sarcoma cell line (SK-N-MC cells). SK-N-MC cells express both PlGF and erythropoietin receptor, and addition of erythropoietin to this cell line results in a dose-dependent increase in PlGF mRNA expression (P.M. et al, unpublished results, June 2003). SK-N-MC cells were treated with erythropoietin for different time intervals followed by isolation of nuclear extracts and hybridization to a radiolabeled oligonucleotide containing MTF-1 response elements (Figure 3). Nuclear DNA binding activity of MTF-1 was observed within 15 minutes of erythropoietin exposure and was competed out by addition of excess nonradiolabeled probe. These studies indicate that erythropoietin induces nuclear translocation and DNA binding activity of MTF-1, suggesting that the increased transcription of PlGF by erythropoietin may involve MTF-1 binding to its response elements in the PlGF gene promoter.
Erythropoietin increases nuclear DNA binding activity of MTF-1 transcription factor. Electrophoretic mobility shift assay (EMSA) on nuclear extracts from the SK-N-MC Ewing sarcoma cell line, which expresses both PlGF and a functional erythropoietin receptor, using a radiolabeled oligonucleotide probe containing MTF-1 response elements. The blot shows increased nuclear DNA binding of MTF-1 to the probe within 15 minutes (lane 3), with a maximal binding at 30 minutes (lane 4). The response is abrogated with excess cold (nonradiolabeled) oligonucleotide MTF-1 probe (lane 5).
Erythropoietin increases nuclear DNA binding activity of MTF-1 transcription factor. Electrophoretic mobility shift assay (EMSA) on nuclear extracts from the SK-N-MC Ewing sarcoma cell line, which expresses both PlGF and a functional erythropoietin receptor, using a radiolabeled oligonucleotide probe containing MTF-1 response elements. The blot shows increased nuclear DNA binding of MTF-1 to the probe within 15 minutes (lane 3), with a maximal binding at 30 minutes (lane 4). The response is abrogated with excess cold (nonradiolabeled) oligonucleotide MTF-1 probe (lane 5).
Plasma PlGF concentrations are elevated in subjects with SCD and are associated with increased incidence of vascular occlusive events
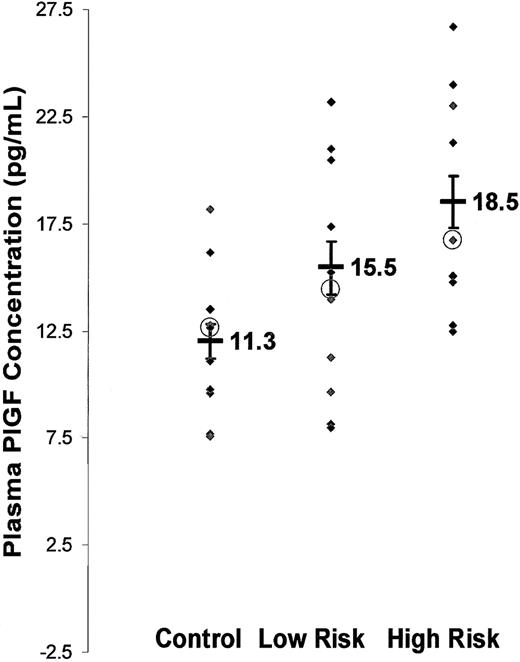
PlGF is a secreted angiogenic growth factor,24 and we observed increased PlGF mRNA in bone marrow of subjects with SCD. We therefore determined whether this resulted in higher amounts of PlGF protein in peripheral blood in subjects with SCD. PlGF concentrations in SCD plasma were 16.8 ± 0.9 pg/mL (mean ± SEM), n = 22, compared with 11.3 ± 0.7 pg/mL, n = 9, in healthy controls (P < .001). Furthermore, PlGF concentrations increased with increasing disease severity (Figure 4): Of the 22 individuals with SCD, those with severe disease (n = 9) had significantly higher circulating PlGF at steady state (18.5 ± 1.2 pg/mL) than those with mild disease (15.5 ± 1.2 pg/mL, n = 13) compared with healthy controls (11.3 ± 0.7 pg/mL, n = 9). Each of these groups was significantly different from each other (P < .05). Therefore, PlGF might modulate the clinical severity, possibly by increasing the inflammatory tone in SCD.
PlGF concentration is increased in plasma of subjects with SCD at steady state and correlates with incidence of VOCs. PlGF concentration was determined on cell-free heparinized plasma from SCD subjects and from healthy controls using ELISA (y-axis). Subjects were classified as mild and severe disease based on the incidence of severe VOCs per year (x-axis). Each point (♦) represents average values of duplicate samples from individual subjects. Mean values are represented as thick black bars, with values adjacent to the symbol. Medians represented as ○. Error bars represent SEM.
PlGF concentration is increased in plasma of subjects with SCD at steady state and correlates with incidence of VOCs. PlGF concentration was determined on cell-free heparinized plasma from SCD subjects and from healthy controls using ELISA (y-axis). Subjects were classified as mild and severe disease based on the incidence of severe VOCs per year (x-axis). Each point (♦) represents average values of duplicate samples from individual subjects. Mean values are represented as thick black bars, with values adjacent to the symbol. Medians represented as ○. Error bars represent SEM.
PlGF activates mononuclear cells and increases production of inflammatory cytokines and chemokines
Elevated levels of PlGF and PlGF/VEGF heterodimers have been observed in inflammatory joint disease.25 Activated monocytes and high concentrations of inflammatory cytokines have been reported in SCD in the absence of vascular occlusion or illness. We therefore determined whether PlGF could mediate activation of leukocytes and increase production of inflammatory molecules.
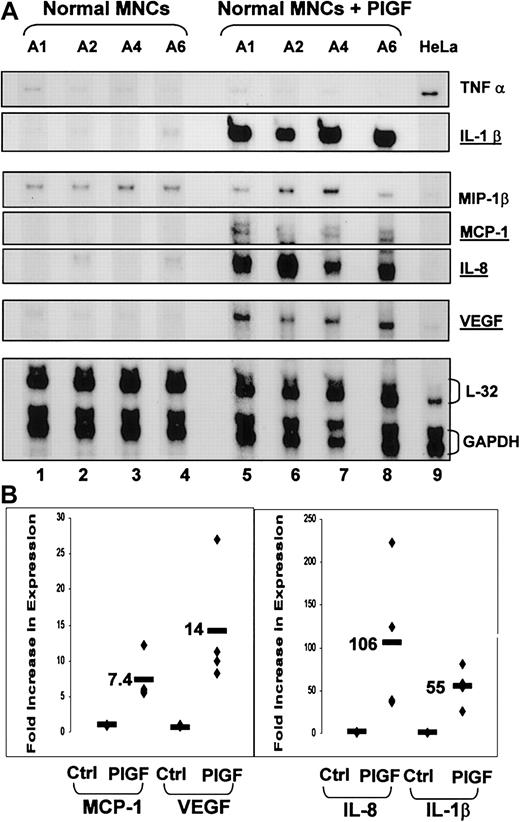
Normal blood MNCs (n = 4) were exposed to PlGF (250 ng/mL), and the mRNA expression profile of inflammatory cytokines TNF-α, IL-1β; chemokines IL-8, MIP1-β, and MCP-1; and VEGF was examined (Figure 5). There was increased mRNA expression of IL-1β (55-fold ± 11-fold, P < .08), IL-8 (106-fold ±44-fold, P < .04), MCP-1 (7.4-fold ± 1.6-fold, P < .01), and VEGF (14-fold ± 4.3-fold, P < .02) as compared with untreated MNCs.
PlGF activates normal mononuclear cells, increasing production of inflammatory cytokine IL-1β, chemokines MCP-1 and IL-8, and the angiogenic growth factor VEGF. (A) Ribonuclease protection assay performed using RNA from normal peripheral blood MNCs from 4 subjects exposed to exogenous PlGF (250 ng/mL) for 1 hour (lanes 5-8) or left untreated (lanes 1-4). HeLa cell RNA was used as a control RNA (lane 9). (B) Densitometry analyses were performed on appropriate exposures on the different bands and normalized for loading.
PlGF activates normal mononuclear cells, increasing production of inflammatory cytokine IL-1β, chemokines MCP-1 and IL-8, and the angiogenic growth factor VEGF. (A) Ribonuclease protection assay performed using RNA from normal peripheral blood MNCs from 4 subjects exposed to exogenous PlGF (250 ng/mL) for 1 hour (lanes 5-8) or left untreated (lanes 1-4). HeLa cell RNA was used as a control RNA (lane 9). (B) Densitometry analyses were performed on appropriate exposures on the different bands and normalized for loading.
The genes inducible by PlGF in normal MNCs were also expressed at high levels in MNCs from SCD subjects at steady state
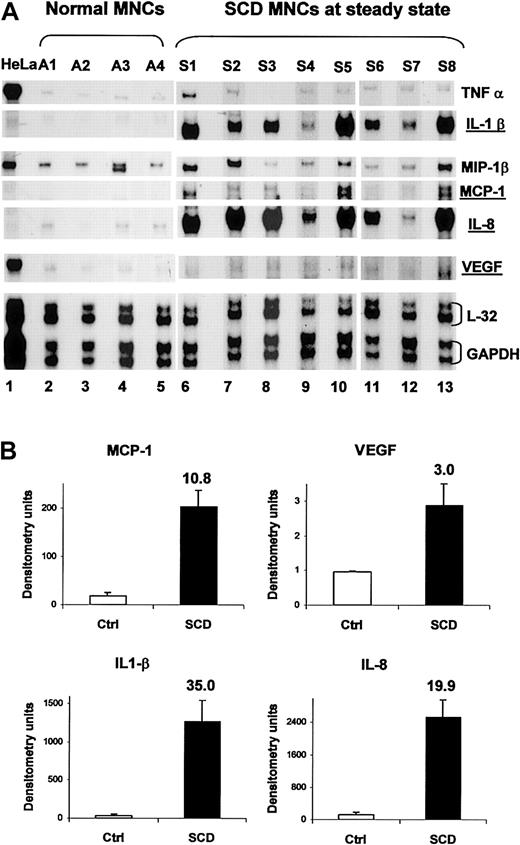
We reasoned that if PlGF were activating normal MNCs, expression of these cytokines and chemokines would also be elevated in SCD MNCs. RNA from peripheral blood MNCs from subjects with SCD at steady state was analyzed. The same pattern of increased mRNA expression of IL-1β, IL-8, MCP-1, and VEGF was observed in MNCs from SCD subjects, compared with controls (Figure 6), with P values ranging from P < .01 to P < .001. These data suggest that the activation of monocytes previously reported in SCD5 may be secondary to increased release of PlGF from erythroid cells.
The cytokines and chemokines that are induced in normal MNCs with exogenous PlGF are also elevated in SCD MNCs at baseline. (A) RNase protection assay was performed using RNA from normal peripheral blood MNCs (A1-A4, lanes 2-5) and those derived from subjects with SCD at steady state (S1-S8, lanes 6-13). HeLa cell RNA was used as a control RNA. (B) Mean densitometry of MCP-1, VEGF, IL1-β, and IL-8 shown in panel A in normal controls (□) and in SCD patients (▪). Error bars represent SEM.
The cytokines and chemokines that are induced in normal MNCs with exogenous PlGF are also elevated in SCD MNCs at baseline. (A) RNase protection assay was performed using RNA from normal peripheral blood MNCs (A1-A4, lanes 2-5) and those derived from subjects with SCD at steady state (S1-S8, lanes 6-13). HeLa cell RNA was used as a control RNA. (B) Mean densitometry of MCP-1, VEGF, IL1-β, and IL-8 shown in panel A in normal controls (□) and in SCD patients (▪). Error bars represent SEM.
PlGF increased chemotaxis of monocytes
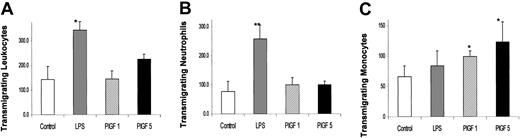
Of the mononuclear fractions, Flt-1, the receptor for PlGF, is expressed on monocyte-macrophages.36 Monocytes have been shown to transmigrate toward a PlGF gradient.37,38 To examine which of the leukocyte subfractions (monocytes or neutrophils) were responding to PlGF, we exposed leukocytes, obtained after hypotonic lysis of whole blood, to a PlGF gradient and measured their transmigration toward PlGF through a 3 μm pore membrane. Figure 7 shows the number of transmigrated leukocytes and their subpopulations (granulocytes and monocytes). PlGF increased the transmigration of leukocytes. However, it was the monocyte fraction that showed a statistically significant, dose-dependent increase in chemotaxis toward PlGF (n = 3), substantiating that PlGF is affecting the monocyte fraction of the mononuclear cells.
PlGF increases monocyte chemotaxis. Total leukocytes from fresh peripheral blood obtained after hypotonic lysis of RBCs were placed in a Boyden chamber against no chemical gradient (white bars, control) or a PlGF gradient of 1 nM (striped bars) or 5 nM (black bars). Lipopolysaccharide (LPS, gray bars) gradient was the positive control, which promotes neutrophil transmigration. Cells that transmigrated across 3 μm pore filters were stained and the total leukocytes counted in 9 fields (A) and a differential count obtained on the transmigrating neutrophils (B) or monocytes (C). Each bar represents mean ± SD (n = 3), and values that were statistically significant from controls are represented with an asterisk: *P < .05 and **P < .01.
PlGF increases monocyte chemotaxis. Total leukocytes from fresh peripheral blood obtained after hypotonic lysis of RBCs were placed in a Boyden chamber against no chemical gradient (white bars, control) or a PlGF gradient of 1 nM (striped bars) or 5 nM (black bars). Lipopolysaccharide (LPS, gray bars) gradient was the positive control, which promotes neutrophil transmigration. Cells that transmigrated across 3 μm pore filters were stained and the total leukocytes counted in 9 fields (A) and a differential count obtained on the transmigrating neutrophils (B) or monocytes (C). Each bar represents mean ± SD (n = 3), and values that were statistically significant from controls are represented with an asterisk: *P < .05 and **P < .01.
Discussion
Although the molecular defect in SCD has been known for years, the reasons for leukocytosis, monocyte and endothelial cell activation at steady state, in the absence of infection or vascular occlusion, are largely unknown. Moreover, the disease phenotype is extremely variable, and prolonged symptom-free episodes between VOCs suggest factors other than sickling promote VOCs. We show that erythroid cells expressed PlGF in response to erythropoietin. PlGF increased the expression of proinflammatory cytochemokines (IL-1β, IL-8, and MCP-1) and VEGF from normal mononuclear cells. This same pattern of increased mRNA expression of cytokines and chemokines was also seen in mononuclear cells from SCD subjects at steady state. We also found that plasma PlGF concentrations were elevated in individuals with SCD at steady state and correlated with the incidence of vaso-occlusive events.
Recently, both PlGF and VEGF-A have been shown to be secreted from erythroid cells in culture, indicating the specific role of erythroid cells in generation of angiogenic factors.24,39 Both VEGF and PlGF have been proposed to interact with either monocyte/macrophages or endothelial cells to promote transmigration of erythroid cells across endothelial barrier. The expanded erythropoiesis in SCD bone marrow may be responsible for increased PlGF observed in SCD bone marrow and in circulation. However, it was interesting that the light density fraction of bone marrow mononuclear cells from healthy or SCD subjects appeared to have nearly comparable amounts of erythroid cells, as determined by expression of glycophorin A, an erythroid-specific cytoskeletal protein. The light density cell separation technique most likely removed the relatively dense erythroid cells observed in SCD secondary to the sickling phenomenon. The density profile of red cells is altered in SCD.40,41 Regardless, the levels of PlGF mRNA were disproportionately high in SCD bone marrow when normalized to glycophorin mRNA, suggesting that the expression of PlGF in SCD erythroid cells is regulated by additional factors.
It was intriguing, however, that despite high glycophorin A signal, there was a very faint band for PlGF in normal bone marrow. Tordjman et al have shown that normal erythroid cells produce significant amounts of PlGF.24 When relatively purified erythroid and nonerythroid populations from the light density mononuclear fractions from normal and SCD bone marrow were analyzed, PlGF protein was present only in erythroid cells. These results are consistent with the previously published results.24
We also found that PlGF transcripts were induced as early as 12 hours after erythropoietin exposure in CD34+ cells. However, Tordjman et al24 have previously reported the appearance of PlGF transcripts in CD34+ cells on the second day of culture. This discrepancy may be due to differences in methodology. In our study we used more cycles of cDNA amplification by PCR, increasing the sensitivity of the assay, and used different erythroid culture conditions.
Anemia causes secondarily increased erythropoietin levels and erythroid hyperplasia, resulting in increased erythropoietin concentrations.33 We observed that PlGF expression was induced by erythropoietin. This response was associated with increased nuclear DNA binding activity of the MTF-1, suggesting that erythropoietin probably increases PlGF transcription via this mechanism.23 PlGF induction has been shown to be MTF-1 mediated in embryonic fibroblasts.23 However, its induction by erythropoietin has not been previously described. We observed more PlGF transcripts in bone marrow from patients with β-thalassemia major, another hemolytic anemia with increased erythropoietic stress. It would be of interest to determine if PlGF is increased in other hemolytic anemias or severe iron deficiency states that result in erythroid hyperplasia. Because erythropoietin increases release of an angiogenic growth factor, higher PlGF levels may be present in hemoglobin SC patients, which may contribute to the retinopathy seen commonly in hemoglobin SC disease. The increased production of angiogenic growth factors mediated by erythropoietin could have even wider implications: Erythropoietin, routinely given to alleviate cancer-therapy–related anemia, could induce angiogenesis in tumors. In a separate study, we have found that pediatric tumor cells express erythropoietin receptor and addition of erythropoietin increases the release of angiogenic growth factors VEGF and PlGF from tumor cells (P.M. et al, unpublished results, June 2003). Administration of erythropoietin to patients with cancer therefore may promote tumor angiogenesis and survival, which needs to be studied. PlGF does play a role in placental angiogenesis: PlGF concentrations have been shown to increase during gestation, and lower PlGF concentrations are associated with pre-eclampsia of pregnancy.42,43
PlGF concentrations in SCD subjects correlated with incidence of vaso-occlusive events, suggesting its putative role in inflammation. One observes high levels of PlGF homodimers and PlGF/VEGF heterodimers in synovial fluid of patients with inflammatory joint disease.25 The mechanism by which PlGF or PlGF/VEGF heterodimers modulate inflammation is unknown, and whether this is due to signaling via different VEGF receptors has largely been undetermined. Studies have shown that VEGF can bind 2 distinct kinase receptors: the fms-like tyrosine kinase, Flt-1 (VEGF receptor-1), and the kinase insert domain-containing receptor/fetal liver kinase, KDR/Flk-1 (VEGF receptor-2).38,44-46 PlGF binds only to Flt-1, while VEGF can bind either Flt-1 or KDR/Flk-1 receptor on endothelial cells.20,47-49 Among the hematopoietic cells, Flt-1 is specifically present on monocytes25,37 or progenitor cells.50 It has also been shown that PlGF can induce VEGF release from monocytes.25 Its role in generating proinflammatory cytokines and chemokines from monocytes has not been described. Our studies show that PlGF causes a very significant increase in proinflammatory cytochemokine mRNA in monocytes. This effect was mediated via the Flt-1 receptor (see accompanying article by Selvaraj et al,51 beginning on page 1515).
These proinflammatory molecules could contribute to the activation of leukocytes and endothelial cells, a phenomenon observed in SCD at steady state.6,7,26 Studies in the literature show that these proinflammatory molecules (TNF-α, IL-1β, IL-8) are increased in subjects with SCD52 and activate neutrophils in vitro.53 Although TNF-α concentrations appeared unchanged under our experimental conditions, there was a transient increase in TNF-α expression with PlGF in peripheral blood monocytes.51 These proinflammatory molecules released by PlGF may be responsible, to some extent, for the increased incidence of vascular occlusions in SCD subjects. Recent studies show that leukocyte adhesion to endothelium may be the primary event in initiating vascular occlusion, secondarily causing RBCs from subjects with SCD to adhere to the leukocytes or to endothelium.15,54 Therefore, it is plausible that PlGF may increase the adhesion of leukocytes either by (1) directly activating monocytes and increasing their adherence to endothelium or (2) secondarily activating neutrophils through release of proinflammatory molecules from monocytes. And, finally, PlGF increases VEGF production from monocytes. PlGF and VEGF have been shown to form heterodimers and may further modulate the inflammatory tone.
Hebbel and colleagues have elegantly shown that RBCs from subjects with SCD adhere abnormally to endothelial cells and their adhesiveness correlates with clinical severity.16,55 Increased expression of adhesion molecules both on sickle RBCs56,57 and endothelial cells8 has been shown to contribute to the increased adhesion. VEGF has been shown to increase the expression of cell adhesion molecules ICAM-1 and VCAM-1 (vascular cell adhesion molecule-1) on the endothelium, thereby increasing endothelial cell adhesivity.8,17,58 PlGF shares significant homology with VEGF and could induce a proadhesive endothelial state either through direct activation of endothelial cells or indirectly, by increasing monocyte release of VEGF or proinflammatory cytochemokines.
A perplexing feature of SCD is its highly variable clinical phenotype and intermittent episodes of vascular occlusions, with prolonged and varied symptom-free intervals, suggesting that phenomena other than sickling may be involved. Several large clinical studies consistently show that the degree of anemia and leukocytosis at baseline conditions are 2 independent factors that predict the severity of SCD.59-64 In view of our findings, SCD subjects who are more anemic would have increased tissue hypoxia and higher erythropoietin concentrations, factors that would lead to higher PlGF expression. This would result in increased vascular occlusive events through its effect on monocytes. Croizat et al have shown that the degree of anemia in SCD correlates directly with the erythropoietin concentrations in plasma and the clinical severity of disease.33 Our data suggest that these factors would increase PlGF levels, which would then secondarily activate monocytes, thus contributing to the clinical severity.
Moreover, very recently, Rafii and colleagues have shown that PlGF mobilizes hematopoietic progenitor cells by increasing matrix metalloproteinase-9 (MMP-9) expression in bone marrow.50 MMP-9 mediates cleavage of c-kit, allowing recruitment and egression of progenitor cells and leukocytes.50 A single injection of PlGF-adenoviral vector in mice resulted in leukocytosis for 2 to 3 weeks that was MMP-9 mediated. In comparison, the adeno-null vector had no effect.50 Therefore, PlGF may be also be contributing to the leukocytosis observed in SCD at steady state via MMP-9. Additionally, the increased cytochemokines, TNF-α and IL-8, released from PlGF-stimulated monocytes could also promote leukocyte chemotaxis.65,66 Leukocytosis has been shown to correlate with the degree of anemia in several clinical studies. It will be interesting to correlate the degree of anemia and leukocytosis with PlGF levels and the degree of monocyte activation.
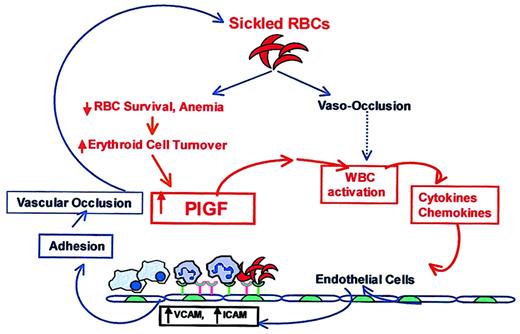
In conclusion, the molecular defect in SCD has been known for years, but the reasons for leukocytosis and activation of monocyte and endothelial cell at steady state,9,10,53,67 in the absence of infection or vascular occlusion, are largely unknown.68 Activation of leukocytes and elevation of cytokines, chemokines, and VEGF in SCD has always been attributed to events secondary to vascular occlusion and the resulting inflammation, as shown in the schema in Figure 8 (shown in blue). PlGF may provide the first link between these 2 phenomena (Figure 8, shown in red): We show that erythroid cells intrinsically release PlGF, a factor that directly activates leukocytes, and specifically monocytes, and may thus modulate the inflammatory tone and clinical severity of SCD.
A working model showing the link between increased erythropoiesis in SCD and inflammation. In SCD, increased sickling of RBCs leads to vaso-occlusion, which has been suggested to activate leukocytes. Alternatively, we propose that increased PlGF, resulting from erythroid hyperplasia and increased erythropoietin levels in SCD, activates leukocytes, specifically monocytes, and results in elevated proinflammatory cytokines and chemokines. These cytokines have been previously shown to up-regulate expression of VCAM and ICAM-1 in cultured endothelial cells and thereby increase the adherence of sickle RBCs and leukocytes through their counterreceptors. The increased adherence of these cells to endothelium has also been shown to cause vaso-occlusion in in vivo models. In this proposed schema of events in SCD, previously published associations are shown in blue, speculations are indicated with a dashed blue line, and our data and its interpretations are depicted in red. We conclude that erythroid cells intrinsically release a factor, PlGF, which directly activates leukocytes, specifically monocytes, to modulate the inflammatory tone and clinical severity of SCD.
A working model showing the link between increased erythropoiesis in SCD and inflammation. In SCD, increased sickling of RBCs leads to vaso-occlusion, which has been suggested to activate leukocytes. Alternatively, we propose that increased PlGF, resulting from erythroid hyperplasia and increased erythropoietin levels in SCD, activates leukocytes, specifically monocytes, and results in elevated proinflammatory cytokines and chemokines. These cytokines have been previously shown to up-regulate expression of VCAM and ICAM-1 in cultured endothelial cells and thereby increase the adherence of sickle RBCs and leukocytes through their counterreceptors. The increased adherence of these cells to endothelium has also been shown to cause vaso-occlusion in in vivo models. In this proposed schema of events in SCD, previously published associations are shown in blue, speculations are indicated with a dashed blue line, and our data and its interpretations are depicted in red. We conclude that erythroid cells intrinsically release a factor, PlGF, which directly activates leukocytes, specifically monocytes, to modulate the inflammatory tone and clinical severity of SCD.
Prepublished online as Blood First Edition Paper, April 24, 2003; DOI 10.1182/blood-2002-11-3422.
Supported by the USC Comprehensive Sickle Cell Center (CSSC) grant HL-PO1-48484 and the Sickle Cell Disease Association of America (SCDAA). Punam Malik is the recipient of the USC CSSC scholar award and Lori Luck the recipient of the SCDAA fellowship award.
The publication costs of this article were defrayed in part by page charge payment. Therefore, and solely to indicate this fact, this article is hereby marked “advertisement” in accordance with 18 U.S.C. section 1734.
We thank Dr Martine Torres for critically reviewing the manuscript and the helpful suggestions and Robert Weihing for help with obtaining patient samples and performing the ELISA for PlGF.


This feature is available to Subscribers Only
Sign In or Create an Account Close Modal