Abstract
Coactivators p300 and CREB (cyclic adenosine monophosphate [cAMP]–response element binding protein)–binding protein (CBP) serve as an integrator for gene transcription. Their relative involvement in regulating cyclooxygenase-2 (COX-2) promoter activity had not been characterized. Using fibroblast and macrophage COX-2 transcription as a model, we determined p300 and CBP levels in nuclear extracts and their binding to a COX-2 promoter probe. CBP level was barely detectable and there was little CBP binding. In contrast, p300 was detectable in nucleus and its binding to a COX-2 promoter probe was enhanced by phorbol 12-myristate 13-acetate (PMA), interleukin-1β (IL-1β), or lipopolysaccharide (LPS). Binding of p300/CBP-associated factor (PCAF) was also up-regulated. COX-2 proteins and promoter activities induced by these agonists were augmented by p300 overexpression. Early region 1A (E1A), but not its deletion mutant, abrogated COX-2 expression induced by inflammatory mediators and with or without p300 overexpression. Molecular analysis of p300 revealed the requirement of multiple domains, including histone acetyltransferase (HAT) for COX-2 transactivation. Furthermore, roscovitine, an indirect inhibitor of p300 HAT, and histone deacetylase-1 transfection completely abolished COX-2 promoter activity. We conclude that p300 is the predominant coactivator that is essential for COX-2 transcriptional activation by proinflammatory mediators.
Introduction
Cyclooxygenase-2 (COX-2) catalyzes the formation of prostaglandin H2 (PGH2), which is the common precursor for synthesis of diverse prostaglandins and thromboxane.1 COX-2 expression is induced by a myriad of proinflammatory cytokines, phorbol esters, lipopolysaccharide (LPS), and mitogenic factors.2 COX-2 is well recognized to play a key role in inflammation.3,4 It has recently been shown that COX-2 is overexpressed in atheromatous plaque and its metabolite, PGE2, plays an important role in plaque instability.5 COX-2 overexpression has also been shown to cause colon cancer growth.6-8 The diverse actions of COX-2 that result in tissue damage and tumor growth depend on COX-2 transcriptional activation. COX-2 promoter activation by proinflammatory mediators and mitogenic factors has been characterized considerably. Human COX-2 core promoter region comprises a canonical twin arginine translocation A (TATA) and several enhancer elements located within approximately 500 base pair (bp) from the transcription start sites.9,10 Several enhancer elements have been demonstrated to be essential for promoter activation by proinflammatory mediators. They include a cyclic adenosine monophosphate (cAMP) response element (CRE) at –53 to –59, a CCAAT/enhancer binding protein (C/EBP) element at –125 to –132, and 2 κB elements (–213 to –222 and –438 to –447).11,12 Each proinflammatory mediator requires binding of a combination of different transactivators to their respective enhancer elements.13 For example, phorbol 12-myristate 13-acetate (PMA) increases binding of activator protein-1 (AP-1) to CRE and C/EBPβ to C/EBP elements while tumor necrosis factor α (TNF-α) induces nuclear factor κB (NF-κB) binding to κB sites.14,15 Several reports have suggested involvement of p300 coactivator in COX-2 transcriptional regulation,15,16 but it remains unclear whether p300 is essential for COX-2 transcription. CREB (cyclic AMP–response element binding protein)–binding protein (CBP) is closely related to p300 with a high degree of sequence homology.17-19 They share binding domains and histone acetyltransferase (HAT) activity.17-19 They interact with DNA-bound transactivators and bind general transcription factors such as transcription factor IIB (TFIIB), thereby integrating the transcriptional signals from external stimuli.17-19 It is generally believed that they have similar functions and play redundant roles in gene expression. However, a recent report suggested that they possess structural differences that may influence their involvement in different gene transcription.20 The role of CBP in COX-2 transcriptional activation had not been previously reported. In order to understand the relative importance of p300 and CBP in COX-2 promoter activation by proinflammatory mediators, we investigated the binding of p300/CBP to COX-2 promoter and the regulation of proinflammatory mediator-induced COX-2 expression by p300/CBP in human foreskin fibroblasts (HFb) and a mouse macrophage, RAW 264.7. Our results show that CBP was barely detectable and its binding was negligible in these 2 cell types. In contrast, the binding of p300 and its interactive protein PCAF (p300/CBP-associated factor) to COX-2 promoter region was up-regulated by PMA, interleukin-1β (IL-1β), and LPS in HFb and by LPS in macrophages. Inhibitors of p300 and HAT significantly reduced COX-2 protein levels and promoter activities in PMA-induced HFb and LPS-induced macrophage. Furthermore, PMA-stimulated COX-2 promoter in HFb and LPS-induced promoter activity in RAW 264.7 were similarly abrogated by transfection of histone deacetylase-1 (HDAC-1). These findings indicate that p300 plays an essential role in COX-2 promoter activation by proinflammatory mediators.
Materials and methods
Plasmids
A promoter region of human COX-2 gene (–891/+9 from the transcription start site) was constructed into a luciferase reporter vector pGL3 as previously described.10 Expression vectors containing full-length p300 (pCL.p300) and its HAT deletion mutant (pCL.p300ΔHAT, Δ1472-1522) were provided by Dr Joan Boyes. Expression vectors containing p300 carboxy- and amino-terminal deletion mutants were constructed by cloning the respective cDNAs amplified by polymerase chain reaction (PCR) into the HindIII/XhoI sites of mammalian expression vector pCMV-Tag2 (Stratagene, La Jolla, CA). Expression vectors for early region 1A (E1A) and its mutant ΔE1A (Δ2-36) were provided by Dr Pradip Raychaudhuri. The expression vector of HDAC-1 was provided by Dr I. Talianidis.
Cell culture and treatment
HFb and RAW 264.7 cells were cultured in Dulbecco modified Eagle medium supplemented with 10% fetal bovine serum and 1:100 dilution of an antibiotic-antimycotic solution. For all experiments, 80% to 90% confluent cells were cultured in serum-free medium for 24 hours. After washing with phosphate-buffered saline (PBS), HFb were incubated in fresh medium in the presence or absence of 100 nM PMA, 10 ng/mL IL-1β, or 2 μg/mL LPS (Escherichia coli O26:B6) (all from Sigma, St Louis, MO) at 37°C for 4 hours. RAW 264.7 cells were incubated with or without LPS (2 μg/mL) at 37°C for 4 hours. We chose 4-hour incubation because previous kinetic experiments show that COX-2 protein levels in either cell type reached plateau at 4 hours after incubation with the indicated agonists for 4 hours.21,22 The treated cells were washed 3 times with chilled PBS, harvested, and processed as in the experiments described in “Western blot analysis.” All the tissue culture reagents were obtained from Life Technologies (Grand Island, NY). Roscovitine was obtained from Sigma.
Transient transfection
The transfection procedure was performed as previously described.15 In brief, 10 μL of Lipofetamine 2000 reagent (Invitrogen, Carlsbad, CA) and 4 μg of DNA constructs were mixed, and the mixture was slowly added to each well of HFb in a 6-well plate and incubated for 24 hours. The cells were washed, incubated in serum-free medium, and treated with an agonist. The expressed luciferase activity was measured in a luminometer (TD-20/20; Turner Design, Sunnyvale, CA).
Western blot analysis
Western blot analysis was performed as previously described with minor modifications.23 In brief, cell pellets were lysed with lysis buffer containing 50 mM Tris-HCl (pH 7.5), 150 mM NaCl, 1 mM EDTA (ethylenediaminetetraacetic acid), 1 mM phenylmethylsulfonyl fluoride (PMSF), 1 μg/mL leupeptin, 5 μg/mL aprotinin, 1% Nonidet P450, 0.5% sodium deoxycholate, and 0.1% sodium dodecyl sulfate (SDS). The lysate was centrifuged at 10 000 g for 10 minutes and the supernatant was collected and boiled for 5 minutes. Protein concentration was determined by Bio-Rad kit (Hercules, CA) using bovine serum albumin as a standard. Lysate proteins were separated in a 4% to 15% SDS-polyacrylamide minigels (Bio-Rad) and then electrophoretically transferred to a nitrocellulose membrane (Amersham Pharmacia Biotech, Piscataway, NJ). Western blots were probed with 1 μg/mL of a specific rabbit polyclonal antibody to COX-2 (Cayman, Ann Arbor, MI), p300, CBP, or PCAF (Santa Cruz Biotechnology, Santa Cruz, CA). To ensure the antibody sensitivity, we also used 1 μg/mL monoclonal immunoglobulin G1 (IgG1) against CBP from Santa Cruz Biotechnology and against p300 from Oncogene (Boston, MA). The protein bands were detected by enhanced chemiluminescence.
Immunoprecipitation
Interaction of p300 with transactivators was determined by immunoprecipitation as previously described.15 Nuclear extract proteins (800 μg) prepared from HFb were incubated with a specific rabbit polyclonal antibody to CREB-2, c-Jun, C/EBPβ, p50/p65 NF-κB, or a nonimmune rabbit IgG (all from Santa Cruz Biotechnology), at a final concentration of 4 μg/mL each, overnight at 4°C. The immune complex was pulled down by protein A/G plus agarose (Santa Cruz Biotechnology), and after washing with RIPA buffer (50 mM Tris-HCL [pH 7.5], 150 mM NaCl, 1 mM EDTA, 1 mM PMSF, 1 μg/mL leupeptin, 5 μg/mL aprotinin, 1% Nonidet P40, 0.5% sodium deoxycholate, and 0.1% sodium dodecyl sulfate) 4 times, the immunoprecipitated proteins were separated by SDS–polyacrylamide gel electrophoresis (PAGE) and analyzed by Western blotting using a p300 antibody (Santa Cruz Biotechnology).
DNA-protein binding assay
Binding of p300 to transactivator–COX-2 promoter DNA complexes was assayed by a new technique recently described.24 Eighty percent to 90% confluent HFb were incubated in serum-free medium and treated with an agonist for 4 hours before nuclear extracts were prepared. The biotin-labeled double-stranded oligonucleotide probes were synthesized by IDT (Integrated DNA Technologies, Coralville, IA) based on human COX-2 promoter sequence –30 to –453.10 A biotinylated nonrelevant sequence, 5′-AGTCATCGAGTCACATGGG-3′ that does not harbor known enhancer elements, was used as a binding control. The binding assay was performed by mixing 200 μg HFb nuclear extracts, 2 μg biotin-labeled DNA oligonucleotides, and 20 μL streptavidin agarose beads with 70% slurry. The mixture was incubated at room temperature for 1 hour with shaking. Beads were then pelleted down by centrifugation at 5000g in a microcentrifuge for 30 seconds and washed with cold PBS 3 times. The binding proteins were separated on 4% to 15% PAGE followed by Western blot analysis probed with antibodies against p300.
Chromatin immunoprecipitation (ChIP)
The assay was done as previously described.15 Eighty percent to 90% confluent HFb were serum starved for 24 hours and treated with an agonist at 37°C for 4 hours. One percent formaldehyde was added to the culture medium, and after incubation for 20 minutes at 37°C, cells were washed twice in PBS, scraped, and lysed in lysis buffer (1% SDS, 10 mM Tris-HCl [pH 8.0], with 1 mM PMSF, pepstatin A, and aprotinin) for 10 minutes at 4°C. Lysates were sonicated 5 times for 10 seconds each and the debris was removed by centrifugation at 15 000g in a microcentrifuge for 4 minutes. One third of the lysate was used as DNA input control. The remaining two thirds of the lysate was diluted 10-fold with a dilution buffer (0.01% SDS, 1% Triton X100, 1 mM EDTA, 10 mM Tris-HCl [pH 8.0], and 150 mM NaCl) followed by incubation with an anti-p300 antibody or a nonimmune rabbit IgG (Santa Cruz Biotechnology) overnight at 4°C. Immunoprecipitated complexes were collected by using protein G–Sepharose beads. The precipitates were extensively washed and incubated in the elution buffer (1% SDS and 0.1 M NaHCO3) at room temperature for 20 minutes. Cross-linking of protein-DNA complexes was reversed at 65°C for 5 hours followed by treatment with 100 μg/mL proteinase K for 3 hours at 50°C. DNA was extracted 3 times with phenol/chloroform and precipitated with ethanol. Pellets were resuspended in Tris-EDTA (TE) buffer and subjected to PCR amplification using specific COX-2 promoter primers: 5′ primer, –709 CTGTTGAAAGCAACTTAGCT –690; and 3′ primer, –32 AGACTGAAAACCAAG CCCAT –51. The resulting product of 678 bp was separated by agarose gel electrophoresis.
Densitometric analysis
Scion Image Software (Frederick, MD) was used to determine the density of protein bands detected by Western blots. The data are expressed as an arbitrary unit.
Statistics analysis
Three separate experiments done in duplicate were performed and mean values and standard errors (SEM) were calculated.
Results
Proinflammatory mediators increased p300 and PCAF binding to COX-2 promoter
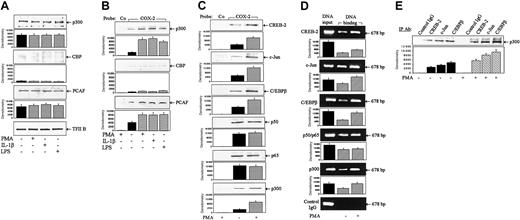
To understand the relative importance of p300, CBP, and PCAF in COX-2 promoter activation by proinflammatory mediators, we investigated the cellular levels of these 3 proteins and their binding to COX-2 promoter and interaction with transcriptional transactivators in a human fibroblast model. We first estimated the cellular level of CBP by Western blots using polyclonal and monoclonal antibodies. Both antibodies yielded a barely detectable basal CBP level in HFb nuclear extracts, which was not increased by PMA, IL-1β, or LPS (Figure 1A). Regardless of the antibodies used, the basal p300 level was detectable, which was not influenced by any of the proinflammatory mediators (Figure 1A). We next evaluated the effect of PMA, IL-1β, and LPS on p300 and CBP binding to a core COX-2 promoter probe by a streptavidin bead pulldown assay. There was little CBP binding (Figure 1B). Basal p300 binding was low but was increased by 4- to 5-fold by PMA, IL-1β, or LPS (Figure 1B). PCAF is a HAT that binds p300 and CBP and modifies chromatin structure.25 We determined whether PCAF was influenced by proinflammatory mediators. The basal level of PCAF in nuclear extracts was not influenced by any of the agonists (Figure 1A), whereas its binding to COX-2 promoter probe was increased by all 3 agonists (Figure 1B). Neither p300 nor CBP or PCAF binding was detected when a control biotinylated probe was used (Figure 1B).
PMA, IL-1β, and LPS increased p300 and PCAF binding without altering their protein levels. (A) Western blot analysis of p300, CBP, and PCAF protein levels in nuclear extracts prepared from HFb treated with or without PMA (100 nM), IL-1β (10 ng/mL), or LPS (2 μg/mL) for 4 hours. General transcription factor TFIIB was used as a nuclear protein control. (B) Binding of p300, CBP, and PCAF to a COX-2 promoter probe. Nuclear extracts prepared from HFb were incubated with a 424-bp biotinylated COX-2 promoter probe (–30 to –453) and streptavidin-agarose beads for one hour, and the complex was obtained after centrifugation. Proteins in the complex were resolved by Western blot analysis. Co indicates a control nonrelevant probe; and COX-2, the COX-2 probe. (C) Correlation of transactivator binding with p300 binding. Binding assays were performed as described in panel B. Multiple proteins were simultaneously analyzed in the pulldown complex. (D) Binding of coactivators and p300 to chromatin COX-2 promoter. ChIP assays were performed as described in “Materials and methods.” DNA input denotes the loading of an equal amount of nuclear extract DNA without immunoprecipitation. Control IgG indicates rabbit normal IgG. (E) Interaction between p300 and transactivators. Nuclear extracts from HFb treated with or without PMA (100 nM) for 4 hours were immunoprecipitated with each indicated antibody, and p300 in the complex was detected by Western blots. IP indicates immunoprecipitation; and control IgG, rabbit normal IgG. In all panels, the top panel shows a representative of 3 experiments and the bottom panel shows mean ± SEM of densitometry from 3 experiments.
PMA, IL-1β, and LPS increased p300 and PCAF binding without altering their protein levels. (A) Western blot analysis of p300, CBP, and PCAF protein levels in nuclear extracts prepared from HFb treated with or without PMA (100 nM), IL-1β (10 ng/mL), or LPS (2 μg/mL) for 4 hours. General transcription factor TFIIB was used as a nuclear protein control. (B) Binding of p300, CBP, and PCAF to a COX-2 promoter probe. Nuclear extracts prepared from HFb were incubated with a 424-bp biotinylated COX-2 promoter probe (–30 to –453) and streptavidin-agarose beads for one hour, and the complex was obtained after centrifugation. Proteins in the complex were resolved by Western blot analysis. Co indicates a control nonrelevant probe; and COX-2, the COX-2 probe. (C) Correlation of transactivator binding with p300 binding. Binding assays were performed as described in panel B. Multiple proteins were simultaneously analyzed in the pulldown complex. (D) Binding of coactivators and p300 to chromatin COX-2 promoter. ChIP assays were performed as described in “Materials and methods.” DNA input denotes the loading of an equal amount of nuclear extract DNA without immunoprecipitation. Control IgG indicates rabbit normal IgG. (E) Interaction between p300 and transactivators. Nuclear extracts from HFb treated with or without PMA (100 nM) for 4 hours were immunoprecipitated with each indicated antibody, and p300 in the complex was detected by Western blots. IP indicates immunoprecipitation; and control IgG, rabbit normal IgG. In all panels, the top panel shows a representative of 3 experiments and the bottom panel shows mean ± SEM of densitometry from 3 experiments.
Coactivator p300 is capable of binding to diverse DNA-bound transactivators including CREB, c-Jun, C/EBPβ, and NF-κB. We determined whether the level of p300 binding was correlated with that of transactivator binding when cells were stimulated by PMA. In agreement with previously reported data, PMA enhanced CREB-2, c-Jun, and C/EBPβ but not p50 or p65 NF-κB binding to COX-2 promoter (Figure 1C). Binding of p300 was concordantly increased by PMA (Figure 1C), whereas CBP binding was not (data not shown). Nuclear extract proteins were immunoprecipitated with antibodies to each of the transactivators, and p300 level in each precipitate was analyzed by Western blots. The results show detectable basal p300 complexed with CREB-2, c-Jun, and C/EBPβ, which was increased by PMA (Figure 1D). Binding of p300 and transactivators was further evaluated by ChIP assay. Results revealed an increased CREB-2, c-Jun, and C/EBPβ binding to the chromatin COX-2 promoter region (Figure 1E). Binding of p300 was also increased. A nonimmune rabbit IgG was used as a control in ChIP, and COX-2 promoter region was undetectable (Figure 1E). DNA input for each experiment is also shown in Figure 1E. Taken together, these data confirm the up-regulation of p300 recruitment to the COX-2 promoter region through increased CREB-2, c-Jun, and C/EBPβ binding by PMA.
Overexpression of p300 or CBP up-regulated COX-2 protein levels and promoter activities
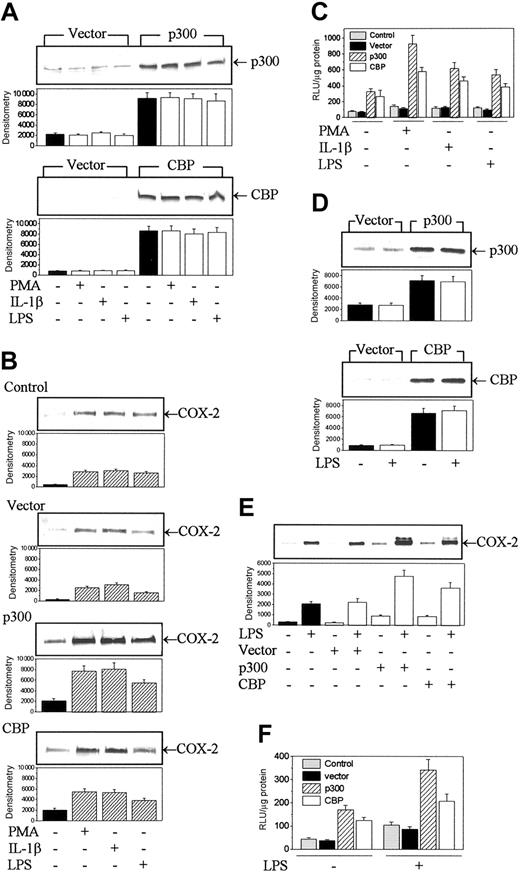
The protein availability of p300 and CBP in nuclear extracts is considered to be a limiting step in gene regulation. In agreement with this concept, we found that overexpression of p300 or CBP by transient transfection of HFb (Figure 2A) up-regulated COX-2 protein expression (Figure 2B) and promoter activity (Figure 2C) stimulated by PMA, IL-1β, or LPS. To ascertain that the up-regulation takes place in other inflammatory cells, we evaluated the effect of p300 and CBP overexpression on COX-2 expression in RAW 264.7 macrophages. Similar to the results in HFb, p300 or CBP expression by transient transfection (Figure 2D) resulted in an increase in COX-2 proteins and promoter activities (Figure 2E-F). Transient transfection of either cell type with a control vector did not increase p300 or CBP level nor did it influence COX-2 protein and promoter levels when compared with nontransfected cells (Figure 2). Thus, COX-2 expression is up-regulated by p300 or CBP overexpression in human fibroblasts and murine macrophages. Judging from very low CBP protein levels in HFb (Figure 1A) and RAW 264.7 (data not shown), p300 plays a more dominant role than CBP in mediating and regulating COX-2 expression in response to inflammatory signals.
CBP and p300 overexpression enhanced COX-2 protein levels and promoter activities in HFb and RAW 264.7. (A) CBP and p300 protein levels in transduced HFb. HFb were transfected with plasmids encoding p300, CBP, or the empty vector (mock control) and then treated with or without PMA, IL-1β, or LPS. Protein levels of p300 were analyzed by Western blots. CBP or p300 level in mock-transfected cells was similar to that in native cells. (B) Influence of p300 and CBP transduction on COX-2 protein levels in HFb. (C) Effect of p300 and CBP transduction on COX-2 promoter activities in HFb. HFb were cotransfected with luciferase expression vectors and plasmids encoding p300 or CBP. The promoter activity was expressed as relative light unit (RLU). (D) CBP and p300 protein levels in RAW 264.7 cells transfected with CBP or p300 vectors. (E) Influence of p300 and CBP transduction on COX-2 protein levels in RAW 264.7 cells. (F) Effect of p300 or CBP overexpression on COX-2 promoter activity. In panels A, B, D, and E, the top panel shows a representative of 3 experiments and the bottom panel shows mean ± SEM of the densitometry from 3 experiments. Each bar in panels C and F also denotes mean ± SEM of 3 experiments.
CBP and p300 overexpression enhanced COX-2 protein levels and promoter activities in HFb and RAW 264.7. (A) CBP and p300 protein levels in transduced HFb. HFb were transfected with plasmids encoding p300, CBP, or the empty vector (mock control) and then treated with or without PMA, IL-1β, or LPS. Protein levels of p300 were analyzed by Western blots. CBP or p300 level in mock-transfected cells was similar to that in native cells. (B) Influence of p300 and CBP transduction on COX-2 protein levels in HFb. (C) Effect of p300 and CBP transduction on COX-2 promoter activities in HFb. HFb were cotransfected with luciferase expression vectors and plasmids encoding p300 or CBP. The promoter activity was expressed as relative light unit (RLU). (D) CBP and p300 protein levels in RAW 264.7 cells transfected with CBP or p300 vectors. (E) Influence of p300 and CBP transduction on COX-2 protein levels in RAW 264.7 cells. (F) Effect of p300 or CBP overexpression on COX-2 promoter activity. In panels A, B, D, and E, the top panel shows a representative of 3 experiments and the bottom panel shows mean ± SEM of the densitometry from 3 experiments. Each bar in panels C and F also denotes mean ± SEM of 3 experiments.
Adenoviral E1A suppressed COX-2 expression induced by proinflammatory mediators
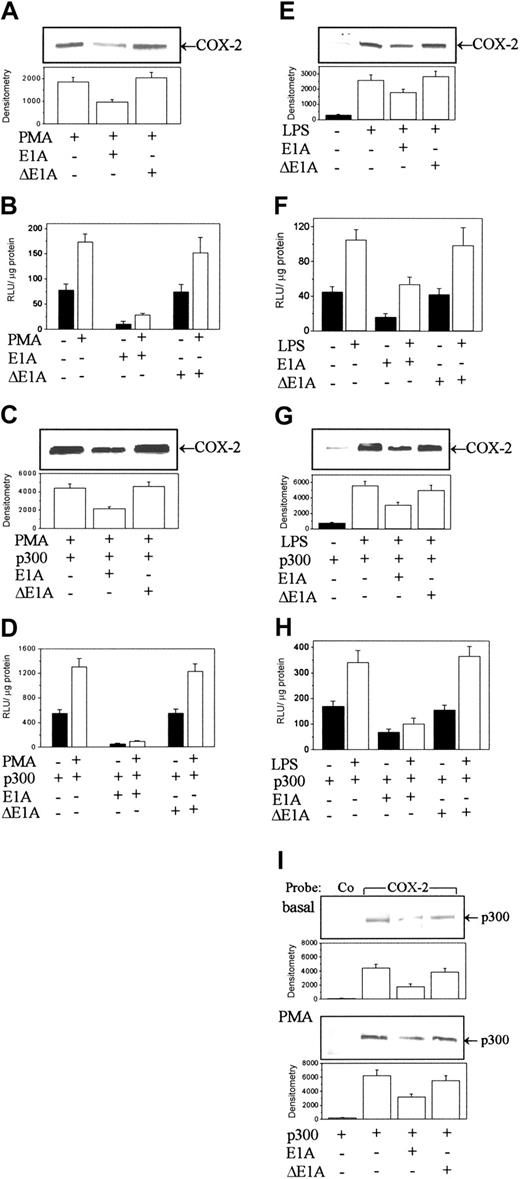
E1A blocks p300 binding and HAT activity.26 Overexpression of E1A in HFb by transient transfection suppressed PMA-induced COX-2 protein levels, whereas overexpression of an E1A deletion mutant (ΔE1A) had no effect (Figure 3A). E1A but not ΔE1A overexpression suppressed COX-2 promoter activity (Figure 3B). E1A overexpression also abrogated COX-2 protein levels (Figure 3C) and promoter activity (Figure 3D) in p300-transduced HFb treated with or without PMA. Again, overexpression of ΔE1A did not reduce COX-2 protein levels (Figure 3C) or promoter activities (Figure 3D). Overexpression of E1A and ΔE1A exerted comparable effects on COX-2 promoter activities and protein levels in native (Figure 3E-F) and p300-transfected (Figure 3G-H) RAW 264.7 cells induced by LPS. We also determined the influence of overexpression of E1A and ΔE1A mutant on p300 binding to COX-2 promoter. The results show that E1A decreased p300 binding to COX-2 promoter in basal or PMA-induced HFb, whereas ΔE1A had no effect (Figure 3I). These results confirm that p300 is essential for COX-2 expression.
E1A but not an E1A deletion mutant (ΔE1A) transduction blocked COX-2 expression and p300 binding to COX-2 promoter in HFb and RAW 264.7 cells. (A-D) Protein levels (A,C) and promoter activities (B,D) in PMA-treated HFb without p300 transfection and with p300 transfection (C-D). (E-H) COX-2 protein levels (E,G) and promoter activities (F,H) in LPS-treated RAW 264.7 cells without (E-F) and with (G-H) p300 transduction. (I) The binding of p300 to a COX-2 versus control probe. Co indicates a control probe; and COX-2, the COX-2 probe. In panels A, C, E, G, and I, the top panel shows a representative blot and the bottom panel shows mean ± SEM of the densitometry from 3 experiments. Other bars also denote mean ± SEM of 3 experiments.
E1A but not an E1A deletion mutant (ΔE1A) transduction blocked COX-2 expression and p300 binding to COX-2 promoter in HFb and RAW 264.7 cells. (A-D) Protein levels (A,C) and promoter activities (B,D) in PMA-treated HFb without p300 transfection and with p300 transfection (C-D). (E-H) COX-2 protein levels (E,G) and promoter activities (F,H) in LPS-treated RAW 264.7 cells without (E-F) and with (G-H) p300 transduction. (I) The binding of p300 to a COX-2 versus control probe. Co indicates a control probe; and COX-2, the COX-2 probe. In panels A, C, E, G, and I, the top panel shows a representative blot and the bottom panel shows mean ± SEM of the densitometry from 3 experiments. Other bars also denote mean ± SEM of 3 experiments.
The p300 deletion mutations abolished the COX-2 expression stimulated by PMA
The p300 contains multiple domains that are involved in interaction with transactivators, nuclear hormone receptor (NHR), cysteinehistidine 1 (C/H1), C/H2, C/H3, CREB-binding domain (KIX), and general transcription factors (C/H3). Its HAT activity resides in a region between C/H2 and C/H3 (Figure 4A). We made several C-terminal and N-terminal deletion mutants to evaluate the p300 molecular requirement for COX-2 promoter activity (Figure 4A). These deletion mutants expressed appropriate polypeptides in HFb detected by Western blots (Figure 4B). Deletion of C-terminal regions containing C/H3 (Del-1), or C/H3 plus C/H2 and HAT domains (Del-2) resulted in a complete loss of basal and PMA-induced COX-2 promoter activity (Figure 4C). Similarly, deletion of a N-terminal region containing KIX plus C/H1 and NHR domains (Del-3) also resulted in a total loss of COX-2 promoter activity (Figure 4B). These results indicate that intact p300 is absolutely required for up-regulating COX-2 promoter activity.
Transfection of N- or C-terminal deletion mutants of p300 abolished the p300 enhancing effect. (A) Linear schemes show wild-type and deletion mutants. Domains are indicated by arrows. (B) Protein levels of the overexpressed wild-type p300 and its mutants determined by Western blots. (C) COX-2 promoter activities in HFb transfected with wild-type p300 and mutants. Control refers to untransfected HFb. Similar results were detected in RAW 264.7 cells (data not shown). Each bar denotes mean ± SEM of 3 experiments.
Transfection of N- or C-terminal deletion mutants of p300 abolished the p300 enhancing effect. (A) Linear schemes show wild-type and deletion mutants. Domains are indicated by arrows. (B) Protein levels of the overexpressed wild-type p300 and its mutants determined by Western blots. (C) COX-2 promoter activities in HFb transfected with wild-type p300 and mutants. Control refers to untransfected HFb. Similar results were detected in RAW 264.7 cells (data not shown). Each bar denotes mean ± SEM of 3 experiments.
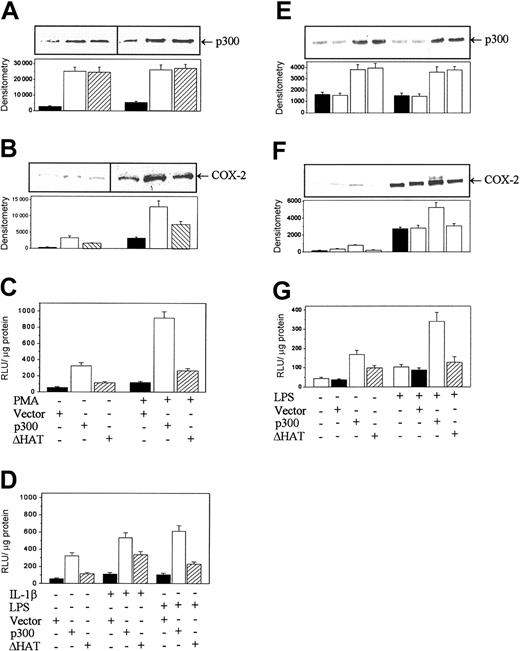
Transfection of HFb with deletion of a core HAT domain (Δ1472-1522, ΔHAT), which did not alter the immunoreactive p300 level (Figure 5A), resulted in an attenuated increase in basal COX-2 protein and a marked reduction in PMA-stimulated protein levels (Figure 5B). Similarly, ΔHAT transfection caused a 2.5-fold increase in PMA-stimulated promoter activity compared with a 9-fold increase with wild-type (WT) p300 overexpression (Figure 5C). ΔHAT transduction also elicited an attenuated COX-2 promoter response to IL-1β and LPS stimulation (Figure 5D). Transfection of ΔHAT mutant exerted a similar effect on COX-2 promoter activity in LPS-induced RAW 264.7 cells (Figure 5E-G).
Deletion mutation of p300 HAT reduced the enhancing effect of p300 on COX-2 expression. (A-B) p300 (A) and COX-2 (B) protein levels in nuclear extracts of ΔHAT- or p300-transduced HFb determined by Western blots. (C-D) COX-2 promoter activities in p300- or ΔHAT-transduced HFb stimulated with or without PMA (C) or with or without IL-1β or LPS (D). (E-F) p300 (E) and COX-2 (F) protein levels in nuclear extracts of RAW 264.7 transduced with ΔHAT or p300. (G) COX-2 promoter activities in transduced RAW 264.7 cells treated with or without LPS. In panels A, B, E, and F, the top panel shows a representative blot and the bottom panel shows mean ± SEM of the densitometry of 3 experiments. Each bar in C, D, and G denotes mean ± SEM of 3 experiments. A mock vector and normal control were included in all experiments with similar results.
Deletion mutation of p300 HAT reduced the enhancing effect of p300 on COX-2 expression. (A-B) p300 (A) and COX-2 (B) protein levels in nuclear extracts of ΔHAT- or p300-transduced HFb determined by Western blots. (C-D) COX-2 promoter activities in p300- or ΔHAT-transduced HFb stimulated with or without PMA (C) or with or without IL-1β or LPS (D). (E-F) p300 (E) and COX-2 (F) protein levels in nuclear extracts of RAW 264.7 transduced with ΔHAT or p300. (G) COX-2 promoter activities in transduced RAW 264.7 cells treated with or without LPS. In panels A, B, E, and F, the top panel shows a representative blot and the bottom panel shows mean ± SEM of the densitometry of 3 experiments. Each bar in C, D, and G denotes mean ± SEM of 3 experiments. A mock vector and normal control were included in all experiments with similar results.
The p300 HAT deletion mutant had a reduced binding
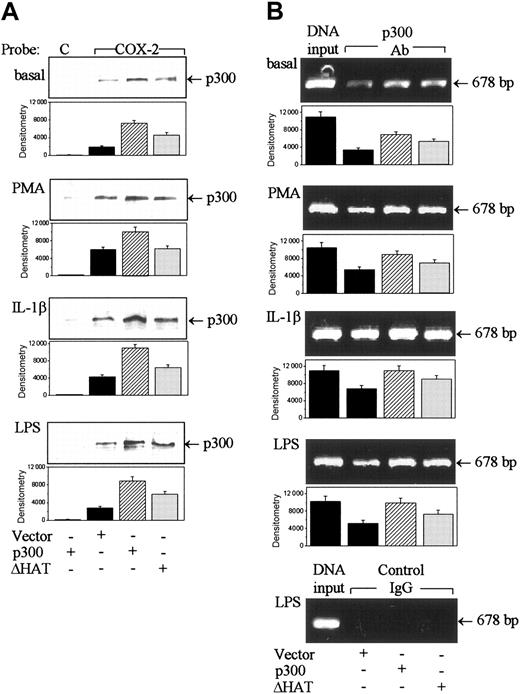
The p300 HAT is known to acetylate chromatin histone thereby increasing accessibility to transactivators.27 We were therefore interested in determining whether overexpression of ΔHAT would influence p300 binding to COX-2 promoter. Immunoreactive p300 proteins in nuclear extracts were equally enhanced by p300 ΔHAT versus WT p300 overexpression (Figure 5A). PMA did not alter the p300 levels in WT and ΔHAT-transfected cells (Figure 5A). The p300 level detectable in DNA-protein complex from ΔHAT-transduced cells was only about 50% of that from WT p300-transduced cells regardless of whether cells were treated with or without PMA, IL-1β, or LPS (Figure 6A). Binding of p300 to chromatin COX-2 promoter in the absence or presence of PMA, IL-1β, or LPS was also consistently lower in ΔHAT than in WT p300-transduced cells (Figure 6B).
Wild-type p300 but not ΔHAT increased p300 binding to COX-2 promoter in HFb. (A) Effect of p300 or ΔHAT overexpression on p300 binding to a biotinylated COX-2 promoter probe analyzed by a streptavidin-agarose pulldown assay. C indicates a control probe; and COX-2, the COX-2 probe. (B) Influence of wild-type p300 versus ΔHAT transfection on p300 binding to a chromatin COX-2 promoter region. Chromatins were immunoprecipitated with a polyclonal p300 antibody and COX-2 promoter region was analyzed by PCR. The left column of each panel shows DNA input. A nonimmune rabbit normal IgG was included with each stimulus as control. Only the LPS-treated cells are shown. In all panels, the top panel shows a representative of 3 experiments and the bottom panel shows mean ± SEM of the densitometry from 3 experiments.
Wild-type p300 but not ΔHAT increased p300 binding to COX-2 promoter in HFb. (A) Effect of p300 or ΔHAT overexpression on p300 binding to a biotinylated COX-2 promoter probe analyzed by a streptavidin-agarose pulldown assay. C indicates a control probe; and COX-2, the COX-2 probe. (B) Influence of wild-type p300 versus ΔHAT transfection on p300 binding to a chromatin COX-2 promoter region. Chromatins were immunoprecipitated with a polyclonal p300 antibody and COX-2 promoter region was analyzed by PCR. The left column of each panel shows DNA input. A nonimmune rabbit normal IgG was included with each stimulus as control. Only the LPS-treated cells are shown. In all panels, the top panel shows a representative of 3 experiments and the bottom panel shows mean ± SEM of the densitometry from 3 experiments.
Roscovitine inhibited COX-2 promoter activity
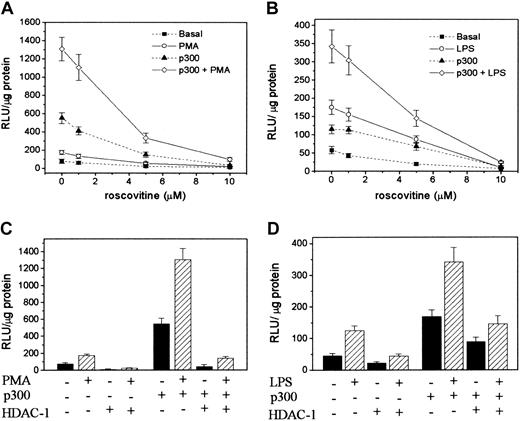
It has been shown that p300 coactivator activity is activated by cyclin E–CdK2.28 Roscovitine, an inhibitor of cyclin E–cyclin-dependent kinase 2 (CdK2), was shown to inhibit p300 activity.28 We evaluated the effect of riscovitine on COX-2 promoter activity. Riscovitine concentration dependently suppressed PMA-induced COX-2 promoter activity in HFb and LPS-induced COX-2 promoter activity in RAW 264.7 (Figure 7A-B). Furthermore, it exerted a similar concentration-dependent suppression of COX-2 promoter activity in p300-transduced HFb with or without PMA treatment or RAW 264.7 with or without LPS treatment (Figure 7A-B).
Roscovitine treatment or HDAC transfection abrogated COX-2 promoter activation. (A-B) Concentration-dependent inhibition of COX-2 promoter activity by roscovitine in HFb and RAW 264.7 with or without p300 transduction and PMA (A) or LPS (B) treatment. (C-D) Influence of HDAC-1 transfection on COX-2 promoter activity in HFb and RAW 264.7 cells with or without p300 transfection and PMA (C) or LPS (D) treatment. Transfection with an empty vector was included in each experiment as control. It yielded basal levels of promoter activities. Each bar denotes mean ± SEM of 3 experiments.
Roscovitine treatment or HDAC transfection abrogated COX-2 promoter activation. (A-B) Concentration-dependent inhibition of COX-2 promoter activity by roscovitine in HFb and RAW 264.7 with or without p300 transduction and PMA (A) or LPS (B) treatment. (C-D) Influence of HDAC-1 transfection on COX-2 promoter activity in HFb and RAW 264.7 cells with or without p300 transfection and PMA (C) or LPS (D) treatment. Transfection with an empty vector was included in each experiment as control. It yielded basal levels of promoter activities. Each bar denotes mean ± SEM of 3 experiments.
Histone deacetlylase-1 repressed COX-2 promoter activity
Gene expression is regulated by a balance between HAT HDACs.29 The role of HDACs in regulating COX-2 expression is unclear. We evaluated the effect of HDAC-1 overexpression on HFb COX-2 promoter activity stimulated with or without PMA. HDAC-1 completely repressed promoter activity in untransduced HFb treated with or without PMA (Figure 7C). Interestingly, HDAC-1 almost completely repressed COX-2 promoter activity in p300-transduced HFb treated with PMA (Figure 7C). HDAC-1 exerted a comparable repression on COX-2 promoter activity in RAW 264.7 cells induced by LPS (Figure 7D). These results suggest that COX-2 promoter activity induced by proinflammatory mediators is up-regulated by p300, which is controlled by deacetylation via the action of HDAC-1.
Discussion
Our results indicate that, of the p300/CBP class of transcriptional coactivators, p300 is a predominant isoform in human fibroblasts and murine macrophages. Its binding to COX-2 promoter is up-regulated by proinflammatory mediators. Coactivator p300 appears to be essential for COX-2 transcriptional activation by proinflammatory mediators, as evidenced by an almost complete repression of COX-2 promoter activity by adenoviral E1A protein, a specific inhibitor of p300/CBP coactivator activity. CBP may not play a significant role in COX-2 promoter activation, as its expression level is barely detectable and its binding to COX-2 promoter probe is negligible. To our knowledge, this is the first report of a selective involvement of p300 in COX-2 transcriptional regulation in human fibroblasts and mouse macrophages. Consistent with previous reports, COX-2 transcription is regulated by the level of p300. Overexpression of p300 by transient transfection augments COX-2 protein level and COX-2 promoter activity induced by proinflammatory mediators. These results are consistent with the interpretation that p300 is expressed in relatively low abundance in cells whose availability poses a limiting step in transcriptional activation.30
A key role of p300 in gene transactivation is conferred by its binding to a myriad of transactivators that are bound to their specific regulatory elements on a promoter. Assessment of p300 binding activity by electrophoretic mobility shift assay was impractical because of the formation of a very large complex that cannot be resolved by gel electrophoresis. In this study, we used a streptavidin-agarose pulldown assay to analyze the levels of p300, CBP, and PCAF in the promoter-protein complex. This assay allows for simultaneous measurements of multiple proteins in the complex.24 Results from this analysis indicate that the levels of p300 and PCAF binding are regulated by PMA, IL-1β, or LPS in HFb. All 3 agonists increased the levels of p300 or PCAF binding to a similar extent. The level of p300 binding stimulated by PMA was in accord with that of CREB-2, c-Jun, and C/EBPβ. These results are consistent with the interpretation that PMA enhances binding of CREB-2, c-Jun, and C/EBPβ to COX-2 promoter in HFb, which in turn recruits an increased level of p300 to the promoter region. As p300 level is increased, its interaction with general transcription factors is more active, thereby up-regulating the promoter activity.
We performed molecular analysis by deletion of mutations of p300 in order to understand the requirement of various binding domains and HAT for COX-2 transcriptional activation by PMA. Our results are consistent with the requirement of multiple binding domains of p300 for COX-2 promoter activity. Deletion of N- or C-terminal domains resulted in a complete loss of p300 transactivating activity. These results support the notion that the p300 transactivation function depends on its interaction with multiple DNA-bound transactivators. Our results further indicate that HAT plays an important role in regulating COX-2 transcriptional activation. Deletion of the core HAT domain results in a marked reduction in p300-mediated COX-2 transcriptional activation. These results are attributed to a major role that p300 HAT plays in opening up the chromatin structure at the COX-2 promoter region to make the enhancer elements in COX-2 promoter accessible to transactivators. It is interesting to note that p300 HAT also plays a role in regulating p300 recruitment to promoter. Despite an identical level of immunoreactive p300 following ΔHAT and wild-type p300 transfection, we found that ΔHAT had a lower level of binding to naked COX-2 promoter probe than WT p300 in cells treated with or without PMA, IL-1β, or LPS. The reason for this is unclear. It is possible that p300 HAT may acetylate transactivators thereby increasing their binding activity, which in turn enhances p300 recruitment. It has been reported that p300 acetylates several transactivators such as p53, GATA-1, p50, p65 NF-κB.31,32 Further studies are needed to determine whether p300 acetylates transactivators involved in COX-2 transcriptional activation induced by these proinflammatory mediators.
It has been reported that CBP is a target of cyclin E–CdK2 phosphorylation and phosphorylated CBP exhibits an increased HAT activity.33 Roscovitine, an inhibitor of cyclin E–CdK2 complex, was shown to suppress HAT activity.28 We, therefore, determined whether roscovitine inhibited COX-2 promoter activity. Our results indicate that cyclin E–CdK2 may be involved in regulating p300-mediated COX-2 transcriptional activation. Furthermore, these results suggest that p300 in human fibroblasts and murine macrophages may be activated by cyclin E–CdK2 in a cell cycle–dependent manner. We have recently shown that COX-2 expression in quiescent fibroblasts treated with serum peaked at G1 phase,34 which coincides with the level of cyclin E–CdK2. This may be explained in part by the stimulating action of cyclin E–CdK2 on p300. These results further support the important role of p300 HAT in COX-2 transcriptional activation.
HDAC-1 is a major isoform of HDAC in mammalian cells. It catalyzes removal of acetyl moieties from core histones.35 Its role in regulating COX-2 promoter activity had not been reported. Our results show that it has a potent effect on COX-2 transcriptional activation by PMA. It abrogates completely the COX-2 augmenting effect of p300. These results suggest that HDAC-1 controls COX-2 transcriptional activation. It provides a check and balance for COX-2 expression. Since COX-2 overexpression induces tumor growth, inflammation, and tissue injury, HDAC represents an important endogenous mechanism for maintaining COX-2 at a physiologically relevant level.
In summary, COX-2 expression in response to stimulation by proinflammatory mediators is regulated by p300/PCAF and controlled by HDAC-1. As COX-2 expressions in inflammatory cells such as macrophages and fibroblasts have recently been suggested to play a major role in atherosclerotic plaque instability, our results provide important insight into the mechanisms by which its overexpression occurs and the potential targets for controlling its overexpression. These findings should be valuable for developing transcription-based anti-inflammatory and plaque stability therapy.
Prepublished online as Blood First Edition Paper, November 20, 2003; DOI 10.1182/blood-2003-09-3131.
Supported by grants from the National Heart, Lung, and Blood Institute (R01 HL-50675) and the National Institute of Neurological Diseases and Stroke (P50 NS-23327) of the National Institutes of Health (NIH).
The publication costs of this article were defrayed in part by page charge payment. Therefore, and solely to indicate this fact, this article is hereby marked “advertisement” in accordance with 18 U.S.C. section 1734.
We thank Dr Joan Boyes at the Institute of Cancer Research, London, United Kingdom; Dr Pradip Raychaudhuri at the University of Illinois at Chicago, IL; and Dr I. Talianidis at the Institute of Molecular Biology and Biotechnology in Crete, Greece for providing valuable plasmid constructs. We thank Ms Susan Mitterling for editorial assistance.


This feature is available to Subscribers Only
Sign In or Create an Account Close Modal