Abstract
Studies establishing that intermittent subcutaneous interleukin-2 (IL-2) therapy can lead to substantial CD4 cell increases in many HIV-infected patients have generally been of limited duration. We studied 77 patients participating in active longitudinal studies of subcutaneous IL-2 therapy at our center in order to determine the long-term feasibility of this approach. Following initial induction, patients in each trial were eligible to receive intermittent 5-day cycles of subcutaneous IL-2 treatment at individualized doses and frequencies capable of maintaining CD4 counts at postinduction levels. The mean duration of study participation to date is 5.9 years (range, 1.0-9.3 years). Mean baseline CD4 cell count and CD4 percent values of 0.521 × 109/L (521 cells/μL) and 27% have risen to 1.005 × 109/L (1005 cells/μL) and 38%, respectively, at 90 months. The mean number of subcutaneous IL-2 cycles required to achieve and maintain these increases was 10 cycles (range, 3-29 cycles), and the current mean interval of cycling required to maintain these elevations is 39 months (median, 35 months; range, 2-91 months). We conclude that subcutaneous IL-2 therapy is capable of maintaining CD4 cell increases for an extended period using a remarkably low frequency of intermittent cycling. These observations may contribute to patients' acceptance of subcutaneous IL-2 as a favorable long-term treatment strategy. (Blood. 2004;103:3282-3286)
Introduction
While initially developed and ultimately licensed in the United States for the therapy of specific forms of refractory malignancy, interleukin-2 (IL-2) treatment has also been studied in the context of reversing the immunologic effects of HIV-1 infection.1 Indeed, experimental treatment of HIV-infected individuals with IL-2 in various forms has been under study in this country since the early 1980s even prior to the etiologic identification of AIDS.2 Over the ensuing years, but in particular since the early 1990s, a robust international phase 2 database of intermittent IL-2 treatment experience has accumulated.3-15 This database provides strong evidence that, for many patients receiving concurrent antiretroviral therapy (ART), an unprecedented rise in CD4+ cell counts can occur as a consequence of a scheduled series of intermittent IL-2 treatment cycles. Building on these observations, the international phase 3 SILCAAT (A Phase III Multicenter Randomized Study of the Biological and Clinical Efficacy of Subcutaneous Recombinant, Human Interleukin-2 in HIV-Infected Patients with Low CD4+ Counts Under Active Antiretroviral Therapy) and ESPRIT (A Randomized, Open-Label, Phase III, International Study of Recombinant IL-2 [Proleukin] in Patients with HIV-1 Infection and CD4 Cell Counts Greater than or Equal to 300 Cells/mm: Evaluation of Subcutaneous Proleukin in a Randomized International Trial) trials were launched and are currently exploring whether these CD4 count changes translate into a demonstrable clinical benefit for IL-2 recipients.16,17
While these end point trials are ongoing, an acknowledged limitation of the current database is that most published studies to date have involved fairly limited treatment schedules without extensive long-term follow-up. For this reason most clinicians remain familiar with only the short-term effects of IL-2 therapy and have little direct knowledge of how this agent could potentially be used within the context of a long-term treatment strategy.
In this report we summarize our long-term experience with 3 cohorts of HIV-infected individuals and their longitudinal response to IL-2 therapy. We explore, in particular, whether an “induction-maintenance” model using this cytokine can reasonably be applied to the overall management of these patients.
Patients, materials, and methods
Patients included in this analysis were those who had enrolled in the extension phase of 3 separate trials of subcutaneous IL-2 plus ART. The initial phase of each trial had a distinct study design, dose, and schedule of subcutaneous IL-2 administration, and entry CD4 cell count requirement (Table 1). These trials enrolled patients during a time period spanning 1993 through early 1997.
Demographics of the study population
. | sc IL-24 . | sc 5003 . | msc IL-25 . |
|---|---|---|---|
| Study purpose | Determine maximally tolerated dose of sc IL-2 | Compare sc IL-2 dosing and schedule | sc IL-2 plus ARV vs ARV alone |
| Study design | Dose escalating | Randomized, open label | Randomized, open label |
| Formal study period | 12 mo | 6 mo | 12 mo |
| CD4 cell count requirement, × 109/L | ≥ 0.2 | ≥ 0.5 | 0.2-0.5 |
| Initial enrollment period | September 1993-June 1994 | May 1995-March 1997 | April 1996-March 1997 |
| Percent protease inhibitor use at baseline | 0 | 15 | 77 |
| Definition for inclusion in long-term follow-up | > 18 mo | > 12 mo | > 18 mo |
| Number enrolled in extension phase | 14 | 43 | 20 |
| Age, y, mean | 38 | 36 | 36 |
| Male sex, % | 93 | 100 | 100 |
| Baseline mean CD4 cell count, × 109/L (%) | 0.371 (22) | 0.655 (32) | 0.425 (24) |
| Baseline mean HIV log10 viral load | 4.14 | 3.51 | 3.15 |
| Total number of IL-2 cycles | 212 | 417 | 162 |
. | sc IL-24 . | sc 5003 . | msc IL-25 . |
|---|---|---|---|
| Study purpose | Determine maximally tolerated dose of sc IL-2 | Compare sc IL-2 dosing and schedule | sc IL-2 plus ARV vs ARV alone |
| Study design | Dose escalating | Randomized, open label | Randomized, open label |
| Formal study period | 12 mo | 6 mo | 12 mo |
| CD4 cell count requirement, × 109/L | ≥ 0.2 | ≥ 0.5 | 0.2-0.5 |
| Initial enrollment period | September 1993-June 1994 | May 1995-March 1997 | April 1996-March 1997 |
| Percent protease inhibitor use at baseline | 0 | 15 | 77 |
| Definition for inclusion in long-term follow-up | > 18 mo | > 12 mo | > 18 mo |
| Number enrolled in extension phase | 14 | 43 | 20 |
| Age, y, mean | 38 | 36 | 36 |
| Male sex, % | 93 | 100 | 100 |
| Baseline mean CD4 cell count, × 109/L (%) | 0.371 (22) | 0.655 (32) | 0.425 (24) |
| Baseline mean HIV log10 viral load | 4.14 | 3.51 | 3.15 |
| Total number of IL-2 cycles | 212 | 417 | 162 |
Populations are as follows: sc IL-2, n = 18; sc 500, n = 53; and msc IL-2, n = 26.
The first subcutaneous IL-2 trial (“sc IL-2”) was designed as an initial 12-month, dose-escalating study aimed at determining the maximum tolerated dose for subcutaneous IL-2 in patients with CD4 counts more than or equal to 0.2 × 109/L (200 cells/μL).4 Eighteen patients were enrolled between September 14, 1993, and June 7, 1994. The second subcutaneous IL-2 trial (“sc 500”) was an initial 6-month, randomized, open-label trial comparing the immunologic effects of 2 different doses of subcutaneous IL-2 and a monthly versus bimonthly schedule of administration in a cohort of HIV-infected patients with less-advanced disease.3 A total of 53 patients with CD4 counts more than or equal to 0.5 × 109/L (500 cells/μL) were enrolled between May 17, 1995, and March 7, 1997. The third trial (“msc IL-2”) was an initial 12-month, multicenter, randomized, open-label trial comparing the effectiveness of subcutaneous IL-2 with standard-of-care ART compared with standard-of-care ART alone in patients with CD4 counts between 0.2 and 0.5 × 109/L (200 and 500 cells/μL).5 Patients were enrolled between April 18, 1996, and March 19, 1997.
An additional minimum study participation of 6 months following the initial formal study period was required for inclusion in the long-term (or “extension”) analysis of these 3 trials. In the extension phase the administration of subcutaneous IL-2 was guided through a common algorithm: serial monitoring of the CD4 cell count and an individualization of subcutaneous IL-2 cycle frequency designed to maintain the CD4 cell count at or above a predefined threshold. For all patients this threshold was the achievable postinduction CD4 count, on average approximately twice the baseline count, present at the time of the individual's entry into the extension phase. Patients were encouraged throughout the study to cycle as often as necessary to maintain their CD4 counts in the postinduction range. However, exceptions were granted for those patients whose postinduction CD4 counts extended high into the supraphysiologic range for this parameter; in such cases a minimum CD4 count in the range of 1 to 1.2 × 109/L (1000-1200 cells/μL) was generally adopted as the cycling threshold. In addition, over time those IL-2 recipients requesting lowering of their individualized thresholds by a few hundred cells were generally permitted to do so as long as a substantial increment over their baseline counts was preserved. As before, subcutaneous IL-2 cycles consisted of twice-daily injections for 5 days at the maximally tolerated dose (≤ 7.5 million International Units [MIU] twice daily) for each individual.
For analysis extension, data were normalized to the mutual time point 0 (day one, cycle one of the initial study period). The data set was closed on January 15, 2003. Individualized end of study dates were used for those patients who ended extension phase participation prior to January 15, 2003.
All patients provided written informed consent as approved through the National Institute of Allergy and Infectious Diseases (NIAID) institutional review board.
Immunologic and virologic (bDNA assay version 3.0; Bayer Diagnostics, Tarrytown, NY) assessments were performed as previously described.5 For analytical purposes, all viral loads below the lower limit of detection (< 50 copies/mL) were assigned a value of 49 copies/mL.
Results
Of a total of 97 patients originally enrolled in the 3 trials, 77 patients continued to receive subcutaneous IL-2 treatment during the extension phase of each study. Of the 20 patients who did not continue IL-2 therapy, 2 were enrolled in the original study but subsequently declined participation following randomization, 10 did not complete the formal study period, 7 chose to discontinue participation after the formal study period, and 1 never received any IL-2 treatment (Figure 1).
Patient flow diagram. The course of the 97 patients originally enrolled in 3 separate trials of subcutaneous IL-2 is outlined.
Patient flow diagram. The course of the 97 patients originally enrolled in 3 separate trials of subcutaneous IL-2 is outlined.
There are currently 61 patients on active follow-up. In this cohort, 4 patients were diagnosed with an AIDS-defining condition at some point during their study participation (Table 2) but remain on active follow-up. Of the 16 patients no longer participating in the extension phase, 1 patient died, 1 patient developed non-Hodgkin lymphoma, 8 patients chose to explore other treatment options, and 6 patients experienced CD4+ T-cell count declines that did not readily respond to IL-2 salvage therapy.
AIDS-defining illnesses in the active study population
Study . | Date . | CD4, × 109/L (%) . | Diagnosis . |
|---|---|---|---|
| msc IL-2 | November 1998 | 0.543 (33) | Mild HIV encephalopathy, NARS 1 |
| sc 500 | January 1996 | 1.041 (49) | Cutaneous Kaposi sarcoma with multiple recurrences, first diagnosis with CD4 1082 (44%) |
| sc IL-2 | April 1999 | 0.209 (18) | Cutaneous Kaposi sarcoma |
| sc 500 | December 1999 | 1.565 (46) | Non-Hodgkin lymphoma, right lower quadrant mass |
Study . | Date . | CD4, × 109/L (%) . | Diagnosis . |
|---|---|---|---|
| msc IL-2 | November 1998 | 0.543 (33) | Mild HIV encephalopathy, NARS 1 |
| sc 500 | January 1996 | 1.041 (49) | Cutaneous Kaposi sarcoma with multiple recurrences, first diagnosis with CD4 1082 (44%) |
| sc IL-2 | April 1999 | 0.209 (18) | Cutaneous Kaposi sarcoma |
| sc 500 | December 1999 | 1.565 (46) | Non-Hodgkin lymphoma, right lower quadrant mass |
NARS refers to the Neuropsychiatric AIDS Rating Scale.
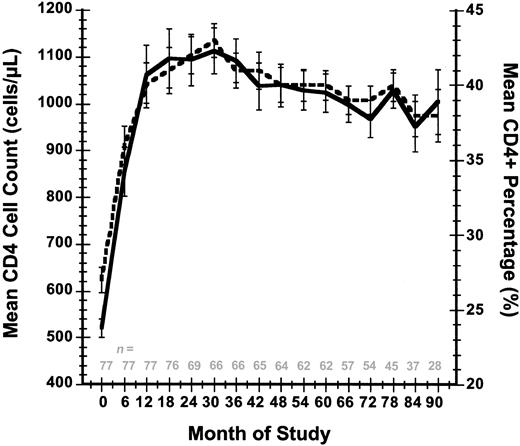
With intermittent IL-2 therapy the CD4 cell counts for these 61 patients have been maintained in the target range for an extended period of time. Relative to mean values for CD4 cell count (0.521 × 109/L [521 cells/μL]) and CD4 percent (27%) at study entry, at month 90 the cohort was still maintaining mean levels of 1.005 × 109/L (1005 cells/μL) and 38%, respectively (Figure 2). Corresponding median and interquartile range (IQR; 25%-75%) values for CD4 cell count and CD4 percent at study entry were 0.521 × 109/L (521 cells/μL) (IQR 0.414-0.606 × 109/L [414-606 cells/μL]) and 27% (IQR 22%-31%), respectively, whereas these same parameters at month 90 were 0.979 × 109/L (979 cells/μL) (IQR 0.717-1.228 × 109/L [717-1228 cells/μL]) and 38% (30%-42%).
Mean CD4+ T-cell count and mean CD4 percent response over time. Standard errors are indicated for these 2 parameters at each time point. The number of evaluable patients at each time point is indicated. Solid line indicates T-cell count; and dashed line, CD4 percent.
Mean CD4+ T-cell count and mean CD4 percent response over time. Standard errors are indicated for these 2 parameters at each time point. The number of evaluable patients at each time point is indicated. Solid line indicates T-cell count; and dashed line, CD4 percent.
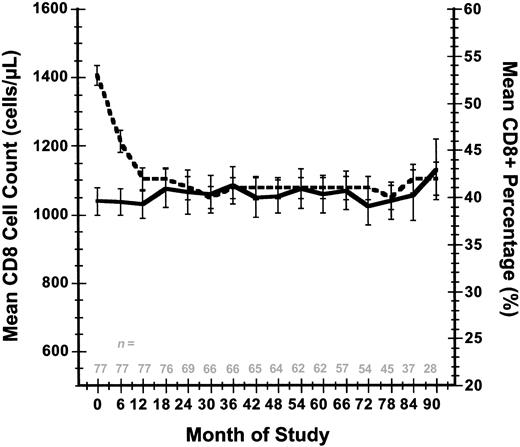
There was a corresponding drop in mean CD8 percent as the mean CD4 percent rose, whereas absolute CD8 counts changed only slightly: mean CD8 percent declined from 53% at study entry to 42% at 90 months, whereas the mean CD8 cell count changed from 1.040 × 109/L (1040 cells/μL) to a level of 1.132 × 109/L (1132 cells/μL) over this same time period (Figure 3). Corresponding median and IQR (25%-75%) values for the CD8 cell count and CD8 percent at study entry were 1.019 × 109/L (1019 cells/μL) (IQR 0.8-1.221 × 109/L [800-1221 cells/μL]) and 53% (IQR 47%-59%), respectively, whereas these same parameters measured at month 90 were 1.003 × 109/L (1003 cells/μL) (IQR 0.814-1.322 × 109/L [814-1322 cells/μL]) and 42% (IQR 39%-48%).
Mean CD8+ T-cell count and mean CD8 percent response over time. Standard errors are indicated for these 2 parameters at each time point. The number of evaluable patients at each time point are indicated. Solid line indicates T-cell count; and dashed line, CD8 percent.
Mean CD8+ T-cell count and mean CD8 percent response over time. Standard errors are indicated for these 2 parameters at each time point. The number of evaluable patients at each time point are indicated. Solid line indicates T-cell count; and dashed line, CD8 percent.
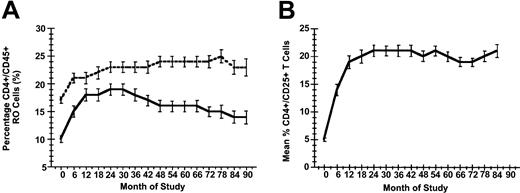
Expression of the alpha chain (CD25) of the IL-2 receptor was induced by IL-2 therapy: mean CD4+/CD25+ percent rose from 5% at study entry to 17% at month 90. Corresponding median and IQR values for CD4+/CD25+ percent expression were 4% (IQR 3%-6%) and 16% (IQR 11%-20%) over this same time period. Similarly, both naive (RO-) and memory (RO+) phenotype expression on CD4+ cells was induced by IL-2: mean RO+ CD45 percent rose from 17% at study entry to 23% at month 90, while mean RO- CD45 percent rose from 10% at study entry to 14% at month 90 (Figure 4). Corresponding median and IQR values for RO+ CD45 percent were 16% (IQR 13%-20%) and 22% (IQR 18%-26%) at study entry and month 90, respectively, whereas RO- CD45 percent expression increased from 9% (IQR 7%-13%) to 13% (IQR 9%-21%) over this same time period.
Changes in CD+ T cell phenotypic expression over time. (A) Mean percentage of naive (CD45 RO-, solid line) and memory (CD45RO+, dashed line) CD4+ cell subsets over time. (B) Mean percentage CD4+/CD25+ T cells over time. In each panel standard errors are indicated for these parameters at each time point.
Changes in CD+ T cell phenotypic expression over time. (A) Mean percentage of naive (CD45 RO-, solid line) and memory (CD45RO+, dashed line) CD4+ cell subsets over time. (B) Mean percentage CD4+/CD25+ T cells over time. In each panel standard errors are indicated for these parameters at each time point.
Viral load did not increase in study participants during the extension period. Using the bDNA version 3.0 assay, the mean HIV log viral load was 3.53 log copies/mL at baseline and it declined to 2.61 log copies/mL at 90 months (Figure 5). The corresponding median and IQR values for viral load at study entry and at month 90 were 3.73 log copies/mL (IQR 3.00-4.33 log copies/mL) and 1.69 log copies/mL (IQR 1.69-3.25 log copies/mL), respectively.
Changes in mean plasma HIV-1 log copies over time. Standard errors are indicated for these 2 parameters at each time point. The number of evaluable patients at each time point is indicated.
Changes in mean plasma HIV-1 log copies over time. Standard errors are indicated for these 2 parameters at each time point. The number of evaluable patients at each time point is indicated.
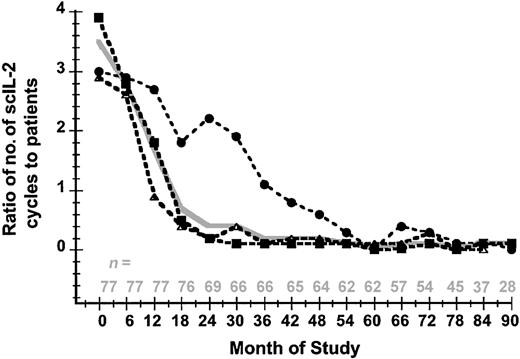
A total of 791 subcutaneous IL-2 cycles (mean, 10; median, 9; range, 3-29 cycles per patient) were administered to 77 patients. The ratio of IL-2 cycles per patient per 6-month interval decreased from 3.5:1 at study entry to 0.1:1 at 90 months (Figure 6). The mean minimal interval since the last cycle was required until closure of the data set was 39 months (median, 35 months; range, 2-91 months).
The mean ratio of IL-2 cycles per patient over time. This is shown in 6-month intervals according to the number of IL-2 cycles for all participants (solid gray line) and for each individual study: sc IL-2 (dashed line, •), sc 500 (dashed line, ▪), and msc IL-2 (dashed line, ▵). The number of evaluable patients at each time point is indicated.
The mean ratio of IL-2 cycles per patient over time. This is shown in 6-month intervals according to the number of IL-2 cycles for all participants (solid gray line) and for each individual study: sc IL-2 (dashed line, •), sc 500 (dashed line, ▪), and msc IL-2 (dashed line, ▵). The number of evaluable patients at each time point is indicated.
In order to evaluate the effect of improved viral suppression through the introduction of highly active antiretroviral therapy (HAART) on CD4+ T-cell counts in our patient population, we also examined CD4 count trends for each patient 12 months before the start of HAART, at the start of HAART, and after 12 months of HAART. Patients in the 3 studies began HAART therapy at a mean of 11 months (median 10 months, range -25-58 months) following initiation of IL-2 therapy. The mean CD4+ T-cell counts 12 months before HAART initiation, at the time of HAART initiation, and 12 months after HAART, were 0.866 × 109/L, 0.903 × 109/L, and 1.004 × 109/L (866 cells/μL, 903 cells/μL, and 1004 cells/μL), respectively.
The potential effects of patient attrition were examined by assigning baseline CD4 values as maximums to all subsequent data points for each patient of the original 97 for whom follow-up data were unavailable. The mean CD4 counts at 48 weeks and at 78 weeks using this method would be 0.867 × 109/L and 0.751 × 109/L (867 cells/μL and 751 cells/μL), respectively, versus values of 1.040 × 109/L and 1.029 × 109/L (1040 cells/μL and 1029 cells/μL) when the analysis is limited to patients participating in the extension phase.
The mean baseline CD4 count for the 61 patients who enrolled in the extension phase was 0.549 × 109/L (549 cells/μL) (27%). The 36 patients who did not enroll in the extension phase or who were lost to follow-up had a mean baseline CD4 count of 0.496 × 109/L (496 cells/μL) (26%). The difference in the mean baseline CD4 count between these 2 groups of patients was not statistically significant (P = .23).
Clinical end points for this patient population were defined as AIDS-defining illnesses or death. One patient with longstanding short-term memory loss was diagnosed with possible mild HIV encephalopathy. Two patients developed cutaneous Kaposi sarcoma and, as mentioned earlier in this section, one patient was diagnosed with non-Hodgkin lymphoma (Table 2). There were 3 patient deaths reported: 2 patients, both IL-2 nonresponders, died of opportunistic infections prior to completion of the initial study phase, and 1 patient on the extension phase died from cardiac disease 2 years after study discontinuation. There were 12 patients enrolled in the extension phase who were diagnosed with either subclinical or mild clinical hypothyroidism; 10 of these patients received synthetic thyroid hormone treatment at some point during their participation.
Discussion
Concerns over the unknown specter of long-term toxicities, costs, and substantial inconvenience associated with combination antiretroviral therapies have continued to promote interest in alternative methods of treating HIV-1 infection.18 Even with the advent of once-daily regimens to facilitate adherence, such considerations as the necessity of a lifelong reliance on these drugs and the ever-present threat of emerging drug resistance have fueled the search for alternative strategies. As primarily a progressive immunodeficiency disease, albeit one with a viral etiology, it has long seemed logical that immunostimulatory therapy should play a larger, more substantive role in the clinical approach to this disorder. Nonetheless, the scope of immune-based treatments being studied for this condition has been comparatively minor in relation to those treatments aimed at direct interference with viral replication. The absence of a broad panoply of suitable candidate agents, our incomplete understanding of the nature of an effective immune response against the virus, the costs and inconveniences associated with largely parenteral therapies, and the absence of well-accepted surrogates for gauging the effectiveness of clinically meaningful immune enhancement remain major obstacles to further development in this field.
Despite these hurdles, at least one immune-based treatment has been under study for a number of years and continues to hold substantial promise either as an adjunct to conventional antiretroviral therapy or potentially even as a stand-alone treatment. As our data show, using the strategy of administering intermittent cycles of recombinant IL-2 at moderate doses to patients receiving antiretroviral medications, it is often possible to elevate the baseline CD4 cell counts in most recipients (although clearly not in all) to levels generally not obtainable through the use of HAART alone. Moreover, this increase occurs in the absence of any adverse effect on plasma virus levels. Increased CD4+/CD25+ expression accompanies these CD4 count increases, and measurement of CD45 isoforms suggests that both phenotypically naive and memory CD4 cell subsets are included in these elevations. As such, these results are wholly consistent with recent in vivo kinetic labeling studies involving either bromodeoxyuridine or deuterium incorporation that suggest that chronic IL-2 administration results in a sustained increase in the survival both of CD25+ naive and central memory CD4 cell subsets.19 While this nonrandomized longitudinal follow-up was not intended to address the overall “functionality” of these CD4 cells, and whereas the subsequent introduction of HAART would confound the interpretation of those results regardless, the notable paucity of significant AIDS-defining events in these long-term IL-2 responders at least provides no evidence of a strong discordance between their CD4 count elevations and the risk of clinical disease progression. Acknowledgment must be made, however, that the high proportion of patients achieving sustained virologic suppression would be expected to lead to a comparatively low rate of disease progression regardless, and that a larger trial involving more advanced patients at higher risk of progression would be required to address this question fully. The results of the SILCAAT trial presently under way may be particularly instructive in this regard.
In this longitudinal follow-up to 3 phase 2 trials of subcutaneous IL-2, we believe we have demonstrated that this agent can be adapted successfully for use according to an induction-maintenance strategy of long-term treatment. Following an initial series of subcutaneous IL-2 cycles to elevate the CD4 count substantially above baseline, a remarkably low frequency of intermittent IL-2 therapy is then required in order to maintain these counts in the physiologic, or even supraphysiologic, range for an extended period of time. At the time the database was closed for this analysis, the mean interval between cycles required to maintain this effect was already in excess of 3 years for that majority of patients continuing in active follow-up. While IL-2 treatment can be associated with transient but significant dose-related side effects even in long-term recipients, a cycling requirement of that low frequency should add considerably to the clinical appeal of this treatment strategy.
While encouraging, these results highlight at least 2 major deficits in our optimal use of IL-2 therapy. To begin with, it remains unclear why a minority of patients with early to moderately advanced disease who undergo aggressive induction with IL-2 treatment do not respond immunologically to this approach despite having otherwise similar baseline CD4 counts and viral loads. Of the 77 patients who originally entered this long-term follow-up study, at least 6 patients experienced no sustained CD4 benefit from subsequent intermittent use of subcutaneous IL-2 therapy. Of the 8 additional patients who eventually chose to pursue other treatment options, it is also possible that personal dissatisfaction with the robustness of the CD4 response may have played some role in their decision to discontinue IL-2 therapy. Thus, the positive findings in the majority of IL-2 recipients who remained in active long-term follow-up may represent some degree of survivor bias in the sense that nonresponders were more likely to terminate their participation earlier. Regardless, it is possible that ongoing in vivo lymphocyte labeling experiments may shed additional insight concerning the kinetics of CD4 cell turnover in this subset of patients and how this parameter may differ from that of IL-2 responders.
Second, if IL-2 induction in the setting of antiretroviral suppression produces CD4 count elevations of this magnitude whose durability is then easily maintained with intermittent cycling, such results beg the obvious question of whether similar increases could either be induced or maintained in the absence of concomitant antiretroviral suppression. That is, could IL-2 therapy be adapted for use as a potential HAART-sparing strategy to obviate or delay the need for initiating antiretroviral treatment in HIV-infected patients? The latter prospects are indeed intriguing, and several pilot trials to address this question are either planned or presently under way.20
Prepublished online as Blood First Edition Paper, January 15, 2004; DOI 10.1182/blood-2003-09-3283.
Supported by an unrestricted grant to C.E.F. from Pfizer Pharmaceuticals Group as administered by the Foundation for the NIH in support of the NIH Clinical Research Training Program.
Two of the authors (J.A.K. and H.C.L.) are listed as coinventors on a patent held by the United States government on the use of interleukin-2 therapy in HIV infection.
The publication costs of this article were defrayed in part by page charge payment. Therefore, and solely to indicate this fact, this article is hereby marked “advertisement” in accordance with 18 U.S.C. section 1734.
The participation of the patient volunteers, the numerous contributions of the Clinic 8 nursing staff, and the ongoing scientific guidance of Dr Anthony S. Fauci, director of NIAID, throughout the performance of these trials are acknowledged with gratitude.



This feature is available to Subscribers Only
Sign In or Create an Account Close Modal