Abstract
Memory B cells are responsible for the rapidly emerging antibody response after antigen reexposure. The signals required for the restimulation of memory B cells have not been fully explained. We used a murine model of anti–factor VIII (FVIII) antibody responses in hemophilia A to study the requirements for the restimulation of FVIII-specific memory B cells and their differentiation into anti-FVIII antibody-producing cells. We were particularly interested in the significance of activated T cells and costimulatory interactions. Our results indicate that the restimulation of FVIII-specific memory B cells is strictly dependent on interactions with activated T cells. These activated T cells can be specific for either FVIII or third-party antigens. Restimulation by T cells specific for third-party antigens requires the presence of FVIII, indicating that signals induced by B-cell receptor (BCR) triggering and by interactions with activated T cells are important. The blockade of B7-1 or B7-2 as well as the blockade of CD40L inhibits the restimulation and differentiation of FVIII-specific memory B cells in vitro and in vivo. The interference with inducible costimulator–inducible costimulator ligand (ICOS-ICOSL) interactions, however, does not cause any modulation. As expected, the production of anti-FVIII antibodies by plasma cells is not dependent on any of the costimulatory interactions tested.
Introduction
The restimulation of memory B cells and their subsequent differentiation into antibody-producing plasma cells is fundamentally important for maintaining antibody-dependent immunologic diseases. Interference with these processes will be the basis for the development of new approaches for treating immune-mediated disorders that involve pathogenic antibodies. Experimental data indicate that interference with costimulatory interactions such as B7-CD28, inducible costimulator–inducible costimulator ligand (ICOS-ICOSL), or CD40-CD40L offers new opportunities for the treatment of patients with immunemediated disorders or after allotransplantation.1-4 Clinical trials using these approaches have been undertaken and have shown at least some success.5-7 Most of the experimental studies in animal models have investigated the prevention of immunologic diseases or of allograft rejection. Little information is available on the potential of these approaches for the treatment of established immune diseases, in particular, diseases that are caused by pathogenic antibodies. Pathogenic antibodies are involved in various immune disorders and are the cause of neutralizing immune responses against therapeutic proteins. An example of such neutralizing immune responses is the development of inhibitory anti–factor VIII (FVIII) antibodies in patients with hemophilia A, a major complication in the replacement therapy with FVIII products.8-12
We used a murine model of hemophilia A to study the importance of activated T cells and of costimulatory interactions such as B7-CD28, ICOS-ICOSL, and CD40-CD40L for the restimulation and differentiation of FVIII-specific memory B cells and for the antibody production by anti-FVIII antibody-secreting plasma cells. Murine hemophilia A in E-17 mice is characterized by complete FVIII deficiency because of a targeted disruption of exon 17 of the FVIII gene.13-15 Intravenous injection of human FVIII into these mice results in high titers of anti-FVIII antibodies.16-19 We and others have previously shown that the blockade of B7-CD28 or CD40-CD40L interactions prevents the development of anti-FVIII antibody responses in these mice.20-22 Furthermore, anti-CD40L antibodies were found to disrupt preexisting germinal centers in the spleen when given after treatment with several doses of FVIII.23 The significance of such interactions in memory-driven antibody production remains, however, unclear. Therefore, we studied the importance of activated T cells and costimulatory interactions for the restimulation and differentiation of FVIII-specific memory B cells.
Materials and methods
Animals
Our colony of fully inbred hemophilic E-17 mice (characterized by a targeted disruption of exon 17 of the FVIII gene) was established with a breeding pair from the original colony13,14 and crossed into the C57BL/6J background as described.15
All mice were male and aged 8 to 10 weeks at the beginning of the experiments. All studies were carried out in accordance with Austrian federal law (Act BG 501/1989) regulating animal experimentation.
Treatment with human FVIII or ovalbumin
Treatment with FVIII. Mice received 4 intravenous doses of 200 ng recombinant FVIII (approximately 80 U/kg FVIII), diluted in 200 μL Dulbecco phosphate-buffered saline (DPBS; Sigma-Aldrich, Irvine, United Kingdom), at weekly intervals. The recombinant human FVIII used throughout the studies was albumin-free bulk material obtained from Baxter BioScience (Thousand Oaks, CA).
Treatment with ovalbumin. Mice were treated with 3 intraperitoneal doses of ovalbumin (Sigma-Aldrich). The first dose was 20 μg, and the second and third doses were both 10 μg. The interval between the first and second injection was 2 weeks and between the second and third injection 1 week. Ovalbumin was diluted in 100 μL DPBS. This treatment regimen was found to give sufficient antibody responses after treatment without adjuvant.
Tissue and blood sampling
Tissue and blood samples were collected 7 days after the last dose of FVIII or ovalbumin if not otherwise indicated. All invasive procedures were carried out under anesthesia with pentobarbital (Nembutal; Richter Pharm, Wels, Austria). Blood samples were obtained by cardiac puncture or tail snipping. The samples obtained from individual mice were added to 0.1 M sodium citrate at a 4:1 (vol/vol) ratio. Plasma was separated by centrifugation and stored at –20° C until further analysis.
Preparation of spleen cells and bone marrow cells
Spleen cells. Spleens were finely minced and passed through a 70-μM nylon cell strainer (Becton Dickinson, Franklin Lakes, NJ). Spleen cells were collected in RPMI 1640 (Life Technologies, Paisley, Scotland) supplemented with 10% preselected fetal calf serum (FCS), 2 mM l-glutamine (both from Hyclone, Logan, UT), 100 U/mL penicillin, 100 mg/mL streptomycin (both from Life Technologies), and 5 × 10–5 M β-mercaptoethanol (Sigma-Aldrich). Single-cell suspensions were cleared of erythrocytes by hemolysis using a hypotonic buffer (pH 7.2) composed of 0.15 M ammonium chloride, 10 mM potassium bicarbonate (both from Merck, Darmstadt, Germany), and 0.1 mM ethylene-diaminetetraacetic acid (Life Technologies).
Bone marrow cells. Bone marrow was extracted from the femur, tibia, and humerus. Cells were passed through a 70-μM nylon cell strainer and collected in RPMI 1640 + 10% preselected FCS as described for spleen cells. Single-cell suspensions were cleared of erythrocytes by hemolysis using a hypotonic buffer as described for spleen cells.
Restimulation of memory B cells in vitro
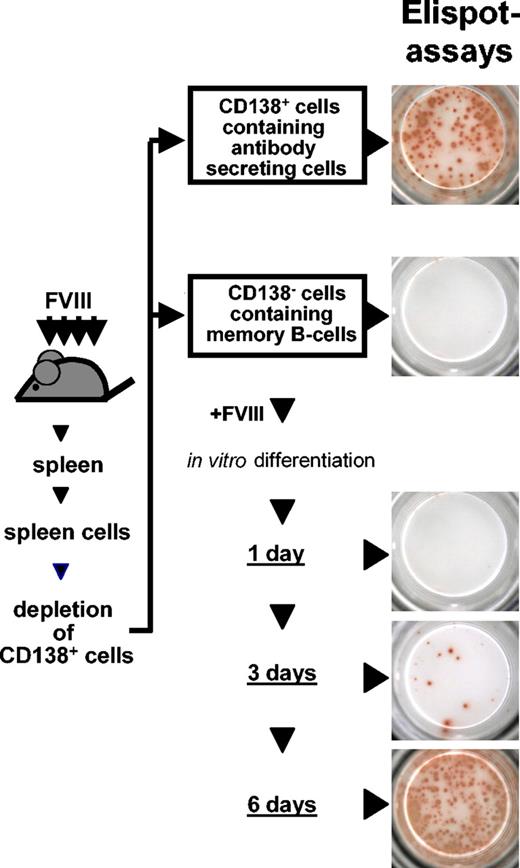
Spleen cells were isolated as described in “Preparation of spleen cells and bone marrow cells,” resuspended in depletion buffer (RPMI 1640 supplemented with 0.1% bovine serum albumin [BSA], 1% glutamine, and 1% sodium pyruvate) and depleted of CD138+ antibody-secreting cells (ASCs). For depletion, cells were incubated with a monoclonal rat antimouse CD138 antibody (PharMingen International, Heidelberg, Germany) coupled to M-450 sheep antirat immunoglobulin G (IgG) Dynabeads (Dynal ASA, Oslo, Norway). After 1 hour of incubation at 4° C, CD138+ cells were depleted by using magnetic devices. CD138– cells, which we called memory cell pool, were cultured at 1.5 × 106 cells/mL in RPMI 1640 (Life Technologies) supplemented with 10% preselected fetal calf serum (Hyclone), 2 mM l-glutamine, 100 U/mL penicillin, 100 mg/mL streptomycin (all from Life Technologies), and 5 × 10–5 M β-mercaptoethanol (both from Sigma-Aldrich) at 37° C for 6 days. Different concentrations of FVIII or ovalbumin were added to the cells on day 0 as indicated. Antibodies and proteins with potential modulating activities or isotype-matched negative control antibodies (hamster IgG for anti-CD40L and anti-CD80, rat IgG2b for anti-CD86, rat IgG2a for anti-ICOSL, human IgG1 for mICOS/Fc and mCTLA4/Fc) were added together with FVIII at day 0. After 3 and 6 days of culture, newly formed ASCs were detected by enzyme-linked immunospot (ELISPOT) assays as described.19
Restimulation of memory B cells in vivo
CD138– memory cell pools were prepared from spleen cells obtained from hemophilic mice treated with 4 doses of FVIII as described in “Restimulation of memory B cells in vitro.” Naive hemophilic mice or hemophilic mice treated with 3 intraperitoneal doses of ovalbumin were each injected intravenously with 107 cells from the memory cell pool resuspended in DPBS. One day after cell transfer, mice were treated with a single intravenous dose of 200 ng FVIII, with 200 μL DPBS (first negative control), with a single intraperitoneal dose of 10 μg ovalbumin or with both FVIII and ovalbumin. For comparison, naive hemophilic mice that did not receive any cell transfer were treated with a single intravenous dose of 200 ng FVIII and served as a second negative control. In some experiments, CD138– memory cell pools were further depleted of T cells (as described in “Depletion of T cells”) before transfer. To evaluate the role of costimulatory signals for the restimulation of FVIII-specific memory cells, recipient mice received a single intravenous dose of blocking monoclonal antibodies or isotype-matched negative control antibodies (hamster IgG for anti-CD40L and anti-CD80, rat IgG2b for anti-CD86, rat IgG2a for anti-ICOSL) 2 hours before the FVIII. A dose of 200 μg antibody or 200 μg competitor protein was given per mouse. Blood samples were taken from each mouse by tail snipping at several time points as indicated.
Antibodies and proteins for the blockade of costimulators
Blocking antibodies against the costimulatory molecules CD80 (B7.1, clone 16-10A1, hamster IgG), CD86 (B7.2, clone P03.1, rat IgG2b), CD40 ligand (CD40L, clone MR1, hamster IgG), and ICOS ligand (ICOSL, clone HK5.3, rat IgG2a) as well as the respective isotype controls were of functional grade and obtained from eBioscience (San Diego, CA). Each antibody was added at 10 μg/mL to the in vitro cell cultures as indicated. Additionally, the importance of ICOS-ICOSL and B7-CD28 interactions were evaluated by using the recombinant competitor proteins murine ICOS/Fc and murine CTLA4/Fc (both are fusion proteins of the murine protein with the Fc-part of human IgG1 and were obtained from R&D Systems, Minneapolis, MN). These proteins were used at a concentration of 10 μg/mL. Murine ICOS/Fc blocks interactions between ICOS and ICOSL, murine CTLA4/Fc blocks interactions between CD80/CD86 and CD28.
All results were normalized for comparing experiments done on different days. The percentage of ASCs compared with the number of ASCs detected in differentiation cultures that were restimulated with FVIII only is presented. Therefore, all results that are smaller than 100% represent an inhibition of memory B-cell differentiation.
Analysis of anti-FVIII ASCs in spleen and bone marrow
Anti-FVIII ASCs in spleen and bone marrow were detected by ELISPOT analysis as described.19
Depletion of T cells
T cells were depleted from CD138– spleen cells using mouse pan-T (Thy 1.2) Dynabeads (Dynal ASA).
Isolation of splenic T cells
Splenic T cells were isolated from either naive hemophilic mice, hemophilic mice treated with 4 intravenous doses of FVIII, or mice treated with 3 intraperitoneal doses of ovalbumin by negative selection using a magnetic cell sorting (MACS) T-cell isolation kit (Milteny Biotec, Auburn, CA).
Cytokine analysis
Murine interleukin 2 (IL-2), interleukin 4 (IL-4), interleukin 5 (IL-5), interleukin 10 (IL-10), interleukin 6 (IL-6), and interferon-γ (IFN-γ) in cell culture supernatants were detected by using a Bio-Plex Mouse Cytokine 6-Plex Assay (Bio-Rad Laboratories, Hercules, CA) specific for the respective cytokines. Samples were measured with a Bio-Plex Protein Array System and analyzed by using the Bio-Plex Manager 2.0 software (all from Bio-Rad Laboratories). All procedures were carried out according to the manufacturer's instructions.
Cell-cell interaction analysis
To find out whether direct interactions between T cells and memory B cells are required for the restimulation of memory B cells, we used tissue culture inserts (Nunc, Roskilde, Denmark) that contain a polycarbonate membrane with a pore size of 0.4 μm. The inserts were put inside a cell culture well. The polycarbonate membrane of the inserts is permeable for soluble proteins such as cytokines but retains cultured cells. We seeded cells of CD138– memory cell pools (described in “Restimulation of memory B cells in vitro”) inside the insert and cells of CD138– memory cell pools depleted of T cells outside the insert in the cell culture well. Both cell populations were adjusted to the same cell concentration (1.5 × 106/mL). We restimulated both culture compartments with FVIII and analyzed newly formed anti-FVIII ASCs after 6 days of culture with use of ELISPOT assays.
Flow-cytometric analysis
The purity of the different cell populations was analyzed by flow cytometry. For this purpose, the following monoclonal antibodies were used: phycoerythrin (PE)–labeled hamster-antimouse CD3e (clone 145-2C11), fluorescein isothiocyanate (FITC–labeled rat-antimouse CD19 (clone 1D3), and PE-labeled rat-antimouse CD138 (Syndecan-1, clone 281-2). To block nonspecific binding sites, cells were incubated with a mixture of antimouse CD16 and antimouse CD32 anti–Fc receptor antibodies (FcBlock) before staining with the monoclonal antibodies mentioned earlier. Appropriate isotype-matched negative control antibodies were included as negative controls for antibody staining. All the antibodies were from PharMingen International. We analyzed 10 000 cells from each sample with use of a fluorescence-activated cell sorter (FACS) Calibur and CellQuest software (Becton Dickinson, Vienna, Austria) after exclusion of cell debris by electronic means.
Detection of anti-FVIII antibodies in blood plasma
Results
Antigen-specific restimulation of FVIII-specific memory B cell in vitro
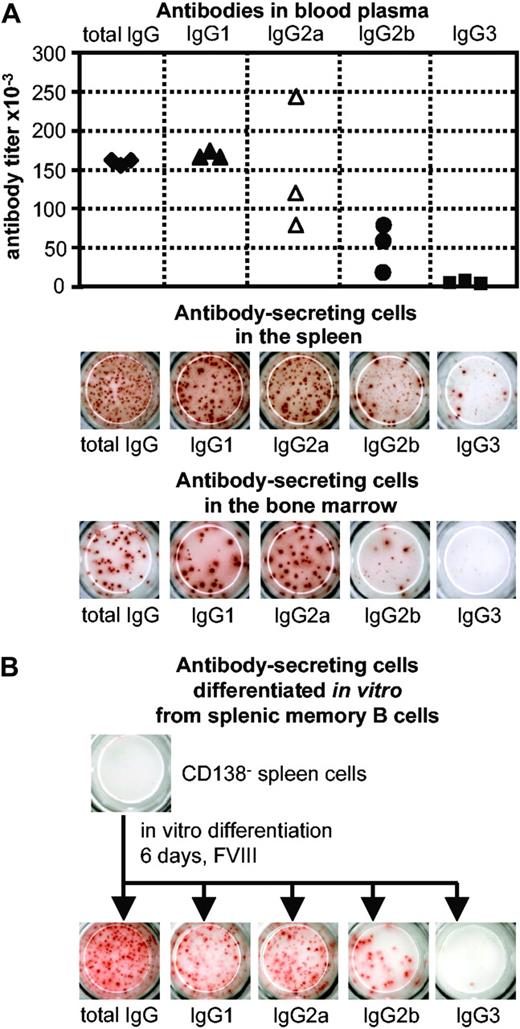
Spleen cells obtained from hemophilic mice treated with FVIII were depleted of anti-FVIII ASCs. This depletion resulted in a cell population that did not contain any anti-FVIII ASCs (Figure 1) but contained FVIII-specific memory B and memory T cells. When this memory cell pool was restimulated with FVIII, newly formed anti-FVIII ASCs could be detected as soon as 3 days after restimulation. The maximum of anti-FVIII ASCs was observed 6 days after restimulation (Figure 1). The IgG subclass distribution of anti-FVIII antibodies produced by in vitro differentiated ASCs was not restricted isotypically and was similar to that of anti-FVIII antibodies found in blood plasma of hemophilic mice treated with FVIII and to that of anti-FVIII antibodies produced by in vivo differentiated anti-FVIII ASCs (Figure 2). When the FVIII-specific memory cell pool was restimulated with ovalbumin, an irrelevant antigen, no formation of anti-FVIII ASCs was observed. Therefore, the restimulation of FVIII-specific memory B cells in vitro was antigen specific.
Restimulation of FVIII-specific memory B cells in vitro. Spleen cells were obtained from hemophilic mice treated with 4 intravenous doses of 200 ng (80 U/kg) FVIII and depleted of CD138+ ASCs. The remaining CD138– cell population was restimulated with 10 ng/mL FVIII and analyzed for newly formed ASCs after 1, 3, and 6 days of culture. ASCs were analyzed by ELISPOT assays as described.19
Restimulation of FVIII-specific memory B cells in vitro. Spleen cells were obtained from hemophilic mice treated with 4 intravenous doses of 200 ng (80 U/kg) FVIII and depleted of CD138+ ASCs. The remaining CD138– cell population was restimulated with 10 ng/mL FVIII and analyzed for newly formed ASCs after 1, 3, and 6 days of culture. ASCs were analyzed by ELISPOT assays as described.19
IgG subclass distribution of anti-FVIII antibodies and anti-FVIII ASCs. (A) The IgG subclass distribution of anti-FVIII antibodies in blood plasma was compared with the IgG subclass distribution of anti-FVIII ASCs in spleen and bone marrow of hemophilic mice treated with 4 intravenous doses of 200 ng (80 U/kg) FVIII. Titers of anti-FVIII antibodies were determined by ELISA (each point represents an individual mouse), and anti-FVIII ASCs were analyzed by ELISPOT assays (spleens and bone marrow of 3 mice were pooled). Blood and tissues were obtained from the same mice. (B) IgG subclass distribution of anti-FVIII ASCs that were differentiated in vitro from CD138– spleen cells. Spleen cells were obtained from hemophilic mice treated with 4 intravenous doses of 200 ng (80 U/kg) FVIII and depleted of CD138+ ASCs. CD138– cells were subsequently restimulated with 10 ng/mL FVIII. Anti-FVIII ASCs were analyzed after 6 days of culture using ELISPOT assays.
IgG subclass distribution of anti-FVIII antibodies and anti-FVIII ASCs. (A) The IgG subclass distribution of anti-FVIII antibodies in blood plasma was compared with the IgG subclass distribution of anti-FVIII ASCs in spleen and bone marrow of hemophilic mice treated with 4 intravenous doses of 200 ng (80 U/kg) FVIII. Titers of anti-FVIII antibodies were determined by ELISA (each point represents an individual mouse), and anti-FVIII ASCs were analyzed by ELISPOT assays (spleens and bone marrow of 3 mice were pooled). Blood and tissues were obtained from the same mice. (B) IgG subclass distribution of anti-FVIII ASCs that were differentiated in vitro from CD138– spleen cells. Spleen cells were obtained from hemophilic mice treated with 4 intravenous doses of 200 ng (80 U/kg) FVIII and depleted of CD138+ ASCs. CD138– cells were subsequently restimulated with 10 ng/mL FVIII. Anti-FVIII ASCs were analyzed after 6 days of culture using ELISPOT assays.
Antigen-specific restimulation of FVIII-specific memory B cell in vivo
The requirements for the differentiation of FVIII-specific memory B cells into anti-FVIII ASCs in vivo was investigated by transferring the memory cell pool that did not contain any anti-FVIII ASCs into naive hemophilic mice. After transfer, mice were treated with a single intravenous dose of FVIII to stimulate the differentiation of memory B cells into anti-FVIII ASCs. Three days after treatment, anti-FVIII antibodies could be detected in blood plasma, indicating the restimulation of FVIII-specific memory B cells. Antibody titers reached a maximum at 7 days after treatment and were detectable for at least 84 days (Figure 3). Animals that did not receive any FVIII treatment after cell transfer or naive mice that did not receive any cell transfer but a single dose of FVIII did not develop anti-FVIII antibodies (Figure 3). This result indicates that FVIII-specific memory B cells did not differentiate into anti-FVIII ASCs without further restimulation by FVIII. Furthermore, only mice that received a transfer of FVIII-specific memory cells developed anti-FVIII antibodies after a single dose of FVIII.
Restimulation of FVIII-specific memory B cells in vivo. CD138– spleen cells were obtained from hemophilic mice treated with 4 intravenous doses of 200 ng (80 U/kg) FVIII and transferred into naive hemophilic mice. Twenty-four hours after transfer, mice were treated with a single dose of either 200 ng FVIII (⋄) or formulation buffer (○). As a negative control, naive hemophilic mice that did not receive any cell transfer were treated with a single intravenous dose of 200 ng FVIII (▵). Blood samples were obtained 1, 3, 7, 14, 42, and 84 days after treatment and analyzed for anti-FVIII antibody titers by ELISA. Each point represents a single mouse. The same mice were analyzed for each time point.
Restimulation of FVIII-specific memory B cells in vivo. CD138– spleen cells were obtained from hemophilic mice treated with 4 intravenous doses of 200 ng (80 U/kg) FVIII and transferred into naive hemophilic mice. Twenty-four hours after transfer, mice were treated with a single dose of either 200 ng FVIII (⋄) or formulation buffer (○). As a negative control, naive hemophilic mice that did not receive any cell transfer were treated with a single intravenous dose of 200 ng FVIII (▵). Blood samples were obtained 1, 3, 7, 14, 42, and 84 days after treatment and analyzed for anti-FVIII antibody titers by ELISA. Each point represents a single mouse. The same mice were analyzed for each time point.
In a control experiment naive mice received a transfer of total spleen cells obtained from hemophilic mice treated with 4 doses of FVIII. After transfer, anti-FVIII antibody titers were detectable in the circulation without any treatment with FVIII for about 8 weeks.
Antigen-specific restimulation and differentiation of FVIII-specific memory B cells depends on interactions with activated T cells that are specific for either FVIII or a third-party antigen
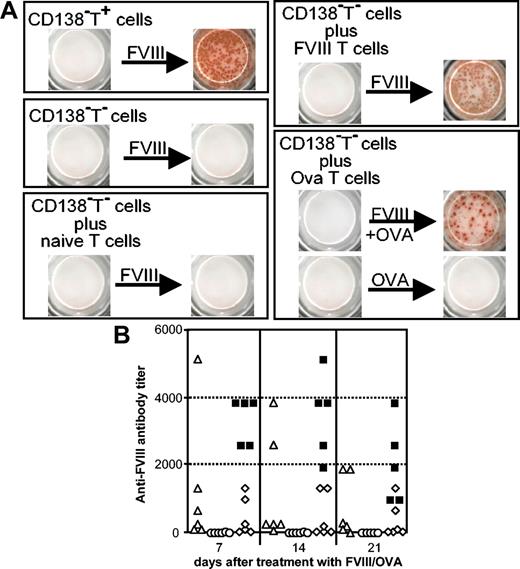
The importance of T cells for the restimulation of FVIII-specific memory B cells was studied by depleting the memory cell pool of T cells before restimulation and comparing the depleted with the nondepleted cell population. Memory cells depleted of T cells did not differentiate into any anti-FVIII ASCs (Figure 4A), indicating that the restimulation of FVIII-specific memory B cells is absolutely dependent on T cells. In another set of experiments, memory cell pools depleted of T cells were reconstituted with T cells from either naive mice, mice treated with FVIII, or mice treated with ovalbumin before restimulation with FVIII. Memory cell pools reconstituted with T cells from mice treated with either FVIII or ovalbumin, but not memory cell pools reconstituted with T cells from naive mice, could be restimulated and differentiated into anti-FVIII ASCs. Ovalbumin alone was not able to restimulate memory cell pools reconstituted with T cells from mice treated with ovalbumin (Figure 4A), indicating that the differentiation of FVIII-specific memory B cells into anti-FVIII ASCs requires both interactions with activated T cells and triggering of the B-cell receptor (BCR) by FVIII.
Restimulation and differentiation of FVIII-specific memory B cells requires FVIII and interaction with activated T cells. Memory cell pool cells (CD138–T+) and memory cell pool cells depleted of T cells (CD138–T–) were obtained from spleen cells of hemophilic mice treated with 4 intravenous doses of 200 ng (80 U/kg) FVIII. (A) CD138–T+ cells, CD138–T– cells, and CD138–T– cells supplemented with T cells from either naive mice (naive T cells), mice treated with 4 intravenous doses of FVIII (FVIII T cells), or mice treated with 3 intraperitoneal doses of ovalbumin (Ova T cells) were cultured in the presence of either FVIII alone, FVIII plus ovalbumin (FVIII + OVA), or ovalbumin alone (OVA). CD138–T– and T cells were always mixed at a ratio of 1:2. Newly formed ASCs were analyzed by using ELISPOT assays after 6 days of culture. (B) Ovalbumin-sensitized mice received memory cell pool cells depleted of T cells and were challenged 1 day after transfer with either ovalbumin together with FVIII (▵) or ovalbumin alone (○). For comparison, naive mice received memory cell pool cells (▪) or memory cell pool cells depleted of T cells (⋄) and were challenged with FVIII 1 day after cell transfer. Blood samples were obtained 7, 14 and 21 days after treatment and analyzed for anti-FVIII antibodies by ELISA. Each point represents a single mouse.
Restimulation and differentiation of FVIII-specific memory B cells requires FVIII and interaction with activated T cells. Memory cell pool cells (CD138–T+) and memory cell pool cells depleted of T cells (CD138–T–) were obtained from spleen cells of hemophilic mice treated with 4 intravenous doses of 200 ng (80 U/kg) FVIII. (A) CD138–T+ cells, CD138–T– cells, and CD138–T– cells supplemented with T cells from either naive mice (naive T cells), mice treated with 4 intravenous doses of FVIII (FVIII T cells), or mice treated with 3 intraperitoneal doses of ovalbumin (Ova T cells) were cultured in the presence of either FVIII alone, FVIII plus ovalbumin (FVIII + OVA), or ovalbumin alone (OVA). CD138–T– and T cells were always mixed at a ratio of 1:2. Newly formed ASCs were analyzed by using ELISPOT assays after 6 days of culture. (B) Ovalbumin-sensitized mice received memory cell pool cells depleted of T cells and were challenged 1 day after transfer with either ovalbumin together with FVIII (▵) or ovalbumin alone (○). For comparison, naive mice received memory cell pool cells (▪) or memory cell pool cells depleted of T cells (⋄) and were challenged with FVIII 1 day after cell transfer. Blood samples were obtained 7, 14 and 21 days after treatment and analyzed for anti-FVIII antibodies by ELISA. Each point represents a single mouse.
We next asked the question whether the in vitro results described earlier could be confirmed in vivo. For this purpose, FVIII-specific memory cell pool cells and, for comparison, FVIII-specific memory cell pool cells depleted of T cells were transferred into either naive mice or mice that had been immunized with ovalbumin. After transfer, mice were treated with a single dose of FVIII, ovalbumin alone, or ovalbumin in combination with FVIII. Naive mice that received memory cell pool cells (not depleted of T cells) showed a fast and long-lasting antibody response to FVIII challenge as quantified by anti-FVIII antibody at days 7, 14, and 21 after challenge (Figure 4B). In contrast, naive mice responded poorly (2 marginal responders, 4 nonresponders) to FVIII challenge when they did receive memory cell pool cells depleted of T cells (Figure 4B). Furthermore, ovalbumin-sensitized mice that received ovalbumin alone after transfer of memory cell pool cells depleted of T cells did not show any significant anti-FVIII antibody response. In contrast, 2 of 6 mice sensitized with ovalbumin that received ovalbumin together with FVIII developed a powerful anti-FVIII antibody response (Figure 4B), suggesting that activated ovalbumin-specific T-memory cells present in ovalbumin-sensitized mice helped the transferred FVIII-specific memory B cells to differentiate into anti-FVIII ASCs.
To study the importance of direct cell-cell interactions between activated T cells and memory B cells, the memory cell pool cells were seeded into culture inserts that contained a membrane that was permeable for proteins and low molecular weight substances but retained cells. These inserts were transferred into culture dishes. Memory cell pool cells depleted of T cells were seeded into the same culture dishes outside the inserts. Both compartments of the culture dish were restimulated with FVIII. After 6 days, newly formed anti-FVIII ASCs were detectable inside the inserts but not outside the inserts. Therefore, the restimulation of FVIII-specific memory B cells requires either direct cell-cell interactions or very close range interactions between activated T cells and memory B cells.
Antigen-specific restimulation of FVIII-specific memory B cells involves CD40-CD40L and B7-CD28 interactions but does not require ICOS-ICOSL interactions
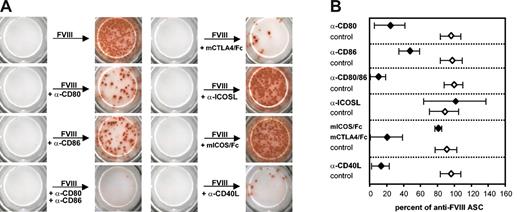
Blocking antibodies against CD40L, CD80 (B7-1), CD86 (B7-2), or ICOSL or recombinant competitor proteins (mICOS/Fc, mCTLA-4/Fc) were added to the memory cell pool cultures immediately before restimulation with FVIII to ascertain the importance of the ligand receptor pairs in restimulating antigen-specific memory cells. After 6 days of culture, newly formed anti-FVIII ASCs were analyzed. The blockade of B7-CD28 or CD40-CD40L interactions significantly inhibited the restimulation of FVIII-specific memory B cells (Figure 5A-B). Both CD80 (B7-1) and CD86 (B7-2) contributed to the required costimulatory interactions with CD28. Blockade of both molecules prevented the restimulation of memory cells almost completely, whereas the blockade of only 1 of the 2 molecules resulted in a partial blockade (Figure 5A-B). In contrast to CD40-CD40L and B7-CD28 interactions, ICOS-ICOSL interactions did not contribute to the restimulation of FVIII-specific memory cells. Neither the addition of a blocking antibody against ICOSL nor the use of a recombinant competitor protein (mICOS/Fc) resulted in a significant alteration in the restimulation of memory B cells (Figure 5A-B). Previous work suggested that the importance of ICOS-ICOSL interactions for the restimulation of memory T cells might depend on the degree of polarization toward either T helper 1 (Th1) or Th2 of these cells.24 In our system memory cell pools obtained from the spleens of hemophilic mice treated with 2, 3, or 4 doses of FVIII (which were expected to show increasing polarization toward Th1 cells) showed a similar lack of dependency on ICOS-ICOSL interaction for effective in vitro restimulation and differentiation into anti-FVIII ASCs. Furthermore, the importance of CD40-CD40L and B7-CD28 interactions did not depend on the number of FVIII doses that were given to the mice.
The antigen-specific restimulation of FVIII-specific memory B cells in vitro involves CD40-CD40L and B7-CD28 interactions but does not require ICOS-ICOSL interactions. Memory cell pool cells (CD138– spleen cells) were obtained from hemophilic mice treated with 4 intravenous doses of 200 ng (80 U/kg) FVIII and restimulated with 10 ng/mL FVIII. Anti-FVIII ASCs were analyzed after 6 days of culture by using ELISPOT assays. To interfere with costimulatory interactions, blocking antibodies (α-CD80, α-CD86, α-ICOSL, α-CD40L as indicated) or competitor proteins (mCTLA-4/Fc, mICOS/Fc as indicated) were added to the cultures at a concentration of 10 μg/mL together with the FVIII. Appropriate isotype-matched control antibodies and human IgG1 were used at the same concentration. (A) ELISPOT data obtained in a representative experiment. (B) Mean values (n = 4) and standard deviations. All results were normalized for comparing experiments done on different days. Results obtained with FVIII only were set as 100%.
The antigen-specific restimulation of FVIII-specific memory B cells in vitro involves CD40-CD40L and B7-CD28 interactions but does not require ICOS-ICOSL interactions. Memory cell pool cells (CD138– spleen cells) were obtained from hemophilic mice treated with 4 intravenous doses of 200 ng (80 U/kg) FVIII and restimulated with 10 ng/mL FVIII. Anti-FVIII ASCs were analyzed after 6 days of culture by using ELISPOT assays. To interfere with costimulatory interactions, blocking antibodies (α-CD80, α-CD86, α-ICOSL, α-CD40L as indicated) or competitor proteins (mCTLA-4/Fc, mICOS/Fc as indicated) were added to the cultures at a concentration of 10 μg/mL together with the FVIII. Appropriate isotype-matched control antibodies and human IgG1 were used at the same concentration. (A) ELISPOT data obtained in a representative experiment. (B) Mean values (n = 4) and standard deviations. All results were normalized for comparing experiments done on different days. Results obtained with FVIII only were set as 100%.
To confirm the importance of CD40-CD40L and B7-CD28 interactions for the restimulation of FVIII-specific memory B cells in vivo, memory cell pools were transferred into naive hemophilic mice. After transfer, mice were treated with blocking antibodies and 2 hours later with a single dose of FVIII. The blockade of either CD40-CD40L or B7-CD28 significantly inhibited the restimulation of FVIII-specific memory B cells in vivo when the effects of blocking antibodies were compared with those of appropriate isotype-matched control antibodies. The blockade of CD40-CD40L interactions was more effective than the blockade of B7-CD28 interactions. However, the blockade of ICOS-ICOSL interactions did not cause any modulation of the restimulation of FVIII-specific memory B cells (Figure 6).
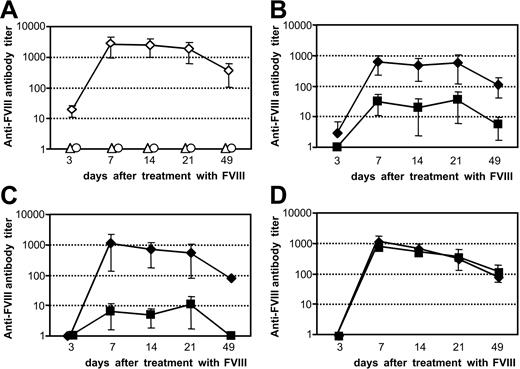
The antigen-specific restimulation of FVIII-specific memory B cells in vivo shows the same dependence on costimulatory interactions as the restimulation in vitro. Memory cell pool cells (CD138– spleen cells) were obtained from hemophilic mice treated with 4 intravenous doses of 200 ng (80 U/kg) FVIII and transferred into naive hemophilic mice. (A) Twenty-four hours after transfer, mice were treated with a single dose of either 200 ng FVIII (⋄) or formulation buffer (○). As a negative control, naive hemophilic mice that did not receive any cell transfer were treated with a single intravenous dose of 200 ng FVIII (▵). (B-D) Twenty-two hours after transfer, mice were treated with intravenous doses of either 100 μg α-CD80 and 100 μg α-CD86 (B; ▪), 200 μg α-CD40L antibody (C; ▪), 200 μg α-ICOSL antibody (D; ▪), or 200 μg appropriate isotype control antibodies (B-D; ♦). Two hours after the application of the antibodies, mice received a single intravenous dose of FVIII. Blood samples were obtained at 3, 7, 14, 21, and 49 days after treatment with FVIII and analyzed for anti-FVIII antibody titers by ELISA. Data shown represent mean values (n=5) and SD.
The antigen-specific restimulation of FVIII-specific memory B cells in vivo shows the same dependence on costimulatory interactions as the restimulation in vitro. Memory cell pool cells (CD138– spleen cells) were obtained from hemophilic mice treated with 4 intravenous doses of 200 ng (80 U/kg) FVIII and transferred into naive hemophilic mice. (A) Twenty-four hours after transfer, mice were treated with a single dose of either 200 ng FVIII (⋄) or formulation buffer (○). As a negative control, naive hemophilic mice that did not receive any cell transfer were treated with a single intravenous dose of 200 ng FVIII (▵). (B-D) Twenty-two hours after transfer, mice were treated with intravenous doses of either 100 μg α-CD80 and 100 μg α-CD86 (B; ▪), 200 μg α-CD40L antibody (C; ▪), 200 μg α-ICOSL antibody (D; ▪), or 200 μg appropriate isotype control antibodies (B-D; ♦). Two hours after the application of the antibodies, mice received a single intravenous dose of FVIII. Blood samples were obtained at 3, 7, 14, 21, and 49 days after treatment with FVIII and analyzed for anti-FVIII antibody titers by ELISA. Data shown represent mean values (n=5) and SD.
Because the restimulation of FVIII-specific memory B cells and their differentiation into anti-FVIII ASCs is dependent on direct or close range interactions with activated T cells, we hypothesized that the inhibition of memory B cell restimulation by the blockade of costimulatory interactions might be because of an inhibition of T-cell activation. To test this hypothesis, patterns of T-cell cytokines were measured in supernatants of memory cell pool cultures 6 days after restimulation with FVIII. The major T-cell cytokines found were IL-10 and IFN-γ (Table 1). The presence of α-CD80 or α-CD86 antibodies reduced the release of IL-10 and IFN-γ significantly, and, when the antibodies were present together, the release of both cytokines was almost completely blocked (Table 1). The prevention of CD40-CD40L interactions significantly reduced the release of T-cell cytokines but did not completely block their release. The blockade of ICOS-ICOSL interactions influenced the release of T-cell cytokines only marginally (Table 1).
Release of T-cell cytokines into supernatants of memory cell pool cultures during 6 days of culture with 10 ng/mL FVIII in the presence or absence of blocking antibodies or appropriate isotype-specific control antibodies (control)
. | Concentrations of cytokines in supernatants, pg/mL . | . | . | . | . | . | |||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Culture conditions . | IL-2 . | IL-4 . | IL-5 . | IL-6 . | IL-10 . | IFN-γ . | |||||
| FVIII only | 12.1 | 3.7 | 10.2 | 13.4 | 636.1 | 358.2 | |||||
| FVIII + α-CD80 | 26.5 | 1.0 | ND | ND | 65.8 | 91.5 | |||||
| FVIII + control | 11.7 | 0.7 | 9.5 | 16.5 | 506.9 | 242.6 | |||||
| FVIII + α-CD86 | 9.5 | ND | ND | ND | 73.9 | 39.8 | |||||
| FVIII + control | 9.3 | ND | 7.8 | 12.8 | 498.7 | 386.5 | |||||
| FVIII + α-CD80/86 | ND | ND | ND | ND | 11.5 | ND | |||||
| FVIII + control | 7.7 | ND | 10.6 | 13.5 | 566.5 | 344.6 | |||||
| FVIII + α-CD40L | 13.2 | ND | 10.4 | 12.5 | 261.8 | 125.2 | |||||
| FVIII + control | 11.7 | 0.7 | 9.5 | 16.5 | 506.9 | 242.6 | |||||
| FVIII + α-ICOSL | 14.7 | 10.2 | 12.4 | 14.5 | 634.1 | 293.7 | |||||
| FVIII + control | 10.8 | 11.9 | 11.6 | 17.8 | 707.5 | 326.5 | |||||
. | Concentrations of cytokines in supernatants, pg/mL . | . | . | . | . | . | |||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Culture conditions . | IL-2 . | IL-4 . | IL-5 . | IL-6 . | IL-10 . | IFN-γ . | |||||
| FVIII only | 12.1 | 3.7 | 10.2 | 13.4 | 636.1 | 358.2 | |||||
| FVIII + α-CD80 | 26.5 | 1.0 | ND | ND | 65.8 | 91.5 | |||||
| FVIII + control | 11.7 | 0.7 | 9.5 | 16.5 | 506.9 | 242.6 | |||||
| FVIII + α-CD86 | 9.5 | ND | ND | ND | 73.9 | 39.8 | |||||
| FVIII + control | 9.3 | ND | 7.8 | 12.8 | 498.7 | 386.5 | |||||
| FVIII + α-CD80/86 | ND | ND | ND | ND | 11.5 | ND | |||||
| FVIII + control | 7.7 | ND | 10.6 | 13.5 | 566.5 | 344.6 | |||||
| FVIII + α-CD40L | 13.2 | ND | 10.4 | 12.5 | 261.8 | 125.2 | |||||
| FVIII + control | 11.7 | 0.7 | 9.5 | 16.5 | 506.9 | 242.6 | |||||
| FVIII + α-ICOSL | 14.7 | 10.2 | 12.4 | 14.5 | 634.1 | 293.7 | |||||
| FVIII + control | 10.8 | 11.9 | 11.6 | 17.8 | 707.5 | 326.5 | |||||
Results were obtained in a representative experiment. ND indicates not detectable (below detection limit).
Production of anti-FVIII antibodies by ASCs does not depend on any of the costimulatory interactions tested
Cells from spleen and bone marrow of hemophilic mice treated with FVIII were isolated and analyzed for the frequency of anti-FVIII ASCs in the presence or absence of blocking antibodies or competitor proteins. Our results demonstrate that the production of anti-FVIII antibodies by ASCs does not depend on any of the costimulatory interactions tested (Figure 7).
Production of anti-FVIII antibodies by ASCs in vitro does not depend on any of the costimulatory interactions tested. Spleen cells and bone marrow cells were obtained from hemophilic mice treated with 4 intravenous doses of 200 ng (80 U/kg) FVIII and analyzed for ASCs by ELISPOT assays. All results were normalized for comparing experiments done on different days. The ELISPOTs above each graph represent the results obtained in a typical experiment. To interfere with B7-CD28, CD40-CD40L, and ICOS-ICOSL interactions, blocking antibodies (α-CD80, α-CD86, α-ICOSL, α-CD40L as indicated) or competitor proteins (mCTLA-4/Fc, mICOS/Fc as indicated) were added at a concentration of 10 μg/mL at the beginning of the cultures. Appropriate isotype-matched control antibodies and human IgG1 were used as controls at the same concentration. Mean values (n = 3) and SDs are presented.
Production of anti-FVIII antibodies by ASCs in vitro does not depend on any of the costimulatory interactions tested. Spleen cells and bone marrow cells were obtained from hemophilic mice treated with 4 intravenous doses of 200 ng (80 U/kg) FVIII and analyzed for ASCs by ELISPOT assays. All results were normalized for comparing experiments done on different days. The ELISPOTs above each graph represent the results obtained in a typical experiment. To interfere with B7-CD28, CD40-CD40L, and ICOS-ICOSL interactions, blocking antibodies (α-CD80, α-CD86, α-ICOSL, α-CD40L as indicated) or competitor proteins (mCTLA-4/Fc, mICOS/Fc as indicated) were added at a concentration of 10 μg/mL at the beginning of the cultures. Appropriate isotype-matched control antibodies and human IgG1 were used as controls at the same concentration. Mean values (n = 3) and SDs are presented.
Discussion
The experiments described in this paper were designed to identify requirements for the restimulation and differentiation of FVIII-specific memory B cells in established anti-FVIII immune responses in hemophilia A. In particular, we were interested in the importance of activated T cells and costimulatory interactions such as B7-CD28, ICOS-ICOSL, or CD40-CD40L. Our results indicate that the restimulation of FVIII-specific memory B cells is strictly dependent on interactions with activated T cells. The appearance of anti-FVIII antibodies in some of the naive mice that were treated with FVIII after transfer of T-cell–depleted memory cell pools is probably because of interactions of FVIII-specific memory B cells with de novo–activated T cells in vivo. The activated T cells do not need to be specific for FVIII, supporting the idea that T cells activated by a third-party antigen can restimulate memory B cells, previously referred to as bystander help.25,26 Activated T cells on their own, however, are not sufficient for the restimulation and differentiation of FVIII-specific memory B cells. Both activated T cells and triggering of the BCR by FVIII are required for this process. These findings seem to contrast with the results published previously by Bernasconi et al,26 who found that human CD27+ memory B cells isolated from peripheral blood could be restimulated with an alloantigen-specific T-cell clone in the absence of BCR triggering. The differences between these results and our results might be because of differences in the strength of the signal provided by bystander T cells and differences in the frequency of activated T cells present in the experimental systems. Therefore, we cannot exclude that a very strong signal from activated T cells, which are present at a high frequency, would overcome the requirement for BCR triggering in our system. We are currently addressing this question in further experiments.
The B7-1/B7-2-CD28/CTLA-4 pathway is the best characterized costimulatory pathway for T-cell activation and is also essential for T-cell tolerance (reviewed in Sharpe and Freeman3 and Rudd and Schneider27 ). Qian et al20 were able to show that B7-2, but not B7-1, was involved in the primary immune response against FVIII in hemophilic mice. Furthermore, the injection of murine CTLA-4-Ig into hemophilic mice prevented a further increase in anti-FVIII antibody titers in hemophilic mice with an established anti-FVIII immune response, indicating that CTLA-4-Ig blocks the restimulation of FVIII-specific memory B cells. In the present study we could confirm these results and show that both B7-1 and B7-2 are required for the restimulation of FVIII-specific memory B cells. The release of T-cell cytokines into the supernatants of in vitro cell cultures was completely inhibited in the presence of blocking antibodies against both B7-1 and B7-2, indicating that the inhibition of memory B-cell restimulation was because of an inhibition of T-cell activation. Comparing our results with those published by Qian et al,20 we conclude that B7-2, but not B7-1, is involved in primary anti-FVIII antibody responses in hemophilic mice and that both molecules are important for the memory-driven antibody response.
ICOS (inducible T-cell costimulator) is a new member of the CD28 family (reviewed in Sharpe and Freeman3 and Rudd and Schneider27).28,29 Previous reports indicated that ICOS-ICOSL interactions are important for the development of T-cell–dependent antibody responses.30-34 We did not, however, see any dependence of the restimulation of FVIII-specific memory B cells on ICOS-ICOSL interactions. There are several possible explanations for the discrepancy between our results and previous reports. McAdam et al30 showed that the disturbed class switching in ICOS–/– mice was restored by stimulation of CD40 using a stimulating monoclonal antibody. This suggests that ICOS-ICOSL interactions are important for the up-regulation of CD40L on activated T cells, which is essential for the costimulatory interaction with B cells. Therefore, it is imaginable that other signals, which induce the up-regulation of CD40L, can substitute the requirement for ICOS-ICOSL interactions. McAdam et al30 showed that not only the stimulation of ICOS but also the stimulation of CD28 induced the expression of CD40L on murine T cells that were activated by triggering the T-cell receptor (TCR). We believe that in our experimental system the expression of CD40L was mediated by the triggering of CD28 because we could clearly show that the restimulation of FVIII-specific T cells was dependent on B7-CD28 interactions. The nature of the antigen and the dose of the antigen used for the induction and restimulation of immune responses probably influence the requirements for certain costimulatory signals. We used human FVIII at concentrations of 10 ng/mL and 100 ng/mL for the in vitro experiments. These concentrations correspond to 10% and 100% of the normal concentrations of FVIII found in blood plasma and, therefore, are physiologically relevant. For in vivo experiments we used doses of 200 ng FVIII without adjuvants for intravenous application. In previous reports different antigens at much higher concentrations were used.30-34
CD40-CD40L interactions are a key event in T-cell–dependent humoral immune responses (reviewed in Foy et al1 ). The results from studies on the significance of these interactions for the differentiation of memory B cells into ASCs, however, conflict. Several reports suggest that CD40 signaling is important for the terminal differentiation of B cells and for antibody secretion.35-38 Other reports show that CD40 signaling prevents the terminal differentiation of B cells.39-43 Our results indicate that the restimulation of FVIII-specific memory B cells and their subsequent differentiation into anti-FVIII ASCs requires CD40-CD40L interaction. The blockade of these interactions prevented the formation of anti-FVIII ASCs in vitro and reduced it significantly in vivo. Furthermore, the blockade of CD40-CD40L interactions reduced the release of T-cell cytokines into culture supernatants but did not completely block it. Therefore, we believe that the blockade of CD40-CD40L interactions in our system down-regulates T-cell activation and, more importantly, blocks the interaction between activated T cells and memory B cells.
One of the reasons for the conflict between the results from different studies might be the difference in the experimental systems that were used for the studies. Erickson et al44 have recently shown that the magnitude of CD40 signaling dramatically influences the outcome of an immune response in vivo. Furthermore, Pype et al45 have identified a novel intracellular CD40-binding protein termed TTRAP (TRAF [tumor necrosis factor receptor-associated factor] and TNF [tumor necrosis factor] receptor-associated protein) that associates with CD40 in response to treatment of cells with CD40L and inhibits the activation of nuclear factor-κB (NF-κB), thereby counteracting signal transduction. The strength of CD40 triggering that is determined by the experimental setup might, therefore, dramatically influence the outcome of any experiment. Both the lack of CD40 signaling and a very strong signal mediated by way of CD40 triggering might block the differentiation of memory B cells into ASCs. Furthermore, Lee et al46 have suggested that the duration of CD40 signaling during B-cell differentiation might be important for the differentiation into ASC.
Two different pathways for the restimulation and differentiation of memory B cells have been described, namely an antigen-dependent pathway and an antigen-independent pathway by ways of Toll-like receptors.26,47 We investigated the antigen-dependent pathway and showed that the restimulation and differentiation of FVIII-specific memory B cells in hemophilic mice requires interactions with activated T cells and costimulatory interactions such as B7-CD28 and CD40-CD40L. An intervention with these costimulatory interactions might prevent the restimulation of FVIII-specific memory B cells and thereby prevent or reduce any additive effects resulting from additional antigenic challenge in patients with hemophilia A. However, our results also show that the secretion of anti-FVIII antibodies by ASCs is not modulated by these costimulatory interactions. Therefore, we believe therapeutic approaches that interfere with B7-CD28 or CD40-CD40L interactions will not suffice for the treatment of patients with established immunemediated disorders that involve pathogenic antibodies.
Prepublished online as Blood First Edition Paper, March 4, 2004; DOI 10.1182/blood-2003-07-2456.
All authors except C.H. are employed by Baxter Healthcare Corporation. The work described in this study might eventually contribute to the development of a potential product by the corporation.
The publication costs of this article were defrayed in part by page charge payment. Therefore, and solely to indicate this fact, this article is hereby marked “advertisement” in accordance with 18 U.S.C. section 1734.
We thank Elisabeth Hopfner and Monika Grewal for technical assistance, Howard M. Reisner for his critical review, and Elise Langdon-Neuner for editing the manuscript.



This feature is available to Subscribers Only
Sign In or Create an Account Close Modal