Abstract
Immunosuppressive agents in current use are nonspecific. The capacity to delete specific CD8 T-cell clones of unique specificity could prove to be a powerful tool for dissecting the precise role of CD8+ T cells in human disease and could form the basis for a safe, highly selective therapy of autoimmune disorders. Major histocompatibility complex (MHC) tetramers (multimeric complexes capable of binding to specific CD8 T-cell clones) were conjugated to 225Ac (an alpha-emitting atomic nanogenerator, capable of single-hit killing from the cell surface) to create an agent for CD8 T-cell clonal deletion. The “suicide” tetramers specifically bound to, killed, and reduced the function of their cognate CD8 T cells (either human anti–Epstein-Barr virus (EBV) or mouse anti-Listeria in 2 model systems) while leaving the nonspecific control CD8 T-cell populations unharmed. Such an approach may allow a pathway to selective ablation of pathogenic T-cell clones ex vivo or in vivo without disturbing general immune function.
Introduction
Immune recognition by CD8+ T cells is determined by binding of αβ T-cell receptors (TCRs) to target cell antigen-derived peptides displayed in the target's major histocompatibility complex (MHC) class I molecule.1-4 These antigenic peptides can be nonnative peptide fragments derived from foreign viral or bacterial proteins, or derived from normal or mutated self proteins.3,5,6 Soluble tetrameric forms of peptide–MHC class I complexes can bind stably to the TCRs on a given specific CD8 T-cell clone.7-9 Such specific tetramers, which are fluorescently tagged, have been used to identify or isolate antigen-specific T cells from peripheral blood and other tissues.10,11
High linear energy transfer (LET) alpha-particle emitters are of unique interest as cytotoxic agents because they need not be internalized to kill cells and are potent enough to selectively kill individual cells within a short range with a single decay. Bi-213 and At-211 alpha-emitting antibody constructs are in human cancer trials.12 Actinum-225 (225Ac) is an alpha-emitting atomic nanogenerator that yields 4 net alpha particle emissions.13-15 Monoclonal antibodies were conjugated to 225Ac for cancer therapy in animal models and have shown that small doses (nCi) of 225Ac-antibody are capable of eradicating tumors without significant toxicity.15 The characteristics of the alpha generators suggest that they also would be useful in arming tetramers to selectively kill their cognate T-cell clones.
In this report, we describe new techniques for making and using armed alpha-emitting tetramers capable of targeting specific peptide–MHC class I–restricted CD8+ T-cell clones. We used well-characterized CD8+ T-cell lines of human or murine origin to demonstrate that small doses of 225Ac-radiolabeled “suicide” tetramers were capable of killing targeted CD8+ T cells while leaving the nonspecific control CD8+ T-cell populations unharmed. This is the first description of a method to kill specific CD8+ T-cell clones.
Materials and methods
Human and mouse CD8+ T-cell lines
Human Epstein-Barr virus (EBV) or influenza virus peptide-specific CD8+ T-cell lines, and the mouse Listeria monocytogenes–specific CD8+ T-cell lines chosen for this study were established as described previously.16-18 The cell lines specific for the human EBV latency membrane protein 1 peptide (LMP1; YLLEMLWRL) and the influenza peptide (Flu58-66; GILGFVFTL) were generated from the HLA-A2 healthy donors. In brief, for the purpose of conducting the assays, T cells were isolated from peripheral blood by using Ficoll-Hypaque gradient separation of mononuclear cells followed by depletion of CD20+ B cells, CD14+ monocytes, and CD56+ natural killer (NK) cells with monoclonal antibody (mAb)–coated immunomagnetic beads (Miltenyl Biotec, Auburn, CA). The aliquots of lymphocyte population were stimulated in vitro by exposure to either irradiated autologous LMP1 or Flu58-66 peptide–loaded, EBV-transformed B cells and cultured in special lymphocyte medium (AIM-V medium; Gibco, Carlsbad, CA) containing 100 IU/mL interleukin 2 (IL-2; BD Biosciences, San Jose, CA). Cells were stimulated weekly and the enriched T-cell cultures were subsequently tested by anti-CD8 mAb and LMP1 tetramer or Flu58-66 tetramer flow cytometry for binding specificity. CD8 T-cell lines positive for tetramer binding were further stimulated and aliquots of the enriched human peptide–specific CD8 cells (> 90% pure) were used in this study.
Murine LLO91-99- or p60217-225-specific CD8+ T-cell lines were established from BALB/c splenocytes 1 week after second immunization with a sublethal dose of Listeria.19-22 The LLO91-99 (GYKDGNEYI) or p60217-225 (KYGVSVQDI) peptide-specific CD8+ T cells were maintained in RPMI medium containing 0.16 μg/mL IL-7 (BD Biosciences) and 0.5 ng/mL IL-2 at 37° C in 5% CO2. Tetramer binding specificity of the CD8+ T cells was reconfirmed by flow cytometry before each experiment. Tetramer binding to CD8+ cell lines was stable over several weeks when cells were maintained in culture with periodic exposure to peptide-pulsed antigen-presenting cells and fresh cytokines. This allowed us to utilize the same mouse cell line repeatedly for study.
Peptide–MHC class I tetramer constructs
Tetramers were prepared as previously described18,23 and provided by the Memorial Sloan-Kettering Cancer Center (MSKCC) Tetramer Core Facility. Briefly, recombinant HLA-A2 or H-2Kd and human β2 microglobulin produced in Escherichia coli were solubilized in urea and reacted with synthetic peptide antigens in a guanidine refolding buffer. The peptides used in this study were synthesized by ResGen (Huntsville, AL) and were more than 90% pure. Refolded peptide–MHC I complexes were purified and then biotinylated. Tetrameric peptide–MHC I complexes were subsequently produced by the stepwise addition of streptavidin-conjugated phycoerythrin (PE) to achieve a 1:4 molar ratio.
Flow cytometry
The binding specificity of tetramers was analyzed by flow cytometry using the human and mouse antigen-specific CD8 T-cell lines. Human LMP1- or Flu58-66-specific CD8 T cells (1 × 105 in 100 μL) were stained with fluorescein isothiocyanate (FITC)–anti-CD8 antibody (BD Biosciences) and PE-labeled cognate peptide/HLA A2 tetramers at different concentrations at 4° C for 60 minutes. In addition, binding of Flu58-66 tetramers to LMP1 CD8 T cells and LMP1 tetramers to Flu58-66-specific CD8 T cells was employed to confirm the specificity of the tetramer staining. Flow cytometry using LLO91-99/H-2Kd or p60217-225/H-2Kd tetramers on mouse CD8 T-cell lines was also conducted using identical conditions. To determine the stability of tetramer binding to TCRs, CD8 T-cell lines were incubated with tetramers at 37° C for 1, 2, 3, 8, and 24 hours in phosphate-buffered saline (PBS) or in 100% serum, and tetramer binding was subsequently quantified by flow cytometry. Fractions of multimeric constructs were collected separately by size exclusion chromatography and binding of dimers, trimers, and tetrameric MHC constructs to their cognate CD8 T cells was determined by flow cytometry.
Preparation of 225Ac-DOTA-biotin and 111In-DTPA-biotin
Radionuclide 111In was purchased from PerkinElmer Life Sciences (Billerica, MA) and 225Ac was obtained from the Oak Ridge National Laboratory (Oak Ridge, TN). The 225Ac nitrate residue was dissolved in 0.2 M Optima grade HCl (Fisher Scientific, Pittsburgh, PA) and biotinylated 1,4,7,10-tetraazacyclodododecane-1,4,7,19-tetraacetic acid (biotin-DOTA) was prepared by the Organic Core Facility at MSKCC (W.B., June 2003, unpublished). Biotin-DOTA or biotinylated diethyenetriaminepentaacetic acid α, w-bis (DTPA; Sigma, St Louis, MO) was dissolved in metal-free water to yield a 10 to 20 mg/mL solution. The same procedure was used to label either biotin-DOTA (1 mg) with 1 mCi (37 MBq) of 225Ac or biotin-DTPA (1 mg) with 2 mCi (74 MBq) 111In. In brief, 5 μL to 20 μL 225Ac dissolved in 0.2 M HCl was added to a NUNC 1.8-mL reaction tube (Fisher Scientific). One milligram of biotin-DOTA solution (100 μL) was added along with 100 μL of 0.2 M HCl, 50 μL of 2 M tetramethylammonium acetate, and 15 μL of 150 g/L I-ascorbic acid (Aldrich Chemical, Milwaukee, WI). The mixture (pH = 4.5-5.0) was then heated to 60° C for 30 minutes and the reaction was terminated by adding 20 μL of 0.10 M EDTA (ethylenediaminetetraacetic acid; Aldrich Chemical). The 111In-DTPA-biotin mixture was prepared without the heating step before termination.
To quantify incorporation of 225Ac or 111In radionuclide, 1 mL Sepadex C-25 resin (Aldrich Chemical) in 0.9% NaCl was packed into a column. A 2-μL aliquot of the radioactive reaction mixture was applied and the column was eluted with 3 mL of 0.9% NaCl. The column was eluted a second time to determine if all radioactivity had been removed. The column and washes were either counted immediately using a Squibb CRC-17 Radioisotope Calibrator (ER Squibb and Sons, Princeton, NJ) to measure 111In activity or counted 20 hours later to determine 225Ac activity levels. The activity contained in the eluate was considered to be the percent 111In or percent 225Ac that was complexed to the chelant moiety.
Biotin reactivity in the radiolabeled component was assayed after application of the radioactive reaction mixture to an immobilized avidin column (Pierce, Rockford, IL). The column was washed twice with 5 mL of 0.9% NaCl to remove unbound material and the column and washes were counted to determine the 111In or 225Ac activity using the same method previously described in the previous paragraph. The percent activity bound to the column was considered to be the percent 111In or percent 225Ac that contained biotin-avidin binding reactivity.
Radiolabeled tetramer constructs
111In has a relatively short half-life (∼ 3 days), and it was selected for use in our early experiments to establish the optimal conditions for tetramer labeling. The freshly prepared 111In-DTPA-biotin products were mixed with freshly prepared biotinylated monomers in the presence of streptavidin at a ratio of 1:3:1 in order to construct radiolabeled tetramers. The product was further purified by size exclusion chromatography using a 10-mL Econo-Pac 10DG column (BioRad, Hercules, CA) with a PBS mobile phase. Both radiolabeled-specific and non-specific tetramers were prepared in this fashion for our in vitro studies. In addition, non-radiolabeled tetramers used for controls or blocking experiments were similarly prepared.
Specific binding and internalization of radiolabeled tetramers
Different dilutions of radiolabeled tetramers in 5 to 8 μL PBS were added to 1 × 107 CD8+ T cells on ice. To determine the influence of incubation temperature on radiolabeled tetramer binding or tetramer internalization, cells were incubated with either radiolabeled tetramers on ice or at 37° C for 1 hour and 4 hours and overnight. The cells were then centrifuged, washed twice with 1 mL ice-cold PBS, and subjected to counting to determine the amount of specific 111In tetramer binding. To quantify internalization of radiolabeled tetramers, the cell-surface–bound radiolabeled tetramers were stripped from the cell pellet by exposure to 1 mL of 50 mM glycine/150 mM NaCl (pH 2.8) for 10 to 15 minutes at room temperature.24 The quantity of surface-bound and internalized radioactivity was determined by counting the samples separately. Both radiolabeled nonspecific tetramers and CD8 T-cell lines bearing TCRs of different peptide specificities served as controls for this assay. All assays were performed in duplicate.
Specific cell killing by 225Ac-labeled tetramers
The killing efficacy of suicide tetramers was quantified using 1 × 106 LMP1-orLLO91-99-specific CD8+ T cells in 96-well plates. Flu58-66-specific or p60217-225-specific CD8+ T cells served as negative controls. Serial dilutions of 225Ac-tetramers were added to the CD8+ T cells and nonspecific cell killing was determined by adding only 225Ac-DOTA or only nonradiolabeled tetramers. To confirm specificity of cell killing, some cells were first incubated with a 50-fold excess of nonradiolabeled specific tetramers for 30 minutes before addition of suicide tetramers. The cells were incubated at 37° C in 5% CO2 for 72 hours and cell viability was subsequently determined by [3H]thymidine incorporation and trypan blue staining. Each assay was performed in triplicate.
In order to further demonstrate that 225Ac-suicide tetramer killing was T-cell specific, the 225Ac-LLO91-99 tetramers were added to a mixed cell culture of LLO91-99-specific CD8+ T cells and LLO tetramer–negative, p60217-225-specific CD8+ T cells. Serial dilutions of suicide 225Ac-LLO91-99 tetramers were added to the cell mixture (50:50) containing LLO91-99 CD8+ cells and p60217-225-specific CD8+ cells. Cell viability was subsequently determined by both trypan blue staining and [3H]thymidine incorporation after incubation at 37° C in 5% CO2 for 72 hours. The remaining viable CD8+ T cells were washed and then restudied by tetramer flow cytometry to define their specificities. Each assay was performed in triplicate.
IFN-γ ELISPOT assay
The interferon γ (IFN-γ) enzyme-linked immunospot (ELISPOT) assay was performed in nitrocellulose-lined 96-well microplates (Millipore MAHA S45) using an IFN-γ ELISPOT kit.25 Plates were coated overnight with antibody to murine IFN-γ and washed 6 times. The LLO91-99 CD8 T cells (1 × 106/mL) or control p60217-225 CD8 T cells (1 × 106/mL) were incubated at 37° C for 72 hours with 225Ac-LLO91-99 tetramers at 5 nCi/mL (1 μg/mL). T cells were then washed and cultured (105/well) with irradiated antigen-presenting cells (APCs; P815 cells), cognate peptides (50 μg/mL), and incubated for 20 hours at 37° C. Wells containing CD8+ T cells, APC cells, and nonspecific control peptide served as negative controls. The spots were counted using a stereomicroscope at a 40-fold magnification and an automated Elispot reader system (Carl Zeiss Vision, Jena Goettingen, Germany) with KS Elispot 4.0 software. The final number of specific IFN-γ spots was obtained after subtracting the number of nonspecific IFN-γ spots produced in the control wells. All assays were performed in duplicate.26,27
Statistics
Statistical analyses were performed using one-way analysis of variance (ANOVA) on GraphPad Instat 3.0 (GraphPad Software, San Diego, CA).
Results
Peptide-specific tetramers bind CD8 T-cell lines with high specificity
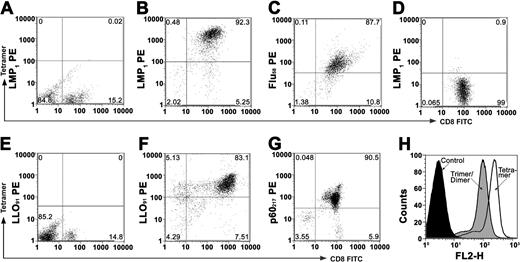
Human and mouse peptide-specific CD8 T-cell lines were established in order to assess specific cell-killing efficacy of our radiolabeled tetramers. The binding specificity of individual nonradiolabeled tetramers was first analyzed by flow cytometry. Only 0.02% of the EBV-negative healthy human peripheral mononuclear cells reacted with the LMP1 tetramers (Figure 1A). More than 90% of enriched human LMP1-specific T cells were positive for both anti-CD8 mAb and LMP1/HLA-A2 tetramer staining (Figure 1B). Similarly, 87% of Flu58-66-specific T cells positively reacted with their cognate Flu58-66/HLA-A2 tetramers and none of these CD8 cells reacted with LMP1/HLA-A2 tetramers (Figure 1C-D). Consistent to previous report, none of the spleen CD8 T cells from normal BALB/c mouse reacted with LLO91-99 tetramers22,23 (Figure 1E). Experiments performed using mouse CD8+ T-cell lines (Figure 1F-H) specific for L monocytogenes peptides showed that more than 80% of the enriched mouse spleen LLO91-99 T cells bound to both anti-CD8 mAb and LLO91-99 tetramers. Similarly, more than 90% of mouse p60217 T cells were double-positive for anti-CD8 mAb and p60217/H2-Kd tetramer staining. Taken together, the tetramer constructs assembled from peptide–MHC class I monomers were highly specific for their cognate antigen-specific CD8+ T cells.
Tetramer staining of human and mouse CD8+ T-cell lines. CD8 T-cell lines were incubated with anti-CD8 mAb and peptide–MHC class I–specific tetramers. The percentage of tetramers and CD8 double-positive T cells is shown in the upper right quadrant of each plot. (A) The panel shows that 0.02% of EBV-negative human peripheral blood mononuclear cells (PBMCs) tested positive for LMP1/HLA-A2 tetramers. (B) The panel shows that 92% of LMP1 peptide–specific CD8 T cells bound to LMP1 tetramers. (C) The panel shows that 87% of Flu-specific CD8 T cells bound to Flu58-66/HLA-A2 tetramers. (D) Less than 1% of the human Flu-specific CD8+ T cells tested positive for the LMP1/HLA-A2 tetramers. (E) The panel shows that 0% normal mouse spleen CD8 T cells bound the LLO91-99/H-2Kd tetramers. (F) More than 80% of the mouse splenic CD8 T-cell line bound LLO91-99/H-2Kd tetramers. (G) The panel shows that 90% of the p60217-225-specific T-cell line tested positive for p60217-225 tetramer staining. (H) Only mildly decreased binding to their cognate CD8 T cells by dimeric/trimeric compared with trimeric/tetrameric MHC constructs was observed.
Tetramer staining of human and mouse CD8+ T-cell lines. CD8 T-cell lines were incubated with anti-CD8 mAb and peptide–MHC class I–specific tetramers. The percentage of tetramers and CD8 double-positive T cells is shown in the upper right quadrant of each plot. (A) The panel shows that 0.02% of EBV-negative human peripheral blood mononuclear cells (PBMCs) tested positive for LMP1/HLA-A2 tetramers. (B) The panel shows that 92% of LMP1 peptide–specific CD8 T cells bound to LMP1 tetramers. (C) The panel shows that 87% of Flu-specific CD8 T cells bound to Flu58-66/HLA-A2 tetramers. (D) Less than 1% of the human Flu-specific CD8+ T cells tested positive for the LMP1/HLA-A2 tetramers. (E) The panel shows that 0% normal mouse spleen CD8 T cells bound the LLO91-99/H-2Kd tetramers. (F) More than 80% of the mouse splenic CD8 T-cell line bound LLO91-99/H-2Kd tetramers. (G) The panel shows that 90% of the p60217-225-specific T-cell line tested positive for p60217-225 tetramer staining. (H) Only mildly decreased binding to their cognate CD8 T cells by dimeric/trimeric compared with trimeric/tetrameric MHC constructs was observed.
The tetramer-specific binding to cognate CD8 T cells incubated in PBS was stable for 24 hours at 37° C, whereas in the presence of 100% mouse or human serum tetramer binding stability was maintained up to 8 hours and then rapidly declined with longer incubation (Table 1). A comparison of dimeric/trimeric and tetrameric MHC constructs binding to their cognate CD8 T cells (Figure 1H) showed that trimeric MHC constructs were closely reactive as tetrameric constructs. Therefore, we hypothesized that the trimeric forms could be rendered potently cytotoxic by conjugation to alpha-emitting isotopes through the free biotin binding site on the tetrameric avidin core.
Stability of tetramer binding to CD8 T-cell receptors at 37°C
CD8 T cells/tetramers (medium) . | Incubation time . | . | . | . | . | ||||
|---|---|---|---|---|---|---|---|---|---|
| . | 1 h, % . | 2 h, % . | 3 h, % . | 8 h, % . | 24 h, % . | ||||
| LMP1/LMP1 (PBS) | 92 | 94 | 95 | 86 | 86 | ||||
| Flu58/LMP1 (PBS) | 0.9 | 1.2 | 1.1 | 1.0 | 1.0 | ||||
| LLO91/LLO91 (PBS) | 68 | 78 | 82 | — | 69 | ||||
| LMP1/LMP1 (serum) | 98 | 94 | 86.4 | 71 | 28.8 | ||||
| LLO91/LLO91 (serum) | 88 | 66 | 62 | — | 10.6 | ||||
CD8 T cells/tetramers (medium) . | Incubation time . | . | . | . | . | ||||
|---|---|---|---|---|---|---|---|---|---|
| . | 1 h, % . | 2 h, % . | 3 h, % . | 8 h, % . | 24 h, % . | ||||
| LMP1/LMP1 (PBS) | 92 | 94 | 95 | 86 | 86 | ||||
| Flu58/LMP1 (PBS) | 0.9 | 1.2 | 1.1 | 1.0 | 1.0 | ||||
| LLO91/LLO91 (PBS) | 68 | 78 | 82 | — | 69 | ||||
| LMP1/LMP1 (serum) | 98 | 94 | 86.4 | 71 | 28.8 | ||||
| LLO91/LLO91 (serum) | 88 | 66 | 62 | — | 10.6 | ||||
— indicates not determined.
Binding of specific radiolabeled tetramers to CD8 T-cell lines
In order to establish reliable conditions for our radiometal labeling procedure, we first used 111In, a pure gamma-emitting isotope with a 3-day half-life. We assembled radiolabeled tetramers by adding one 111In-DTPA-biotin molecule and 3 biotinylated monomers for each streptavidin molecule, since each molecule of streptavidin has 4 biotin binding sites. Highly purified 111In-biotinylated DTPA (99%) was prepared for tetramer labeling. To separate the radiolabeled multimers from nonlabeled small size products, the mixture was passed through a BioRad Econo-Pac10G column. Fluorescent tetramers were tested for specific binding and served as an additional quality control for use when assembling radiolabeled tetramers.
The 111In-radiolabeled multimers were incubated with cognate and control CD8+ T-cell lines to determine if they displayed specific binding and to determine their binding kinetics and internalization. 111In-labeled LMP1 tetramers exhibited dose-dependent specific binding to the LMP1 CD8+ clone (Figure 2A). In contrast, there was little binding of 111In-LMP1 tetramers to the Flu-specific control CD8+ T cells. As another control, 111In-Flu tetramers showed little binding to LMP1 CD8+ T cells even at very high concentrations. These results strongly indicated that the peptide-specific tetramers could be successfully radiolabeled with maintenance of their binding specificity for their cognate CD8+ T cells. These observations were confirmed in the mouse model by a similar experiment testing 111In-labeled LLO91 tetramer binding against murine LLO91-99-specific CD8+ T cells (Figure 2B).
Specific binding and internalization of 111In-radiolabeled tetramers. (A) LMP1-specificorFlu58-66-specific CD8 cell lines were incubated with 111In-LMP1 tetramers or 111In-Flu58-66 tetramers at different concentrations on ice. Binding of 111In-LMP1 tetramers to LMP1-specific CD8 T cells (•) was dose dependent. Little binding to 111In-LMP1 tetramers was observed when tested against the Flu-specific CD8 T cells (○). There was also little binding of the 111In-Flu tetramers to LMP1 CD8 T cells (▴). (B) Dose-dependent binding of 111In-labeled LLO91-99 tetramers to mouse LLO91-99-specific CD8 T cells (•) and little binding to the control p60217-225-specific CD8 T cells (○) were observed. (C) 111In-LLO91-99 tetramers were added to LLO91-99-specific CD8 (left bars) or control p60217-225-specific CD8 (right bars) T-cell lines. Cells were incubated at 37° C. Surface binding (▪) and internalization (□) of 111In-LLO91-99 tetramers were measured at different time points. Both cell-surface binding and internalization of 111In-LLO91-99 tetramers to LLO91-99 CD8 T cells progressively increase with prolonged incubation. There was little binding or tetramer internalization with the control CD8 T-cell line (P < .0001). Data represent the mean of 2 tests from a single representive experiment that was done 3 times. Error bars indicate SEM.
Specific binding and internalization of 111In-radiolabeled tetramers. (A) LMP1-specificorFlu58-66-specific CD8 cell lines were incubated with 111In-LMP1 tetramers or 111In-Flu58-66 tetramers at different concentrations on ice. Binding of 111In-LMP1 tetramers to LMP1-specific CD8 T cells (•) was dose dependent. Little binding to 111In-LMP1 tetramers was observed when tested against the Flu-specific CD8 T cells (○). There was also little binding of the 111In-Flu tetramers to LMP1 CD8 T cells (▴). (B) Dose-dependent binding of 111In-labeled LLO91-99 tetramers to mouse LLO91-99-specific CD8 T cells (•) and little binding to the control p60217-225-specific CD8 T cells (○) were observed. (C) 111In-LLO91-99 tetramers were added to LLO91-99-specific CD8 (left bars) or control p60217-225-specific CD8 (right bars) T-cell lines. Cells were incubated at 37° C. Surface binding (▪) and internalization (□) of 111In-LLO91-99 tetramers were measured at different time points. Both cell-surface binding and internalization of 111In-LLO91-99 tetramers to LLO91-99 CD8 T cells progressively increase with prolonged incubation. There was little binding or tetramer internalization with the control CD8 T-cell line (P < .0001). Data represent the mean of 2 tests from a single representive experiment that was done 3 times. Error bars indicate SEM.
While efficacy of killing by the alpha-emitting elements should be increased if the armed tetramers are internalized, it is not a prerequisite for killing. Time-dependent tetramer internalization of the tetramers was seen, but only small amounts (6%-8%) of 111In-LMP1 tetramers were internalized after a 1-hour incubation at 37° C when tested on LMP1 CD8+ T cells. Maximum internalization (∼ 22%) was achieved with a 24-hour incubation (Figure 2C).
Specific killing of CD8+ T cells by armed 225Ac-labeled tetramers
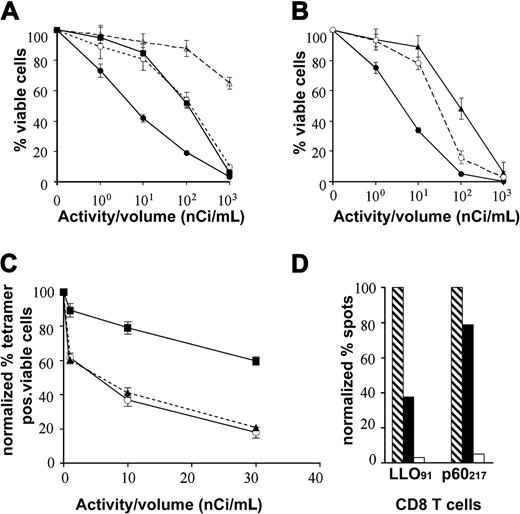
225Ac, a radiometal atomic generator that decays to yield 4 net alpha particles, can be stably conjugated to proteins by use of bifunctional DOTA chelates.15 Therefore, using the MHC multimer radiolabeling method that was developed for 111In tracing (see the previous section), we next constructed radiolabeled suicide tetramers armed with 225Ac generators. Biotinylated DOTA was labeled with 225Ac at high yields (> 96%). The armed 225Ac-LMP1 tetramers effectively killed the targeted LMP1 CD8+ T-cell clones at small doses (ED50 = 5-8 nCi/mL or 1-1.6 μg/mL; Figure 3A). In contrast, the armed 225Ac-LMP1 tetramers at 5 nCi/mL to 8 nCi/mL exhibited much less toxicity to control Flu-specific CD8+ T cells. Nonspecific cytotoxicity was induced at 15- to 40-fold-higher doses (ED50 = 110-200 nCi) when using 225Ac DOTA alone. Much higher levels of unlabeled specific LMP1 tetramers (100-140 μg/mL) were required to induce mild cytotoxicity in targeted CD8+ T cells. In the murine system, similar high-potency specific cell killing by suicide LLO91-99 tetramers was also demonstrated. LLO91-99 peptide–specific CD8+ T cells were effectively killed after incubation with 225Ac-LLO91-99 tetramers (Figure 3B); as another specificity control, the T-cell killing by suicide LLO91-99 tetramers was partially blocked by the addition of a 50-fold excess of unlabeled LLO91-99-specific tetramers.
Antigen-specific CD8 T-cell killing by armed 225Ac-tetramers. (A) 225Ac-tetramers (LMP1) were added to LMP1-CD8 (•) or Flu58-66-CD8 T cells (○) and then incubated at 37° C for 72 hours. The 225Ac-LMP1 tetramers (5 nCi/μg) efficiently killed the targeted LMP1-specific CD8 T cells with great potency (•, ED50 = 5-8 nCi/mL or 1-1.6 μg/mL). About 15- to 40-fold–higher doses (ED50 = 110-200 nCi) of 225Ac-LMP1 tetramers (○) or equivalent 225Ac DOTA alone (▪) were required for nonspecific killing (P < .001). Much higher levels (100-140 μg/mL) of unlabeled specific LMP1 tetramer alone (▵) were required to induce mild cytotoxicity in targeted LMP1 CD8+ T cells. (B) 225Ac-LLO91-99 tetramers (•) specifically killed mouse LLO91-99-specific CD8+ T cells (ED50 = 5-7 nCi/mL or 1-1.4 μg/mL). More than 15-fold–larger amounts of 225Ac (▴) were required to kill control p60217-225-specific CD8+ T cells. Addition of excess (50-fold) unlabeled LLO91 tetramers (○) partially blocked cell killing of 225Ac-LLO91-99 tetramers. (C) 225Ac-LLO91-99 tetramers at 1 to 30 nCi/mL (0.2-6 μg/mL) were added to mixed cell (50:50) cultures of target LLO91-99-specific CD8 T cells and p60217-225-specific CD8 T cells. 225Ac-LLO91 tetramers (▴) selectively killed LLO91-CD8 T cells in a mixed cell culture, and were as effective as killing in cultures of purified LLO91-CD8 T cells alone (○). 225Ac-LLO91 tetramers produced minimal cytotoxicity in control p60217-225-specific CD8 T cells in a mixed cell culture (▪; P < .0001). Data represent the mean plus or minus the standard error (SE) of 3 tests in a single representive experiment done 2 times. (D) 225Ac-LLO91-99 tetramers (5 nCi/mL or 1 μg/mL) reduce IFN-γ–secreting clones in targeted LLO91-99-specific CD8+ T cells. The level of IFN-γ–secreting cells in suicide tetramer–treated LLO91-99-specific CD8+ T cells (▪) decreased significantly (83 ± 14 colonies) when compared with the nontreated control T cells (□; 219 ± 21 colonies). The control p60217-specific CD8 T cells showed only modest nonspecific reduction in IFN-γ–secreting cells (133 ± 7 vs 105 ± 5). Negative control stimulations with irrelevant peptides produce few spots (□). Bars represent normalized percent of spots from each CD8 cell line either treated with 225Ac-LLO91 tetramers or controls.
Antigen-specific CD8 T-cell killing by armed 225Ac-tetramers. (A) 225Ac-tetramers (LMP1) were added to LMP1-CD8 (•) or Flu58-66-CD8 T cells (○) and then incubated at 37° C for 72 hours. The 225Ac-LMP1 tetramers (5 nCi/μg) efficiently killed the targeted LMP1-specific CD8 T cells with great potency (•, ED50 = 5-8 nCi/mL or 1-1.6 μg/mL). About 15- to 40-fold–higher doses (ED50 = 110-200 nCi) of 225Ac-LMP1 tetramers (○) or equivalent 225Ac DOTA alone (▪) were required for nonspecific killing (P < .001). Much higher levels (100-140 μg/mL) of unlabeled specific LMP1 tetramer alone (▵) were required to induce mild cytotoxicity in targeted LMP1 CD8+ T cells. (B) 225Ac-LLO91-99 tetramers (•) specifically killed mouse LLO91-99-specific CD8+ T cells (ED50 = 5-7 nCi/mL or 1-1.4 μg/mL). More than 15-fold–larger amounts of 225Ac (▴) were required to kill control p60217-225-specific CD8+ T cells. Addition of excess (50-fold) unlabeled LLO91 tetramers (○) partially blocked cell killing of 225Ac-LLO91-99 tetramers. (C) 225Ac-LLO91-99 tetramers at 1 to 30 nCi/mL (0.2-6 μg/mL) were added to mixed cell (50:50) cultures of target LLO91-99-specific CD8 T cells and p60217-225-specific CD8 T cells. 225Ac-LLO91 tetramers (▴) selectively killed LLO91-CD8 T cells in a mixed cell culture, and were as effective as killing in cultures of purified LLO91-CD8 T cells alone (○). 225Ac-LLO91 tetramers produced minimal cytotoxicity in control p60217-225-specific CD8 T cells in a mixed cell culture (▪; P < .0001). Data represent the mean plus or minus the standard error (SE) of 3 tests in a single representive experiment done 2 times. (D) 225Ac-LLO91-99 tetramers (5 nCi/mL or 1 μg/mL) reduce IFN-γ–secreting clones in targeted LLO91-99-specific CD8+ T cells. The level of IFN-γ–secreting cells in suicide tetramer–treated LLO91-99-specific CD8+ T cells (▪) decreased significantly (83 ± 14 colonies) when compared with the nontreated control T cells (□; 219 ± 21 colonies). The control p60217-specific CD8 T cells showed only modest nonspecific reduction in IFN-γ–secreting cells (133 ± 7 vs 105 ± 5). Negative control stimulations with irrelevant peptides produce few spots (□). Bars represent normalized percent of spots from each CD8 cell line either treated with 225Ac-LLO91 tetramers or controls.
To validate that the cell selectivity of killing by 225Ac-LLO91-99 tetramers of LLO91-99 peptide–specific CD8+ T cells within a mixture of possible target cells (as might occur in most applications), 225Ac-LLO91-99 tetramers at concentrations of 1 nCi/mL to 30 nCi/mL (0.2-6 μg/mL) were added to mixed cultures of LLO91-99-specific CD8+ and p60217-225-specific CD8+ T cells. After a 72-hour incubation with 225Ac-LLO91-99 tetramers, significant cell killing was demonstrated in the whole population as judged by [3H]thymidine incorporation and confirmed by reductions in viable cells determined with trypan blue staining. When the remaining viable cells in the population were analyzed by tetramer flow cytometry to define their specificities, there was a significant reduction in LLO91-99-specific CD8+ T cells (P < .001; Figure 3C). In contrast, the nontargeted p60217-225-specific CD8+ T-cell population in the mixed cell culture showed only modest reductions even when exposed to higher quantities of 225Ac-LLO91-99 tetramer (10 nCi/mL or 2 μg/mL).
Functional assays confirmed a significant reduction in the level of IFN-γ secretion in suicide tetramer–treated LLO91-99 CD8+ T cells when compared with the nontreated control T cells (Figure 3D). In the control p60217-225-specific CD8 T-cell population, modest nonspecific reduction in IFN-γ secretion was observed. These results demonstrate that armed tetramers can selectively delete both numbers and function of specific cytotoxic T lymphocytes (CTLs) with high specificity and induce little cytotoxicity within the other CD8+ T-cell populations, mimicking most applications envisioned.
Discussion
The choice of agents to arm MHC tetramers for specific cytotoxicity is limited. The selected agent must be capable of extraordinary potency, killing with a limited number of bound tetramers, most or all of which remain outside the targeted T cell. In addition, the cytotoxic effect must be of short range to allow selective killing of only the individual targeted cognate T cell while sparing bystander cells. As a consequence, nearly all drugs and toxins will be weakly effective. Among the radioisotopes, only alpha-emitting nuclides offer a solution. The 225Ac isotope (an atomic nanogenerator) has a 10-day half-life, emits one alpha, and is the parent molecule in a decay cascade that produces a net of 6 new elements and 4 alpha particles. 225Ac-radiolabeled monoclonal antibodies have been proposed for use in cancer therapy, as these targeted nanogenerators are capable of specifically killing individual cancer cells at extremely low doses without significant toxicity.15 Because it takes several hours to prepare purified radiolabeled tetramers, the much shorter half-life of other alpha particle emitters of possible clinical utility (At-211, B1-213) may limit their potential application for these purposes. These initial experiments now can be extended to study clonal deletion of antigen-specific CD8+ T cells in vivo. Alternatively, this strategy may hold potential for ex vivo purging of specific CTLs prior to bone marrow and stem cell transplantation to prevent graft-versus-host disease. Such an approach would require identification of key immunodominant targets in the human system, which are currently unknown. Previous studies showed that soluble MHC tetramers injected into mice could modulate antigen-specific T-cell response in vivo,28 suggesting stability in vivo. Moreover, our previous work with the alpha-emitting Bi-213–labeled antibodies has demonstrated the ability to target hematopoietic cells in patients in the bone marrow, liver, spleen, and blood within 10 minutes, suggesting that significant depletions within these compartments in a short time-frame is possible.12 However, the induction of clonal deletion of T cells distant from the well-vascularized organs in vivo by suicide tetramers may be limited by structural instability of the molecules; techniques to stabilize the constructs may be necessary. Predictions as to the consequences of the rapid expected clearance of these molecules from plasma are difficult without in vivo study. These methods for selective T-cell clonal deletion may allow advances in our understanding of the role of antigen-specific CD8+ T cells in diverse disease processes including infection, autoimmune disease, and other CD8+ cell-mediated disorders and may provide a pathway to possible T-cell–selective therapies of autoimmune or neoplastic origin.
Prepublished online as Blood First Edition Paper, June 24, 2004; DOI 10.1182/blood-2004-01-0324.
Supported by National Institutes of Health grants RO1 CA55349 and PO1 CA33049, The Experimental Therapeutics Center, The Joseph Leroy and Anne C. Warner Fund, The Lacher Foundation, and the Lymphoma Foundation.
D.A.S. is a Doris Duke Distinguished Clinical Scientist.
The publication costs of this article were defrayed in part by page charge payment. Therefore, and solely to indicate this fact, this article is hereby marked “advertisement” in accordance with 18 U.S.C. section 1734.
We thank Patrick Guirnalda and Eva Menet for technical assistance.


This feature is available to Subscribers Only
Sign In or Create an Account Close Modal