Abstract
Gene therapy has been shown to be a highly effective treatment for infants with typical X-linked severe combined immunodeficiency (SCID-X1, γc-deficiency). For patients in whom previous allogeneic transplantation has failed, and others with attenuated disease who may present later in life, the optimal treatment strategy in the absence of human leukocyte antigen (HLA)–matched donors is unclear. Here we report the failure of gene therapy in 2 such patients, despite effective gene transfer to bone marrow CD34+ cells, suggesting that there are intrinsic host-dependent restrictions to efficacy. In particular, there is likely to be a limitation to initiation of normal thymopoiesis, and we therefore suggest that intervention for these patients should be considered as early as possible.
Introduction
X-linked severe combined immunodeficiency (SCID-X1) is caused by mutations in the gene encoding the common cytokine receptor gamma chain (γc), inducing the complete absence of mature T and natural killer (NK) cells.1 Attenuated forms of γc-deficiency have been described, and due to atypical clinical and immunological phenotypes, may well be underrecognized.2 Human leukocyte antigen (HLA)–genoidentical hematopoietic stem cell transplantation (HSCT) confers a 90% chance of disease-free survival in the first 10 to 20 years. In contrast, haploidentical HSCT is associated with an overall 75% survival rate at 5 years and a significant risk of graft-versus-host disease (GVHD).3,4 In addition, over a long term, there is often evidence for persistent B-cell deficiency and, in some cases, progressive defects of cellular immunity as a result of an exaggerated decline in thymic output.5-8
Recently, 2 clinical studies of somatic gene therapy for SCID-X1 have demonstrated sustained correction of immunodeficiency in 15 out of 16 infants with classical disease despite the occurrence of a clonal proliferation in 2 of them.9-12 However, it remains unclear whether this strategy can be effectively applied to patients in whom the disease phenotype is attenuated, and also to patients with residual defects or failing immunity following previous HSCT.
Study design
Patients
Approval was obtained from the review boards of the Institute of Child Health and the Great Ormond Street Hospital for Children (London), the Hôpital Necker and the INSERM (Paris), and the Cincinnati Children's Research Foundation (OH) for these studies. Informed consent was provided according to the Declaration of Helsinki.
Patient 1. Patient 1 (P1) was the first child of consanguineous parents and was 20 years old at the time of gene therapy. He was diagnosed soon after birth and found to have a 16 base pair (bp) deletion within exon 5 of the γc gene leading to a frameshift at F221 and abolition of cell-surface protein expression. He received a whole HLA phenoidentical paternal HSCT at the age of 3 weeks without any prior myeloablative conditioning. Over the following years he developed bronchiectasis, idiopathic hepatitis, and protein-losing enteropathy. Reassessment of his immune status revealed marked T-cell deficiency, absent NK cells, and low numbers of B cells. A magnetic resonance imaging (MRI) of his chest confirmed bronchiectasis, and absence of thymic tissue. Attempts to locate his estranged father failed, ruling out the possibility of a supplemental HSCT. An unrelated donor search was also unsuccessful. Following ethical and regulatory approval, and with his fully informed consent, he was treated by gene therapy.
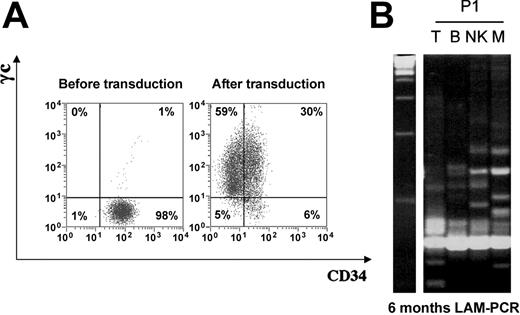
CD34+ cell transduction and LAM-PCR analysis of peripheral leukocyte subsets. (A) γc expression on P1 CD34+ cells before and after transduction. (B) LAM-PCR analysis of the vector insertion site restriction length polymorphism present in peripheral blood leukocytes of P1. DNA samples of approximately 1 to 50 ng isolated directly from sorted peripheral blood leukocytes were analyzed at 6 months after gene therapy. Prominent bands in all lanes at the bottom of the gel are internal PCR controls. Left lane shows 100 bps DNA marker; other lanes: T, CD3+ cells; B, CD19+ cells; NK, CD16/56+ cells; and M, CD33+ myeloid cells.
CD34+ cell transduction and LAM-PCR analysis of peripheral leukocyte subsets. (A) γc expression on P1 CD34+ cells before and after transduction. (B) LAM-PCR analysis of the vector insertion site restriction length polymorphism present in peripheral blood leukocytes of P1. DNA samples of approximately 1 to 50 ng isolated directly from sorted peripheral blood leukocytes were analyzed at 6 months after gene therapy. Prominent bands in all lanes at the bottom of the gel are internal PCR controls. Left lane shows 100 bps DNA marker; other lanes: T, CD3+ cells; B, CD19+ cells; NK, CD16/56+ cells; and M, CD33+ myeloid cells.
Patient 2. P2 was born in 1985 to unrelated parents. From 5 years of age, he developed recurrent respiratory tract infections, protracted diarrhea, and extensive molluscum contagiosum. Immunologic investigations revealed a profound CD4+ lymphopenia, although T, NK, and B cell counts were normal. Serum levels of immunoglobulin (Ig) G, IgA, and IgM were normal or increased. IgG2, IgG3, and IgG4 levels were low and antibodies to immunization antigens were undetectable. In vitro T-cell responses showed normal proliferation to mitogens and very low or absent proliferation to specific antigens. Over the subsequent 10-year period, his clinical status deteriorated and he developed bronchiectasias, chronic pulmonary failure, protracted diarrhea, and recurrent herpes zoster virus infection. The search for a γc gene mutation revealed a 664 C to A, R222S mutation in exon 5. While γc expression was weakly detected on T cells, CD34+ cells did not express the protein. At the age of 15 years, he was considered for HSCT but no genoidentical or HLA-matched unrelated donors were available. Following ethical and regulatory approval, and with his fully informed consent, he was treated by gene therapy.
Gene transfer protocols
In vitro investigation
Lymphoid and hematopoietic cell subsets were phenotypically characterized by the labeling of whole blood with combinations of directly conjugated monoclonal antibodies, all purchased from Becton Dickinson Biosciences (San Jose, CA). Proliferation assays, T-cell receptor (TCR) spectratypes, T-cell receptor signal joint excision circles (TRECs), and provirus integration (MFG-B2-γc) were measured as previously described.10,11 The copy number of the γ-chain transgene was determined by real-time polymerase chain reaction (PCR) after extracting DNA from cells described. Insertion site analysis for visualization of the clonal contribution to T lymphopoiesis and other lineages was performed by linear amplification-mediated PCR (LAM-PCR) as previously described.11,13
Results and discussion
Following transduction, 2.8 × 106 and 35 × 106 total cells/kg were infused into P1 (body weight = 53 kg) and P2 (body weight = 34 kg) respectively, 30% and 13% of which coexpressed CD34 and γc (Figure 1). This transduction rate is comparable to that obtained for patients younger than 1 year of age as previously reported.9,10 No adverse events were observed. Both patients were followed up for at least 180 days (Table 1).
Immunologic and molecular investigations of the patient's peripheral blood leukocytes
. | - 15 d . | . | + 15 d . | . | + 60 d . | . | + 120 d . | . | + 180 d . | . | Control values . | |||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
. | P1 . | P2 . | P1 . | P2 . | P1 . | P2 . | P1 . | P2 . | P1 . | P2 . | . | |||||
| ALC, × 109/L | 3.93 | 3.07 | 3.4 | 2.57 | 4.44 | 3.24 | 5.29 | 4.05 | 4.64 | 3.39 | 800-4000 | |||||
| CD3+, % | 95 | 93 | 94 | 93 | 96 | 91 | 96 | 92 | 96 | 90 | 70-80 | |||||
| CD4+, % | 28 | 4 | 25 | 5 | 28 | 6 | 28 | 7 | 22 | 8 | 60-80 | |||||
| CD8+, % | 68 | 88 | 69 | 84 | 70 | 85 | 70 | 85 | 75 | 81 | 20-40 | |||||
| CD4+CD45RA+, % | 0.2 | 0.08 | 2 | 0.05 | 2 | 0.06 | ND | 0.03 | 3 | 0.08 | 30-40 | |||||
| CD4+CD45RO+, % | 27 | 3.92 | 23 | 4.95 | 18 | 5.94 | ND | 6.9 | 20 | 7.9 | 30-40 | |||||
| CD8+CD45RA+, % | 12 | 23 | 16 | 17 | 18 | 14 | ND | 6.8 | 17 | 8.1 | 10-20 | |||||
| CD8+CD45RO+, % | 53 | 67 | 43 | 67 | 41 | 74 | ND | 79 | 43 | 73 | 10-20 | |||||
| CD19+, % | 4 | 3 | 5 | 3 | 3 | 2 | 3 | 3 | 3 | 5 | 10-20 | |||||
| CD16/56+, % | 0.5 | 2 | 0.1 | 1.5 | 0.1 | 2 | 0.2 | 0.9 | 0.1 | 1 | 5-10 | |||||
| IgG, g/L | 16.6 | ND | 9.61 | ND | 12.4 | ND | 11.6 | ND | 13.2 | 13.2 | 6.4-12.4 | |||||
| IgA, g/L | 0.26 | ND | < 0.06 | ND | 0.09 | ND | < 0.06 | ND | 0.07 | 2.37 | 0.8-3.4 | |||||
| IgM, g/L | 0.46 | ND | 0.56 | ND | 0.58 | ND | 0.53 | ND | 0.6 | 0.89 | 0.5-1.5 | |||||
| γc integration study | ||||||||||||||||
| PBL | - | - | +* | - | + | + | + | + | + | - | - | |||||
| CD3+ | - | - | - | ND | ND | ND | + | + | + | - | - | |||||
| CD3- | - | - | - | ND | ND | ND | + | ND | + | ND | - | |||||
. | - 15 d . | . | + 15 d . | . | + 60 d . | . | + 120 d . | . | + 180 d . | . | Control values . | |||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
. | P1 . | P2 . | P1 . | P2 . | P1 . | P2 . | P1 . | P2 . | P1 . | P2 . | . | |||||
| ALC, × 109/L | 3.93 | 3.07 | 3.4 | 2.57 | 4.44 | 3.24 | 5.29 | 4.05 | 4.64 | 3.39 | 800-4000 | |||||
| CD3+, % | 95 | 93 | 94 | 93 | 96 | 91 | 96 | 92 | 96 | 90 | 70-80 | |||||
| CD4+, % | 28 | 4 | 25 | 5 | 28 | 6 | 28 | 7 | 22 | 8 | 60-80 | |||||
| CD8+, % | 68 | 88 | 69 | 84 | 70 | 85 | 70 | 85 | 75 | 81 | 20-40 | |||||
| CD4+CD45RA+, % | 0.2 | 0.08 | 2 | 0.05 | 2 | 0.06 | ND | 0.03 | 3 | 0.08 | 30-40 | |||||
| CD4+CD45RO+, % | 27 | 3.92 | 23 | 4.95 | 18 | 5.94 | ND | 6.9 | 20 | 7.9 | 30-40 | |||||
| CD8+CD45RA+, % | 12 | 23 | 16 | 17 | 18 | 14 | ND | 6.8 | 17 | 8.1 | 10-20 | |||||
| CD8+CD45RO+, % | 53 | 67 | 43 | 67 | 41 | 74 | ND | 79 | 43 | 73 | 10-20 | |||||
| CD19+, % | 4 | 3 | 5 | 3 | 3 | 2 | 3 | 3 | 3 | 5 | 10-20 | |||||
| CD16/56+, % | 0.5 | 2 | 0.1 | 1.5 | 0.1 | 2 | 0.2 | 0.9 | 0.1 | 1 | 5-10 | |||||
| IgG, g/L | 16.6 | ND | 9.61 | ND | 12.4 | ND | 11.6 | ND | 13.2 | 13.2 | 6.4-12.4 | |||||
| IgA, g/L | 0.26 | ND | < 0.06 | ND | 0.09 | ND | < 0.06 | ND | 0.07 | 2.37 | 0.8-3.4 | |||||
| IgM, g/L | 0.46 | ND | 0.56 | ND | 0.58 | ND | 0.53 | ND | 0.6 | 0.89 | 0.5-1.5 | |||||
| γc integration study | ||||||||||||||||
| PBL | - | - | +* | - | + | + | + | + | + | - | - | |||||
| CD3+ | - | - | - | ND | ND | ND | + | + | + | - | - | |||||
| CD3- | - | - | - | ND | ND | ND | + | ND | + | ND | - | |||||
ND indicates not done; ALC absolute lymphocyte counts; — indicates no control value.
Positive signal studied by quantitative PCR shows < 1% level of gene marking
For P1, despite persisting PCR signals indicating the presence of transgene at low levels, there was no significant change in immunological parameters or clinical status up to 2 years after gene therapy. Specifically, there was no evidence for production of naive CD45RA+ or CD27hiCD45RO- T cells, and the numbers of TRECs remained very low (< 20/105 peripheral blood lymphocytes [PBLs]). Molecular analysis of integration sites in sorted blood lineages performed 6 months after gene therapy revealed polyclonal marking in B, NK, and myeloid cells comparable to that observed in other patients treated by the same protocol. This indicated successful transduction and engraftment of CD34+ cells. However, the pattern of T-cell marking showed few clones (Figure 1, right panel). These observations are consistent with levels of marking in purified T, B, and myeloid lineages less than 1% determined by quantitative PCR up to 2 years after engraftment, and are suggestive of a host-dependent restriction to T-cell development.
For P2, PCR analysis of PBLs or CD3+ sorted cells also revealed a very low γc transgene signal, which disappeared 6 months after gene therapy (Table 1). CD4+ lymphopenia persisted, as did the absence of T-cell proliferative responses. There was no evidence for the emergence of naive T cells and the numbers of TRECs remained very low (< 20/105 PBLs). No improvement of his clinical condition was observed. Despite intensive therapy for recurrent skin and pulmonary infections, his condition deteriorated and he died of respiratory failure 1.5 years after gene therapy. To verify successful gene transfer, cells preserved at completion of gene transfer were thawed and cocultured on a MS-5 feeder cell line. After 5 weeks of culture, both myeloid (CD33+) and lymphoid (CD19+) precursors were present. Immunoselected CD33+ cells were strongly positive for the γc transgene by PCR, indicating that early hematopoietic precursors had been transduced and were viable at the time of engraftment (data not shown).
The failure of gene therapy to produce therapeutic effects in these 2 patients raises important issues for the extension of this type of therapy, as CD34+ progenitor cells were successfully transduced in vitro. The capacity to initiate (or reinitiate) thymopoiesis is likely to be time dependent and influenced by chronic infection, previous GVHD, and physiological aging. The absence of ongoing thymocyte and thymic epithelial cell (TEC) interaction results in disorganization of thymic architecture and hypoplasia, which may become irreversible.14-18 This likely explains why allogeneic HSCT performed for SCID patients in the neonatal period results in superior thymic output compared with those performed later.6,7,19 Similar arguments may well apply to patients with attenuated disease in whom thymopoiesis is severely compromised. The results of gene therapy in these 2 patients with SCID-X1 indicate that there may well be age-related restrictions to efficacy. Therefore, we suggest that intervention should be considered at the earliest possible indication.
Prepublished online as Blood First Edition Paper, February 1, 2005; DOI 10.1182/blood-2004-12-4837.
Supported by grants from INSERM, lst Association Française contre les Myopathies, Le Programme Hospitalier de Recherche Clinique of the French Health Ministry, the Assistance Publique-Hôpitaux de Paris, EC contract no. QLK3-LT 2001 (coordinator, G. Wagemaker), the Jeffrey Modell Foundation, The Welcome Trust (A.J.T.), the Primary Immunodeficiency Association, the Jeans for Genes Appeal, and the Chronic Granulomatous Disease Research Trust.
A.J.T. and S.H.-B.-A. contributed equally to this work.
An Inside Blood analysis of this article appears in the front of this issue.
The publication costs of this article were defrayed in part by page charge payment. Therefore, and solely to indicate this fact, this article is hereby marked “advertisement” in accordance with 18 U.S.C. section 1734.
We are indebted to families of the patients for their continuous support, and to the medical and nursing staff of the Pediatric Immunology Unit at the Necker Hospital and the Immunology Unit at Great Ormond Street Hospital. We would also like to acknowledge Fabian Gross, Chantal Martinache, and Jean-Marc Luby for technical contribution.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal