Abstract
Human T-cell leukemia virus type 1 (HTLV-1) is the causative agent of adult T-cell lymphoma/leukemia (ATL). The HTLV-1 envelope gene exhibits limited variability when examined from infected individuals, but has not been tested using infectious clones of the virus in animal models. In vitro assays indicate that HTLV-1 envelope (Env) Ser75Ile, Asn95Asp, and Asn195Asp surface unit (SU) mutants are able to replicate in and immortalize lymphocytes. Herein, we examined the effects of these Env mutants in rabbits inoculated with HTLV-1 immortalized ACH.75, ACH.95, or ACH.195 cell lines (expressing full-length molecular clones with the SU mutations) or the ACH.1 cell line (expressing wild-type SU). All rabbits became infected, and the fidelity of the mutations was maintained throughout the 8-week study. However, SU point mutations resulted in decreased antibody responses to viral group-associated antigen (Gag) and Env antigens. ACH.195 rabbits had a selective decreased antibody response to SU, and one ACH.195 rabbit had an antibody response to both HTLV-1 and HTLV-2 SUs. Some mutant inoculation groups had altered proviral loads. However, peripheral-blood mononuclear cell (PBMC) proviral loads did not correlate with antibody responses. Our data are the first to demonstrate that mutations in critical determinants of HTLV-1 Env SU altered antibody responses and proviral loads, but do not prevent viral replication in vivo.
Introduction
Human T-cell leukemia virus type 1 (HTLV-1) is a complex retrovirus etiologically linked with adult T-cell leukemia/lymphoma (ATL), HTLV-1-associated myelopathy/tropical spastic paresis (HAM/TSP), and a variety of other immune-mediated disorders.1 ATL is an aggressive T-cell leukemia that typically manifests in middle-aged to elderly patients who were originally infected with the virus neonatally during breastfeeding.2 There are 4 clinically overlapping subsets of ATL. In order of decreasing prognostic survival time, these include smoldering, chronic, lymphoma, and acute.3 The most severe form is the acute form, which manifests as an aggressive T-cell leukemia with concomitant hypercalcemia, bone marrow involvement, generalized lymphadenopathy, skin lesions, and spleen and liver involvement.4 Circulating leukemic cells in patients with ATL have a characteristic lobulated nuclear morphology and are most often CD2+, CD3+, CD4+, and CD8- T cells.1
HTLV-1 envelope protein (Env) is unique among retroviral envelope proteins in that when isolates from ATL or HAM/TSP patients are compared, there is a high degree of sequence conservation in the env region, with variability ranging from 1% to at most 8%.5-10 HTLV-1 Env is a 488-amino-acid protein. It is synthesized as a polyprotein precursor (gp62), which is subsequently glycosylated and cleaved into 2 proteins, surface unit gp46 (SU) and transmembrane gp21 (TM).11,12 SU is required for entry into the target cell by mediating specific attachment to a cellular receptor, which was recently reported to be the glucose transporter, GLUT-1.13 The TM supports fusion between viral and cellular membranes to allow viral entry.
HTLV-1 SU is a 312-amino-acid protein. The C-terminal half of SU is very antigenic in humans, being recognized by antibodies in serum from over 95% of HTLV-1-infected individuals.10 Amino acids 187-196 of SU are a major target for neutralizing antibodies, as evidenced by the use of monoclonal antibodies to inhibit syncytium formation or infectivity as well as enzyme-linked immunosorbent assay (ELISA) peptide binding assays and in vivo peptide immunization studies.10,14-19 Early studies using site-directed mutagenesis demonstrated functional domains within SU involved in intracellular maturation, syncytium formation, and the association between SU and TM.10,14,20-22 Subsequent development of a cell-to-cell transmission assay allowed for separation of fusion events from infectivity events and verified that cell-to-cell fusion and cell-to-cell transmission are independent events.23,24
A major breakthrough in HTLV-1 research came with the development of the ACH HTLV-1 molecular clone in the mid-1990s.25 Subsequent mutagenesis of the wild-type ACH HTLV-1 molecular clone allowed for characterization of mutations of key viral proteins in the context of the entire virus.26-28 Wild-type and mutant HTLV-1 molecular clones were used to create immortalized human peripheral-blood mononuclear cell (PBMC) lines, which were inoculated into rabbits to define how mutations in key viral proteins alter viral infectivity in vivo.26-29 It is through such studies that the necessity of the viral proteins Rex, p12I, and p30II for establishment and maintenance of infection in vivo has been determined.
Based on earlier studies using transient transfections of HTLV-1 env plasmids in key determinants of Env, Tsukahara et al30 created identical mutations in the Env region of the ACH molecular clone. Specifically, point mutations were generated to alter key amino acids 75, 81, 95, 101, 105, and 195 of the SU protein to create the full-length proviral plasmids ACH.75, ACH.81, ACH.95, ACH.101, ACH.105, and ACH.195, respectively. Transient transfections of these molecular clones into 293 T cells showed similar levels of Gag (p55Prgag, p24, p19) and Env (gp62, gp46, gp21) production compared with wild-type ACH, with the exception of ACH.81, which produced slightly lower levels of gp46.30 Cell-free and cell-to-cell transmission assays showed these clones to be equally infectious compared with the wild-type ACH molecular clone, with the exception of ACH.81 infection, which resulted in slightly decreased levels of p19 production.30 With the exception of ACH.101, these molecular clones were then shown to be capable of immortalizing human PBMCs in vitro.30
Unresolved is the question of whether these in vitro findings correlate with virus replication events in vivo. Specifically, given that there is little sequence variation in the HTLV-1 Env protein in the natural infection, would mutations in Env that are maintained in in vitro immortalization assays be tolerated in the in vivo infection? Would these mutations alter the antibody responses in the in vivo infection? In this study, we address these questions by comparing the antibody responses and proviral loads between rabbits inoculated with the wild-type ACH.1 cell line to rabbits inoculated with the ACH.75, ACH.95, and ACH.195 cell lines. These mutations all fall within regions predicted to be important for binding of SU to the viral receptor based on syncytium assays, and the ACH.195 mutation falls within a region that is a major target for neutralizing antibody responses.14-19 Our data indicate that following infection with these cell lines, rabbits became infected, and the fidelity of the mutations was maintained through 8 weeks of infection. The ACH.75 and ACH.95 mutations caused a decreased antibody response as measured by whole-virus ELISA. Interestingly, when assayed by Western blot, the ACH.195 mutation was associated with a decreased antibody response to the SU protein, and in one ACH.195 rabbit we were able to detect an antibody response to the HTLV-2 SU protein. In another ACH.195 rabbit we were able to demonstrate provirus in the PBMCs in the absence of a detectable antibody response. Comparison of PBMC proviral loads between inoculation groups indicated that the ACH.75 group had higher proviral loads than the ACH.1 and ACH.95 groups. However, correlation analysis did not demonstrate a relationship between PBMC proviral loads and the SU antibody responses. Our data confirm that earlier results obtained with ACH Env mutants via in vitro assays correlate with in vivo findings. Additionally, our data support previous reports of the importance of the region in SU between amino acids 187-196 for immunogenicity in vivo. Our study is the first to demonstrate that mutations in critical determinates of HTLV-1 SU, while altering proviral loads and antibody responses against HTLV-1 Env, do not prevent virus replication in vivo.
Materials and methods
Viral clones and cell lines
The derivation and infectious properties of the full-length ACH viral clone have been reported.25,31 The derivation and in vitro infectious properties of ACH.75, ACH.95, and ACH.195 viral clones have also been reported.30 ACH point mutants contain the env genes of HTLV-1 envelope mutants from the HTE series20 inserted between the SphI site at position 5121 and the NsiI site at position 6565 in the ACH clone.25
ACH.1, ACH.75, ACH.95, and ACH.195 cell lines were obtained from the outgrowth of immortalized PBMCs previously transfected with the ACH, ACH.75, ACH.95, and ACH.195 clones, respectively.30,31 PBMCs were isolated from healthy human donors by Ficoll-Hypaque (Pharmacia, Peapack, NJ) centrifugation. Cells were maintained in RPMI 1640 supplemented with 15% fetal bovine serum, l-glutamine (0.3 mg/mL), penicillin (100 U/mL), streptomycin (100 ìg/mL), and recombinant interleukin 2 (IL-2; 10 U/mL) (complete medium).
Detection of viral p19 matrix antigen
To estimate and compare levels of virus production among the ACH.1, ACH.75, ACH.95, and ACH.195 cell lines, duplicate samples of 106 cells from each cell line were washed and seeded in a 24-well plate in 1 mL complete RPMI. Culture samples were collected at 72 hours, serially diluted 10-fold, and tested for HTLV-1 p19 matrix antigen by a commercially available ELISA (Zeptometrix, Buffalo, NY). According to the manufacturer, sensitivity of detection is 25 pg/mL.
Detection of proviral sequences
For detection of provirus in cell lines and rabbit PBMCs, genomic DNA was harvested by affinity column (Qiagen, Valencia, CA) and examined for the presence of HTLV-1 sequences following polymerase chain reaction (PCR) amplification. For cell lines, 1 μg DNA was amplified using a primer pair specific for the HTLV-1 env region (5321, 5′-CAGCAGATCAGGCCCTACAG-3′; and 5885, 5′-GGAGAGTATAGGACGTGCCAAG-3′). For sequencing of provirus in rabbit PBMCs, 1 μg DNA was amplified using a primer pair specific for the HTLV-1 pX env region (ACH.75: 5321 and 5885; ACH.95: 5321 and 5580, 5′-CTTCCAGTAGGGGCTGGAGA-3′; ACH.195: 5718, 5′-CAGCCAACTGCCTCCCACCG-3′ and 5885). After an initial 10-minute incubation period at 94°C to activate the Taq polymerase (AmpliTaq Gold; Applied Biosystems, Foster City, CA), 40 cycles of PCR were performed with the following cycle parameters: denaturation at 94°C for 1 minute, annealing for 1 minute at 60°C (cell lines 5321, 5885), 64.8°C (PBMC 5321, 5885), or 55°C (PBMC 5718, 5885; PBMC 5351, 5580), and extension at 72°C for 45 seconds, followed by a final extension at 72°C for 5 minutes. The amplified products were separated in a 10% polyacrylamide gel and visualized with ultraviolet light following ethidium bromide staining.
HTLV-1-specific PCR products were sequenced to confirm specificity. PCR products were purified (Qiagen) and sequenced by the automated dye terminator cycle sequencing method (3700 DNA analyzer and Big Dye terminator cycle sequencing chemistry; Applied Biosystems) using the 5′ primer used for the PCR amplification.
Real-time PCR
The technique used for real-time PCR has been previously reported.32 DNA was extracted from the PBMCs with the Qiagen Blood Mini Kit according to the manufacturer's protocol (Qiagen). Quantity and quality of the DNA was assessed by NanoDrop spectroscopy (NanoDrop Technologies, Wilmington, DE). HTLV-1 tax gene primers at final concentration of 300 nM each of forward and reverse primers and 200 nM of dual-labeled probe, as well as the real-time PCR conditions, were previously described.32 Final concentrations of each forward and reverse 18S DNA primer were 900 nM, and final concentration of the dual-labeled probe was 200 nM. Sequences of the 18S primers were forward: 5′-CGGCTACCACATCCAAGGAA-3′; reverse: 5′-GCTGGAATTACCGCGGCT-3′; probe: 5′-VIC-TGCTGGCACCAGACTTGCCCTC-TAMRA-3′. The HTLV-1 tax gene standard was constructed by subcloning a 161 base pair fragment (nucleotides 6985 to 7145, NCBI accession no. AF033 817) from the pACH HTLV-1 proviral clone25 into the pCR 4-TOPO vector (Invitrogen, Carlsbad, CA). Standard curves of the HTLV-1 tax gene or 18S DNA endogenous control were included on the same optic plate as the unknown test specimen samples. For each run, a standard curve was generated from triplicate samples of log-10 dilutions of plasmid DNA in DNAse/RNAse-free water. The lower limit of detection was estimated to be 82 copies per microgram of DNA. The HTLV-1 proviral copy number per 1 μg genomic DNA was determined, corrected for amplification efficiency, and normalized to the copy number of 18S DNA determined for each sample. The normalized copy number per cell was calculated using the following formula: copy number per cell = (copy number / 1μg DNA) / (134 600 cells / 1 μg DNA).
Rabbit inoculation
To test the in vivo replication capacity of each cell line, 12-week-old female specific pathogen-free New Zealand white rabbits (Harlan, Indianapolis, IN) were inoculated via the lateral ear vein. Inocula were equilibrated by comparing viral p19 protein production per cell measured by ELISA as described in “Detection of viral p19 matrix antigen.” Inoculations were performed in 2 experiments. In the first, a total of 107 ACH.1 cells (n = 2), ACH.95 cells (n = 2), or uninfected PBMCs (n = 1) were inoculated (Table 1). In the second experiment, a total of 107 ACH.1 cells (n = 1), 107 ACH.75 cells (n = 4), 1.43 × 107 ACH.95 cells (n = 2), or 1.35 × 107 ACH.195 cells (n = 4) were inoculated (Table 1). All cells were gamma irradiated (75 Gy) prior to injection to prevent outgrowth of the cellular inoculum in vivo but allow virus transmission.27
Rabbit groups and inocula
Rabbit . | Inoculum type* . | No. of cells, × 107 . |
|---|---|---|
| R1 | ACH.95 | 1 |
| R2 | ACH.95 | 1 |
| R16 | ACH.95 | 1.43 |
| R17 | ACH.95 | 1.43 |
| R11 | ACH.75 | 1 |
| R12 | ACH.75 | 1 |
| R13 | ACH.75 | 1 |
| R14 | ACH.75 | 1 |
| R18 | ACH.195 | 1.35 |
| R19 | ACH.195 | 1.35 |
| R20 | ACH.195 | 1.35 |
| R21 | ACH.195 | 1.35 |
| R5 | ACH.1 | 1 |
| R10 | ACH.1 | 1 |
| R22 | ACH.1 | 1 |
| R9 | PBMC | 1 |
Rabbit . | Inoculum type* . | No. of cells, × 107 . |
|---|---|---|
| R1 | ACH.95 | 1 |
| R2 | ACH.95 | 1 |
| R16 | ACH.95 | 1.43 |
| R17 | ACH.95 | 1.43 |
| R11 | ACH.75 | 1 |
| R12 | ACH.75 | 1 |
| R13 | ACH.75 | 1 |
| R14 | ACH.75 | 1 |
| R18 | ACH.195 | 1.35 |
| R19 | ACH.195 | 1.35 |
| R20 | ACH.195 | 1.35 |
| R21 | ACH.195 | 1.35 |
| R5 | ACH.1 | 1 |
| R10 | ACH.1 | 1 |
| R22 | ACH.1 | 1 |
| R9 | PBMC | 1 |
Twelve-week-old specific pathogen-free New Zealand white rabbits were inoculated via the lateral ear vein as described in “Materials and methods.” ACH.95, ACH.75, ACH.195, and ACH.1 cell lines were obtained by outgrowth of immortalized PBMCs previously transfected with the full-length HTLV-1 molecular clones ACH.95, ACH.75, ACH.195, and ACH, respectively.30,31 Inocula were equilibrated by p19 gag production on a per-cell basis prior to inoculation
Serologic and clinical analyses
The plasma antibody response to HTLV-1 in inoculated rabbits was determined with a commercial ELISA with solid phase consisting of purified HTLV-1 viral lysate and recombinant HTLV-1 TM (p21) antigen (BioMerieux, Durham, NC), which was adapted for use with rabbit plasma by substitution of horseradish peroxidase-conjugated goat anti-rabbit immunoglobulin G (1:3000 dilution; Chemicon, Temecula, CA). Plasma was diluted 1:12 000 to obtain values in the linear range of the assay, and data were expressed as absorbance values. Absorbance values greater than or equal to 3 standard deviations above the average absorbance value for all week 0 samples were considered to indicate a positive antibody titer. Estimated specificity according to the manufacturer is more than 99%. Reactivity to specific viral antigenic determinants was detected with a commercial HTLV-1 Western blot assay (Genelabs Diagnostics, Singapore, Republic of Singapore) adapted for rabbit plasma by use of alkaline phosphatase-conjugated goat anti-rabbit immunoglobulin G (1:1000 dilution; BioMerieux). Plasma showing reactivity to Gag (p24 or p19) and Env (p21 or gp46) antigens was classified as positive for HTLV-1 seroreactivity. For determination of positive and negative antibody responses, plasma was diluted at 1:100. In order to quantitate antibody responses, densitometry was performed on Western blots using a commercial software package (AlphaEase; Alpha Innotech, San Leandro, CA). Because plasma dilutions of 1:100 saturated the Western blot detection system, Western blots were run at plasma dilutions of 1:2000 for densitometry analysis. Rabbits were regularly evaluated for any overt signs of clinical disease. Rabbits were euthanized and necropsied at a postinoculation interval of 8 weeks.
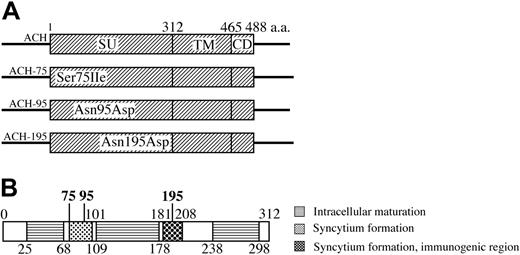
Construction of the ACH-envelope clones. (A) ACH.75, ACH.95, and ACH.195 contain the env gene of the HTE mutants20 between an SphI site and an NsiI site in the ACH molecular clone. The point mutants are designated by the wild-type amino acid, the position (in comparison to initiator methionine), and the mutant amino acid. SU indicates surface protein; TM, transmembrane protein; CD, cytoplasmic domain. (B) HTLV-1 SU protein. Schematic summary of functional domains of SU.10,14-22 The position of amino acid mutants examined in this study is indicated in bold.
Construction of the ACH-envelope clones. (A) ACH.75, ACH.95, and ACH.195 contain the env gene of the HTE mutants20 between an SphI site and an NsiI site in the ACH molecular clone. The point mutants are designated by the wild-type amino acid, the position (in comparison to initiator methionine), and the mutant amino acid. SU indicates surface protein; TM, transmembrane protein; CD, cytoplasmic domain. (B) HTLV-1 SU protein. Schematic summary of functional domains of SU.10,14-22 The position of amino acid mutants examined in this study is indicated in bold.
Results
In vitro analysis of viral clones
We have reported that the amino acid point mutations Ser75Ile, Asn95Asp, and Asn195Asp within the Env SU protein do not affect the ability of the virus to immortalize PBMCs in vitro.30 ACH Env SU point mutants contain the env genes of HTLV-1 envelope mutants from the HTE series20 inserted between the SphI site at position 5121 and the NsiI site at position 6565 in the ACH clone25,30 (Figure 1). To further investigate the effects of these point mutations, we developed immortalized human T-cell lines that continually produce either wild-type HTLV-1 (ACH.1) or HTLV-1 containing point mutations Ser75Ile, Asn95Asp, or Asn195Asp in the Env SU protein (ACH.75, ACH.95, ACH.195, respectively). As we have reported, these cell lines were representative of the phenotype of T cells immortalized by HTLV-1 and had typical expression profiles of CD4 and CD8.30,31 To ensure that the mutation was present prior to inoculation, a region of env containing the mutation site was amplified by PCR from each of the ACH.1, ACH.75, ACH.95, and ACH.195 cell lines. The amplified fragment was sequenced. Sequencing results confirmed the presence of the expected mutation in each cell line.
Serologic response of rabbits to viral clones
To evaluate the effects of point mutations within Env SU in vivo, we compared the abilities of the ACH.1, ACH.75, ACH.95, and ACH.195 cell lines to establish and maintain infection in our rabbit model. To ensure comparable infection potentials, inocula were equilibrated by HTLV-1 p19 antigen production on a per-cell basis (Table 1). Prior to inoculation, the fidelity of the mutations was confirmed by sequencing. Three rabbits were inoculated with the ACH.1 cell line. For each of the ACH.75, ACH.95, and ACH.195 cell lines, a group of 4 rabbits was inoculated. One rabbit was inoculated with uninfected PBMCs as a negative control.
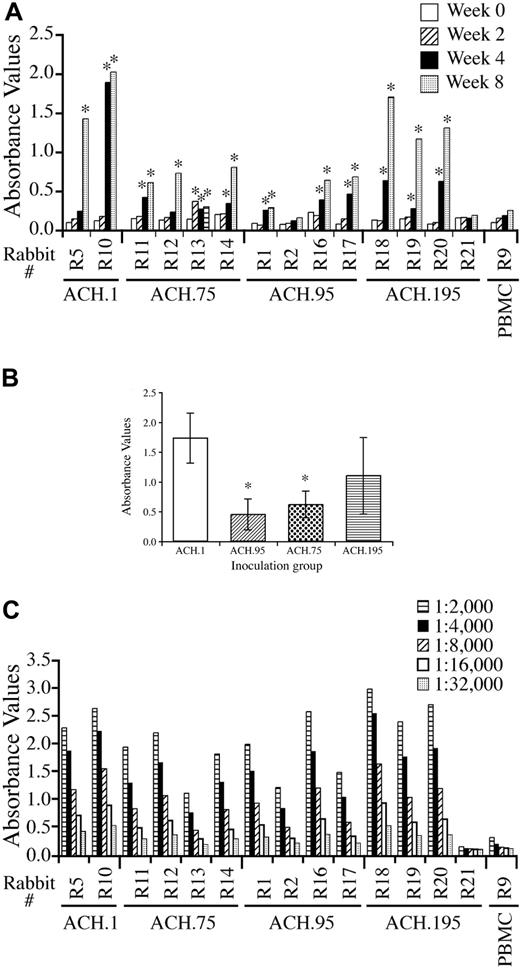
Serologic response of rabbits to the inocula was determined by measuring titers of antibody directed against purified HTLV-1 viral lysate and recombinant TM (p21) protein by ELISA (Figure 2A). Responses were assayed from each rabbit's serum at 0, 2, 4, and 8 weeks after inoculation. By week 4, a positive antibody titer was detected in 1 of 2 ACH.1-inoculated rabbits, and 3 of 4 rabbits in each of the ACH.75-, ACH.95-, and ACH.195-inoculated groups. All rabbits developed a positive antibody titer by week 8 except 1 ACH.95-inoculated rabbit and 1 ACH.195-inoculated rabbit. Pair-wise comparisons between each inoculum group using a Bonferroni adjustment for multiple comparisons with an α level of 0.05 did not reveal a statistically significant difference in ELISA absorbance values between any 2 inoculum groups at week 4. Using a similar analysis on week 8 samples (1:12 000 dilution), pair-wise comparisons showed statistically significant lower absorbance values in the ACH.95 group compared with the ACH.1 group (P = .034) and borderline statistically significant lower absorbance values in the ACH.75 group compared with the ACH.1 group (P = .107; Figure 2B). Serial dilution (1:2000 to 1:32 000) analysis indicated similar patterns of serologic reactivity (Figure 2C).
ELISA absorbance values on serum samples. (A) ACH represents a group of 2 animals. ACH.75, ACH.95, and ACH.195 represent a group of 4 animals. PBMC represents a negative control rabbit inoculated with uninfected PBMCs. The data shown are absorbance values from plasma diluted 1:12 000 and determined by anti-HTLV-1 antibody ELISA. *Positive antibody titer. (B) The data shown are from the week-8 serum samples. A statistically significant lower antibody titer was seen in ACH.95- and ACH.75-inoculated rabbits compared with wild-type ACH.1-inoculated rabbits (pair-wise comparisons with Bonferroni adjustment, P = .034 and P = .107, respectively). (C) Serial dilution (1:2000 to 1:32 000) comparison of week-8 serum samples from rabbits.
ELISA absorbance values on serum samples. (A) ACH represents a group of 2 animals. ACH.75, ACH.95, and ACH.195 represent a group of 4 animals. PBMC represents a negative control rabbit inoculated with uninfected PBMCs. The data shown are absorbance values from plasma diluted 1:12 000 and determined by anti-HTLV-1 antibody ELISA. *Positive antibody titer. (B) The data shown are from the week-8 serum samples. A statistically significant lower antibody titer was seen in ACH.95- and ACH.75-inoculated rabbits compared with wild-type ACH.1-inoculated rabbits (pair-wise comparisons with Bonferroni adjustment, P = .034 and P = .107, respectively). (C) Serial dilution (1:2000 to 1:32 000) comparison of week-8 serum samples from rabbits.
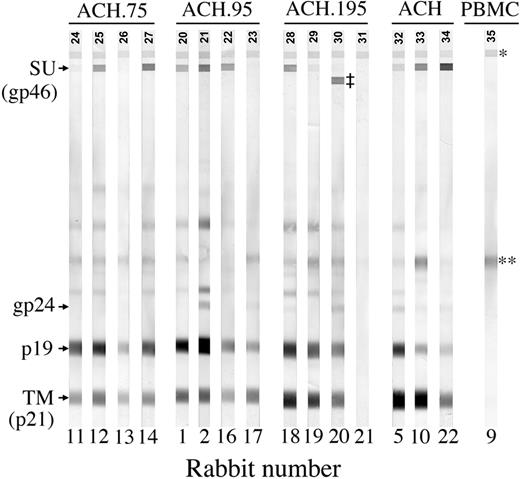
HTLV-1-specific antibody response by Western blot. Data represents 1:2000 dilutions of week-8 postinoculation plasma. R11, R12, R13, and R14 were inoculated with ACH.75. R1, R2, R16, and R17 were inoculated with ACH.95. R18, R19, R20, and R21 were inoculated with ACH.195. R5, R10, and R22 were inoculated with ACH.1. R9 was inoculated with uninfected PBMCs as a negative control. *Serum control band; **cellular antigen; ‡HTLV-2 SU band.
HTLV-1-specific antibody response by Western blot. Data represents 1:2000 dilutions of week-8 postinoculation plasma. R11, R12, R13, and R14 were inoculated with ACH.75. R1, R2, R16, and R17 were inoculated with ACH.95. R18, R19, R20, and R21 were inoculated with ACH.195. R5, R10, and R22 were inoculated with ACH.1. R9 was inoculated with uninfected PBMCs as a negative control. *Serum control band; **cellular antigen; ‡HTLV-2 SU band.
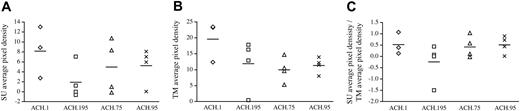
Semiquantitative analysis of HTLV-1 SU- and TM-specific antibody responses. Densitometry analysis was performed on the Western blots shown in Figure 3. (A) SU average pixel density. The ACH.195 SU average pixel density was lower than that seen in other inoculation groups. (B) TM average pixel density. The ACH.195 TM average pixel density was similar to that seen in other inoculation groups. (C) SU average pixel density/TM average pixel density. ACH.195 rabbits mounted a lower SU antibody response relative to their TM antibody response compared with rabbits in other inoculation groups.
Semiquantitative analysis of HTLV-1 SU- and TM-specific antibody responses. Densitometry analysis was performed on the Western blots shown in Figure 3. (A) SU average pixel density. The ACH.195 SU average pixel density was lower than that seen in other inoculation groups. (B) TM average pixel density. The ACH.195 TM average pixel density was similar to that seen in other inoculation groups. (C) SU average pixel density/TM average pixel density. ACH.195 rabbits mounted a lower SU antibody response relative to their TM antibody response compared with rabbits in other inoculation groups.
Reactivity to specific HTLV-1 antigens was confirmed at 0, 2, 4, and 8 weeks after inoculation by Western blot analysis with plasma dilution of 1:100 (Table 2). With the exception of 1 ACH.195 inoculated rabbit (R21), all rabbits showed strong antibody responses by week 4 after inoculation. In R21, we were able to detect a weak antibody response to the TM protein transiently at week 2. Interestingly, in another ACH.195-inoculated rabbit (R20), in addition to strong antibody responses against HTLV-1 Gag and Env TM proteins and a weak antibody response to HTLV-1 Env SU protein, we were also able to detect a strong antibody response to HTLV-2 SU (Figure 3). Because the plasma dilution of 1:100 saturated our detection system, we repeated the Western blots with a 1:2000 plasma dilution followed by densitometry analysis in order to detect differences in reactivity to Env proteins between the inoculation groups (Figure 4). Interestingly, results showed that compared with other inoculation groups, the ACH.195 inoculation group mounted a weaker antibody response to the SU protein as evidenced by both a lower SU average pixel density and a lower SU/TM average pixel-density ratio.
Western blot assay summary of antibody response to HTLV-1 antigens
. | Antibody responses . | . | . | . | . | ||||
|---|---|---|---|---|---|---|---|---|---|
| Inoculum . | Rabbit . | Week 0 . | Week 2 . | Week 4 . | Week 8 . | ||||
| ACH.95 | R1 | – | + | +++ | +++ | ||||
| ACH.95 | R2 | – | + | +++ | +++ | ||||
| ACH.95 | R16 | – | +++ | +++ | +++ | ||||
| ACH.95 | R17 | ∥ | +++ | +++ | +++ | ||||
| ACH.75 | R11 | – | +++ | +++ | +++ | ||||
| ACH.75 | R12 | – | +++ | +++ | +++ | ||||
| ACH.75 | R13 | – | + | +++ | +++ | ||||
| ACH.75 | R14 | – | +++ | +++ | +++ | ||||
| ACH.195 | R18 | – | +++ | +++ | +++ | ||||
| ACH.195 | R19 | – | * | +++ | +++ | ||||
| ACH.195 | R20 | – | + | +++§ | +++§ | ||||
| ACH.195 | R21 | – | * | – | – | ||||
| ACH.1 | R5 | – | + | +++ | +++ | ||||
| ACH.1 | R10 | – | * | +++ | +++ | ||||
| ACH.1 | R22 | – | +++ | +++ | +++ | ||||
| PBMC | R9 | – | – | – | – | ||||
. | Antibody responses . | . | . | . | . | ||||
|---|---|---|---|---|---|---|---|---|---|
| Inoculum . | Rabbit . | Week 0 . | Week 2 . | Week 4 . | Week 8 . | ||||
| ACH.95 | R1 | – | + | +++ | +++ | ||||
| ACH.95 | R2 | – | + | +++ | +++ | ||||
| ACH.95 | R16 | – | +++ | +++ | +++ | ||||
| ACH.95 | R17 | ∥ | +++ | +++ | +++ | ||||
| ACH.75 | R11 | – | +++ | +++ | +++ | ||||
| ACH.75 | R12 | – | +++ | +++ | +++ | ||||
| ACH.75 | R13 | – | + | +++ | +++ | ||||
| ACH.75 | R14 | – | +++ | +++ | +++ | ||||
| ACH.195 | R18 | – | +++ | +++ | +++ | ||||
| ACH.195 | R19 | – | * | +++ | +++ | ||||
| ACH.195 | R20 | – | + | +++§ | +++§ | ||||
| ACH.195 | R21 | – | * | – | – | ||||
| ACH.1 | R5 | – | + | +++ | +++ | ||||
| ACH.1 | R10 | – | * | +++ | +++ | ||||
| ACH.1 | R22 | – | +++ | +++ | +++ | ||||
| PBMC | R9 | – | – | – | – | ||||
– indicates no response; +++, strong response (strong reactivity to both Gag and Env antigens); +, weak response (weak reactivity to both Gag and Env antigens).
Indeterminate response (reactivity to only 1 Gag or Env antigen)
Antibody response to HTLV-2 SU
Slight response to Env TM was seen and was interpreted as a cross-reaction due to the high concentration of serum (1:100) in the reaction
Proviral loads
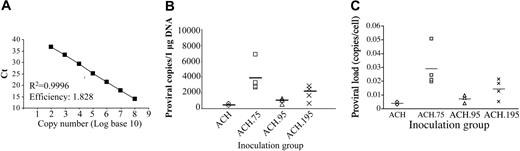
To measure the ability of each HTLV-1 clone to maintain viral loads in vivo, we determined the number of provirus copies per cell in rabbit PBMCs by real-time PCR for all rabbits at 8 weeks after inoculation. The proviral load per 1 μg genomic PBMC DNA was determined at 8 weeks after inocuation (Figure 5B) using the standard curve shown in Figure 5A. The average proviral copy numbers per 1 μg PBMC DNA for ACH.1, ACH.75, ACH.95, and ACH.195 were 523, 3908, 957, and 1932, respectively. The proviral load per PBMC was calculated and the mean values for ACH.1, ACH.75, ACH.95, and ACH.195 were 0.0039, 0.0290, 0.0071, and 0.0143, respectively (Figure 5C). The negative control rabbit inoculated with uninfected PBMCs had no detectable proviral load. The overall t test (analysis of variance [ANOVA] test) yielded a significant group effect (P < .001). Pairwise comparisons using Bonferroni to adjust for multiplicity were performed using an overall α-level of 0.05. The ACH.75-inoculated rabbits had statistically significant higher proviral loads compared with ACH.1- and ACH.95-inoculated rabbits (P = .009 and P = .037, respectively). There was no statistically significant difference in proviral loads between ACH.95-, ACH.195-, and ACH.1-inoculated rabbits. The ACH.195-inoculated rabbit, R21, which did not have a detectable antibody response to HTLV-1 structural antigens, had low but detectable proviral loads (0.0052 copies per cell). Interestingly, other rabbits such as ACH.95-inoculated R16 and R17 and ACH.1-inoculated R5 and R10 had similar or lower proviral loads as R21 but mounted strong antibody responses (Figure 5C).
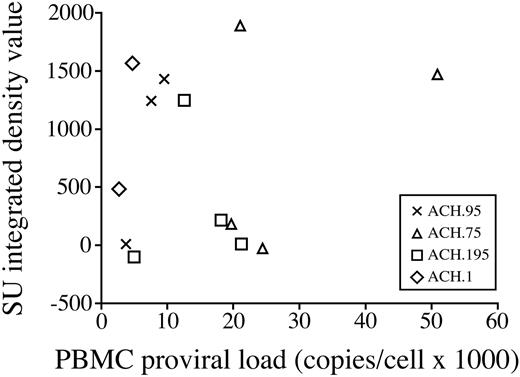
In order to determine if there is a relationship between the antibody response to SU and PBMC proviral loads, we performed correlation analysis of PBMC proviral loads against the Western blot densitometry values obtained for the SU protein (Figure 6). No correlation was found between PBMC proviral loads and SU densitometry values (Spearman correlation coefficient 0.19).
Sequence and restriction enzyme analysis
For each rabbit week-8 PBMC sample, we extracted DNA and sequenced the PCR products derived from env sequence-specific primer pairs across the region spanning the mutations to ensure that there were no unexpected mutations or reversions within env. Sequence analysis showed that all env mutations were maintained in all rabbits through the 8-week postinoculation time point.
Proviral loads in week-8 PBMCs. (A) The HTLV-1 tax gene standard curve was derived by plotting the log copy number versus the mean crossing point (Ct). (B) DNA was extracted from rabbit PBMCs at 8 weeks after inoculation. Proviral loads per 1 μg PBMC DNA were determined with real-time PCR as described in “Materials and methods.” (C) The proviral load per PMBC was calculated as described in “Materials and methods.” Proviral loads in ACH.75-inoculated rabbits were statistically higher compared with ACH.1- and ACH.95-inoculated rabbits (pair-wise comparisons with Bonferroni adjustment, P = .009 and P = .037, respectively).
Proviral loads in week-8 PBMCs. (A) The HTLV-1 tax gene standard curve was derived by plotting the log copy number versus the mean crossing point (Ct). (B) DNA was extracted from rabbit PBMCs at 8 weeks after inoculation. Proviral loads per 1 μg PBMC DNA were determined with real-time PCR as described in “Materials and methods.” (C) The proviral load per PMBC was calculated as described in “Materials and methods.” Proviral loads in ACH.75-inoculated rabbits were statistically higher compared with ACH.1- and ACH.95-inoculated rabbits (pair-wise comparisons with Bonferroni adjustment, P = .009 and P = .037, respectively).
PBMC proviral loads do not correlate with antibody response to SU. PBMC proviral loads were analyzed for correlation with degree of antibody response to SU. No correlation was found (Spearman correlation coefficient 0.19).
PBMC proviral loads do not correlate with antibody response to SU. PBMC proviral loads were analyzed for correlation with degree of antibody response to SU. No correlation was found (Spearman correlation coefficient 0.19).
Discussion
Although numerous studies have addressed the effects of point mutations in the HTLV-1 env gene in an in vitro setting,10,14,20-22,30,33 no studies to date have correlated in vitro findings to the replication capacity or immunogenicity of the virus in vivo. In this study, we expanded the findings of Tsukahara et al,30 who demonstrated that the amino acid point mutations Ser75Ile, Asn95Asp, and Asn195Asp within the Env SU protein do not alter the ability of HTLV-1 to infect and immortalize lymphocytes in vitro. At 2 months after exposure, all ACH.75-, ACH.95-, ACH.195-, and wild-type ACH.1-inoculated rabbits were infected with HTLV-1 with the fidelity of the mutation maintained. ACH.75 and ACH.95 rabbits had decreased antibody response to purified HTLV-1 viral lysate and recombinant TM (p21) by ELISA compared with wild-type ACH.1 rabbits. With the exception of one ACH.195 rabbit, all rabbits mounted strong antibody responses by 8 weeks after inoculation. Interestingly, one ACH.195 rabbit had no detectable antibody response by ELISA and Western blot, but proviral loads were comparable to rabbits that mounted strong antibody responses. Western blot and densitometry analysis indicated that the ACH.195 group mounted a weaker antibody response to the SU protein compared with all other groups. Within the ACH.195-inoculated group, a single rabbit also mounted an antibody response to the HTLV-2 SU protein. ACH.75 rabbit PBMCs had higher proviral loads than the ACH.1 and the ACH.95 groups. Interestingly, correlation analysis did not reveal a relationship between PBMC proviral loads and the antibody response to HTLV-1 SU.
In this study, we confirmed that mutations in HTLV-1 SU, which were tolerated by the virus in in vitro lymphocyte immortalization assays, do not alter the ability of HTLV-1 to replicate, adding validity to in vitro lymphocyte immortalization assays. The ACH.195 inoculation group was the most variable in its antibody responses, which is consistent with data from previous in vitro syncytium and infectivity inhibition studies, peptide-binding ELISA studies, and in vivo peptide immunization studies, which implicate this amino acid to be in a highly immunogenic region.14-19 Serologic studies of HTLV-1-infected humans have clearly implicated amino acid 195 to lie within a region known to elicit neutralizing antibodies during HTLV-1 infection.15-18 In addition, in vivo vaccine studies in animals have indicated that synthetic peptides spanning the region containing amino acid 195 have the potential to protect against HTLV-1 infection in rabbits.19,34 However, in this study, we found no correlation between anti-Env antibody responses and proviral loads, indicating that a robust initial antibody response to Env did not correlate with increased protection from infection. Within humans, during the clinically asymptomatic HTLV-1 infection, proviral loads in PBMCs range from 0.04 to 0.001 per cell.35-39 With the exception of one ACH.75-inoculated rabbit, all rabbit PMBC proviral loads in our study were within this range, indicating that our rabbit model closely mimics the natural asymptomatic infection common in most HTLV-1-infected humans.
Two rabbits within the ACH.195 inoculation group were of particular interest in our study. R21 had a PBMC proviral load consistent with that seen within asymptomatic HTLV-1-infected individuals. However, with the exception of a transient antibody response to the TM protein at week 2, we were unable to detect an antibody response in this rabbit. It is likely that this animal did mount an antibody response, but that it was below the level of our assay detection sensitivity. The serologic and PCR results in this animal were consistent with reports of rare HTLV-1-infected individuals who are serologically negative while PCR positive for the virus in their PBMCs.40-42 Another rabbit of interest in the ACH.195 group was R20, which mounted an antibody response not only to HTLV-1 Gag and Env proteins, but also to the HTLV-2 SU protein. In this rabbit, the antibody response to HTLV-2 SU was stronger than that mounted against HTLV-1 SU. That a single amino acid change in the HTLV-1 SU could result in a seropositive result for HTLV-2 in an animal that clearly was not infected with HTLV-2 has interesting implications for serologic screening in humans.42-44
The HTLV-1 env region maintains a high degree of sequence conservation during the in vivo infection, with variability ranging from 1% to 8%.5-10 There are various reports of single base pair changes in the env sequence in HTLV-1 isolates from infected individuals.5,45 We are not aware that the amino acid changes made in this study have been demonstrated as common variants in the human infection. Given the low degree of env sequence variation found in humans, it leads one to predict that the mutations would not be tolerated in vivo. However, studies on the in vivo expansion of HTLV-1 have demonstrated that HTLV-1 spreads in vivo mainly by the relatively error-free process of clonal expansion of infected lymphocytes rather than by the error-prone process of reverse transcription,46,47 and it may be that the absence of sequence variation is due more to an absence of the need to undergo reverse transcription rather than an inability for sequence variants to maintain infection. The maintenance of single point mutations in Env over the 8-week course of our study in rabbits is more supportive of clonal expansion of infected lymphocytes versus cell-free infection.
Future studies are needed to further define how specific mutations (eg, ACH.195) alter the in vivo pathogenesis of HTLV-1. We believe that our current study adds validity to both the use of in vitro lymphocyte immortalization assays as a model to study HTLV-1 Env, as well as the rabbit as a model of HTLV-1 infection. Additionally, this study provides further evidence to implicate the region spanning Env amino acid 195 to be highly immunogenic during the in vivo infection.
Prepublished online as Blood First Edition Paper, July 26, 2005; DOI 10.1182/blood-2005-03-1076.
Supported by National Institutes of Health grants CA-100 730 (M.D.L.), CA-63 417 (L.R.), and CA-70 529 and CA-09 338 (awarded through the Ohio State University Comprehensive Cancer Center).
The publication costs of this article were defrayed in part by page charge payment. Therefore, and solely to indicate this fact, this article is hereby marked “advertisement” in accordance with 18 U.S.C. section 1734.
We thank Tim Vojt for preparation of figures.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal