Abstract
Herpes simplex viruses (HSV) infect human and murine dendritic cells (DCs) and interfere with their immunostimulatory functions in culture. HSV-2 infection increases human immunodeficiency virus (HIV) spread in patients, and DCs also promote HIV infection. We have studied these topics in rhesus macaque monocyte-derived DCs (moDCs) to set the stage for future studies of these issues in animals. We provide the first evidence that macaque DCs become infected by HSV-2. Structural viral proteins (ICP5 [infected cell protein 5], glycoprotein D [gD], envelope) were detected in the cell periphery, and a functional protein (infected cell protein 8 [ICP8]) was predominantly found in the nucleus after infection. Infectious HSV-2 induced apoptotic death, decreased expression of HLA-DR, CD40, CD80, CD83, and CD86, and increased release of interleukin-6 (IL-6), tumor necrosis factor-α (TNF-α), macrophage inflammatory protein-1α (MIP-1α) (CCL3), and RANTES (regulated on activation normal T cells expressed and secreted) (CCL5) but not IL-12 or interferon-α (IFN-α) by macaque DCs. This coincided with HSV-2–infected DCs stimulating weak T-cell responses, including impaired SIV-specific responses. Comparable HSV-2 protein expression, DC apoptosis, as well as membrane immunophenotype and functional modifications were observed in HSV-2–exposed human moDCs. Such HSV-2–induced modifications of macaque and human DCs could augment DC-driven immunodeficiency virus infection. This work affords the basis for future macaque studies to explore how HSV-2 impacts the efficacy of strategies being developed to prevent HIV transmission.
Introduction
Dendritic cells (DCs) are involved in innate and adaptive immunity.1-5 DCs survey for pathogens to which they respond innately while also processing pathogens and presenting antigenic determinants to induce adaptive immune responses. DCs need to be activated or matured to stimulate potent adaptive immunity.6 Maturation involves the up-regulation of molecules on the DC surface and secretion of cytokines and chemokines that encourage the DC–T cell interactions needed to elicit strong immunity. Many pathogens trigger these pathways, modifying DC functions to encourage effective immune activation and clearance of infection.7-10 Yet, other pathogens like immunodeficiency viruses (human [HIV] and simian [SIV])11-15 and herpes simplex viruses (HSV)16-18 exploit DC biology to facilitate infection and elicit immune responses incapable of preventing or eradicating infection. Moreover, there is a strong correlation between genital HSV (HSV-2) infection and the probability of acquiring HIV.19 Understanding how this might be orchestrated at the DC level is central to developing strategies to prevent DC-driven HIV spread.12
Primary HSV-2 infection, occurring at the mucosal surfaces, is typically followed by the establishment of latency in the sacral root ganglia.20 Neutralizing antibodies (Abs) and antiviral CD4+ and CD8+ T cells are induced,20 which ultimately restrict virus replication at local sites to resolve (primary and reactivated) lesions.21,22 DCs likely play a key role by orchestrating responses to HSV,21,23-25 although low-level productive infection of DCs might also contribute to virus spread.18 HSV infection of human monocyte-derived DCs (moDCs) is cytopathic23,26,27 and results in the down-modulation of several surface markers involved in the activation of T cells.16-18 While this would result in mediocre anti-HSV immunity allowing the establishment of HSV infection (but not preventing infection), it must be sufficient to clear virus upon reactivation.24,25 Herpetic lesions also comprise activated leukocyte infiltrates and enable direct blood contact, providing mechanisms for increased HIV spread. It is possible that HSV-2 infection of immature DCs additionally alters innate DC responses and compromises the ability of DCs to elicit potent adaptive responses to other pathogens, thereby further exacerbating HIV infection.
The initial step toward dissecting this biology in a relevant animal model was to validate HSV-2 infection of macaque DCs. We provide the first proof that immature macaque moDCs can be infected by HSV-2. We observed DC apoptosis upon exposure to infectious HSV-2 as well as reduced immunostimulatory functions in HSV-2–infected DCs. Notably, HSV-2–infected DCs stimulated weaker SIV-specific T-cell responses. These data will enable future studies in the macaque to explore how HSV-2 infection affects DC-driven HIV transmission and if the presence of coinfections, like HSV-2, influences the efficacy of preventative or therapeutic approaches.
Materials and methods
Animals
Adult rhesus macaques (Macaca mulatta) were bred and housed at the Tulane National Primate Research Center ([TNPRC] Covington, LA). At the commencement of these studies all naive animals tested negative by polymerase chain reaction (PCR) for simian type D retroviruses, simian T-cell leukemia virus, and SIV. Healthy SHIV162P-infected macaques (infected about 6 years ago) were used as a source of virus-primed T cells for SIV-specific immune assays.15 Protocols were reviewed and approved by the Institutional Animal Protocols Care and Use Committee of the TNPRC. Animal care procedures were in compliance with the regulations detailed under the Animal Welfare Act28 and in the “Guide for the Care and Use of Laboratory Animals.”29
Generation of immature moDCs
Peripheral blood mononuclear cells were isolated from heparinized macaque blood or human leukopacks (New York Blood Center, New York, NY) using Ficoll-Hypaque density gradient centrifugation (Amersham Pharmacia Biotech, Uppsala, Sweden). CD14+ monocytes were isolated using the nonhuman or human primate CD14 magnetic cell sorting (MACS) system (Miltenyi Biotec, Auburn, CA) and moDCs generated by culturing these cells in 100 U/mL recombinant human interleukin-4 (IL-4) (R&D Systems, Minneapolis, MN) and 1000 U/mL recombinant human granulocyte-macrophage colony-stimulating factor (GM-CSF) (Berlex Laboratories, Montville, NJ).30,31 After 6 to 7 days, immature DCs were collected, an aliquot taken for flow cytometry, and the remainder used for HSV-2 infections. DC phenotype was assayed using fluorescein isothiocyanate (FITC)–conjugated anti–HLA-DR (clone L243; BD Biosciences, San Jose, CA) in combination with phycoerythrin (PE)–conjugated immunoglobulin G1 (IgG1) (clone X40, BD Biosciences Immunocytometry Systems, San Jose, CA), IgG2b (clone 27-35; BD Biosciences Pharmingen), anti-CD25 (clone 2A3; BD Biosciences Immunocytometry Systems), anti-CD80 (clone L307.4; BD Biosciences Immunocytometry Systems), anti-CD83 (clone HB15A; Immunotech, Marseille, France), or anti-CD86 (clone IT2.2; BD Biosciences Pharmingen).
Culture medium (R1) was RPMI 1640 (Cellgro; Fisher Scientific, Springfield, NJ) containing 2 mM L-glutamine (GIBCO Life Technologies, Grand Island, NY), 10 mM HEPES (N-2-hydroxyethylpiperazine-N′-2-ethanesulfonic acid) (GIBCO Life Technologies), 50 μM 2-mercaptoethanol (Sigma Chemical, St Louis, MO), penicillin (100 U/mL) and streptomycin (100 μg/mL) (GIBCO Life Technologies), and 1% heparinized human plasma.
HSV-2 infection
Viral stocks were propagated in Vero cells (American Type Culture Collection [ATCC] Manassas, VA), titered by plaque formation on Vero cells,32 and aliquots stored at –80°C. Immature moDCs were exposed to HSV-2 (strain G; ATCC) at 1 or 5 plaque-forming units (pfu) per cell for 1 hour at 37°C. Controls were set up alongside where DCs were exposed to the medium in which the viral stocks were grown (Dulbecco modified Eagle medium [DMEM] containing 2% fetal bovine serum [FBS]). Typically, 106 cells were added to Eppendorf tubes (precoated with RPMI 1640 containing 10% fetal calf serum [FCS] on ice for 5 to 10 minutes) in a final volume of 100 μL R1 containing 1 to 5 pfu HSV-2 per cell. After incubation, cells were washed 5 times with 500 μL cold R1 (3 minutes, 3000 rpm) to remove unbound virus. Cells were recounted, resuspended in medium with 1000 U/mL GM-CSF and 100 U/mL IL-4, and plated at 0.5 × 106 to 1 × 106 cells per well of a 24-well plate (2 mL) or 2 × 105 to 5 × 105 cells per well of a 48-well plate (250 μL to 1 mL). UV-inactivated virus preparations were used to verify the effects of infectious virus. Aliquots (100 to 200 μL) of virus were plated in a 96-well flat-bottomed tray on ice and exposed to UV light (Blak Ray Lamp, Model UVL-21; UVP, Upland, CA) for 4 hours. Virus inactivation (4 to 5 log reduction in titer) was verified using the Vero cell plaque assay.
Detection of HSV-2 proteins by flow cytometry
DCs (1 × 106) were collected after reculture, washed with 500 μL of 1× Dulbecco phosphate-buffered saline (DPBS) containing 2% FCS (Cellgro; Fisher Scientific), resuspended in 250 μL per tube of Fixation/Permeabilization Solution (BD Biosciences Pharmingen), and incubated for 20 minutes at 4°C in the dark. Cells were washed twice with 500 μLof1 × Perm/Wash Buffer (BD Biosciences), resuspended in the same buffer, and plated into a 96-well V-bottomed tray (about 1 × 105 cells per well). Cells were stained with 1 μg/mL anti–HSV-2 ICP8 monoclonal antibody (mAb) (IgG2a isotype; Virusys, North Berwick, ME) or 1 μg/mL mouse IgG2a isotype control (R&D Systems). After 30 minutes of incubation (room temperature in the dark), cells were washed 4 times with 1 × DPBS (100 μL per well). Bound Abs were detected with biotin-SP (Spacer)–conjugated AffiniPure Donkey Anti-mouse IgG (5 μg/mL; Jackson ImmunoResearch Labs, West Grove, PA) and incubated for 30 minutes at room temperature in the dark. Cells were then washed (as above). Cellbound biotinylated Abs were detected by adding Qdot 525 streptavidin conjugate (10 nM final concentration; Quantum Dot, Hayward, CA) and incubated (30 minutes at room temperature).
For double staining with PE-conjugated Abs to monitor DC phenotype, the cells were then incubated (15 minutes, 4°C) with 5% normal mouse serum (Jackson ImmunoResearch Labs) before being incubated with PE-conjugated IgG2a (BD), anti-IgG2b (BD Pharmingen), anti–HLA-DR (BD), anti-CD40 (BD Pharmingen), anti-CD80, anti-CD83, or anti-CD86 for 30 minutes (4°C in the dark). Cells were washed 4 times with 1 × DPBS. Ab dilutions were made in the Qdot incubation buffer (Quantum Dot). The cells were acquired on a FACSCalibur flow cytometer (BD Biosciences) and data analyzed using FlowJo software (Tree Star, Ashland, OR).
Immunofluorescence microscopy
Recultured DCs were collected into 1.5 mL Eppendorf tubes, washed 4 times in serum-free RPMI 1640, and brought to 1 × 106 cells per mL; 25 μL (2.5 × 104 cells) was placed onto a spot of an Alcian blue–coated slide30,33 and allowed to adhere for 8 minutes at 4°C. After confirming cells had adhered, the supernatant was carefully aspirated and the adhered cells washed 5 times with 1 × DPBS. Cells were then fixed with a drop of freshly prepared 4% paraformaldehyde (PFA) (Electron Microscopy Sciences, Washington, PA) in 1 × DPBS (20 minutes, room temperature). PFA was carefully aspirated and cells washed 5 times with 1 × DPBS. To quench endogenous peroxidase activity, a drop of 0.3% H2O2 in 1 × DPBS (made fresh each time) was added to each spot of cells for 1 hour (room temperature). The slides were then washed 5 times with 1 × DPBS and the last drop of 1 × DPBS left on for 5 minutes; 1 × DPBS was then aspirated, and nonspecific binding was blocked with the blocking buffer (30 minutes, room temperature) (TSA Fluorescein System; Perkin Elmer, Boston, MA). Cells were washed 5 times with 1 × DPBS, and 25 μL of the diluted anti-HSV Abs was added to each spot of cells for 30 minutes (room temperature): 1 μg/mL anti–HSV-2 ICP8 and mouse IgG2a isotype control; 5 μg/mL anti-HSV ICP5 mAb (IgG1 isotype; Virusys), anti–HSV-2 glycoprotein D (gD) mAb (IgG1 isotype; Biodesign International, Saco, ME), anti–HSV-1 and -2 envelope glycoprotein mAb (IgG1 isotype; United States Biological, Swampscott, MA), and mouse IgG1 isotype control (R&D Systems). Cells were washed 5 times with 1 × DPBS before 25 μL of an 8 μg/mL solution of peroxidase-conjugated AffiniPure [Heavy + Light chain] Donkey Anti-Mouse IgG (H+L, Jackson ImmunoResearch Labs) was added and the slides incubated for 30 minutes (room temperature). After washing, bound peroxidase was then labeled with fluorescein tyramide, diluted 1:50 in the amplification diluent (TSA Fluorescein System) for 4 minutes (room temperature; 25 μL per spot). The TSA (Tyramide Signal Amplification) Fluorescein System amplifies signals allowing sensitive visualization of proteins by microscopy that are difficult to detect by other staining methods.33 Cells were washed 5 times with 1 × DPBS. Nuclei were stained with DAPI (4′, 6′-diamidino-2-phenylindole) (1.75 ng/mL, 25 μL per spot; Molecular Probes, Eugene, OR) for 2 minutes (room temperature) before the last 5 washes with 1 × DPBS. Slides were mounted in Aqua-Poly/Mount (Polysciences, Warrington, PA) and were examined using an Olympus AX70 epifluorescence microscope (Universal Imaging, West Chester, PA) with Plan Apo 60×/1.40 oil, DIC H, α/0.17, WD, 0.21 lenses (Nikon, Tokyo, Japan). The images were acquired using Image-Pro Plus 5.1 (MediaCybernetics, San Diego, CA) and analyzed using Photoshop 6.0 (Adobe Systems, San Jose, CA).
Apoptosis analysis
Untreated DCs and DCs exposed to infectious or UV-treated HSV-2 were stained with annexin V–FITC and propidium iodide (PI) (BD Biosciences Pharmingen). Cells were washed twice with cold 1 × DPBS and then resuspended in 1 × binding buffer (BD Biosciences Pharmingen) at a concentration of 1 × 106/mL. A total of 100 μL of the solution (1 × 105 cells) was left unstained or incubated with annexin V–FITC 1:20 and PI 1:20 for 15 minutes at room temperature in the dark. A total of 400 μL of 1 × binding buffer was added to each tube, and samples were analyzed by flow cytometry within 1 hour of labeling using a BD FACSCalibur.
Measurement of T-cell proliferation
Mixed leukocyte reactions (MLRs) were set up using human T cells.34 Immature moDCs (exposed or not to live versus UV-treated HSV-2) were collected after 4 hours of reculture and viable cells recounted and resuspended in fresh medium. Human T cells were prepared from leukopacks by depleting CD11b+CD14+CD16+HLA-DR+ cells (using Miltenyi MACS beads). T-cell purity was typically more than 95% as verified by fluorescence-activated cell sorter (FACS) analysis of cells stained with mAbs against CD3 versus HLA-DR, CD11b, CD14, CD16, or CD20. Triplicate cultures of 2 × 105 T cells mixed with different doses of macaque or allogeneic human DCs (input doses normalized as viable large cells/DCs) were set up in 96 well flat-bottomed trays (200 μL final). Controls of DCs (highest dose) and T cells cultured in medium alone were included. The plate was incubated for 5 days and then pulsed with tritiated thymidine (3H-TdR; 1 μCi [2.7 × 10–17 Bq] per well) for 8 hours before being harvested using a cell harvester (Brander, Gaithersburg, MD). The amount of 3H-TdR incorporated by the cells was measured using the Liquid Scintillation & Luminescence Counter, 1450 microbeta Wallac Jet (PerkinElmer Life and Analytical Sciences, Boston, MA).
Luminex and ELISA analysis of cytokines and chemokines
Cell-free tissue culture supernatants were collected from the MLRs (highest DC/T cell ratio) and the recultured DCs, spun at 1000 g for 3 minutes, and stored at –20°C prior to testing. Cytokine and chemokine levels were measured using the Beadlyte Human 22-plex Detection System according to the manufacturer's instructions (Upstate Biotechnology, Lake Placid, NY), washing the plates between steps with a MultiScreen Vacuum Manifold (Millipore, Billerica, MA). The plate was read on a Luminex 100 Instrument (Luminex, Austin, TX). Data were analyzed using STarStation software (Applied Cytometry System, Sacramento, CA). IL-12 levels were also monitored in the DC supernatants using the macaque-reactive IL-12 (p70 and p40) enzyme-linked immunosorbent assay (ELISA) (BioSource International, Camarillo, CA), and interferon-α (IFN-α) levels were measured using the monkey–cross-reactive Human IFN-α ELISA Kit (BioSource), according to the manufacturer's instructions.
SIV-specific assay
Blood from healthy, SHIV162P-infected animals was used as a source of SIV-primed T cells15 to monitor the presentation of SIV antigens by autologous DCs.15,31,34 Peripheral blood leukocytes were separated into CD14– and CD14+ fractions,31 and the CD14+ monocytes were cultured in GM-CSF/IL-4 to generate immature DCs (as described under “Generation of immature moDCs”). CD14– cells were cultured in R1 (at least 2 × 107/mL) with 5 mL fresh R1 being added to the CD14– cultures every 2 days when the DCs were fed with cytokines. On day 6, the DCs were collected and infected with 5 pfu per cell of HSV-2 or not (as described under “HSV-2 infection”). Medium-treated versus HSV-2–infected DCs were then recultured (2.5 × 105 DCs in 250 μL of medium with GM-CSF/IL-4 in a 48-well plate) for 4 hours at 37°C in the presence or absence of aldrithiol 2 (AT-2)–inactivated SIV E11S (30 ng p27 per 105 DCs).31,33,35,36 The DCs were collected 4 hours later, washed 5 times in medium, and the viable large cells recounted. Coincidently, the CD14– cells were collected and the T cells purified by depleting HLA-DR+CD11b+CD16+ cells (Miltenyi MACS system).
A range of doses of medium or AT-2 SIV-pulsed HSV-2–infected and uninfected DCs were plated with 2 × 105 autologous T cells in an anti-IFN-γ–coated enzyme-linked immunospot (ELISPOT) plate (triplicates per condition).15 T-cell and DC controls cultured alone were included. For each experiment, 2.5 × 103 DCs and 5 × 104 T cells were cultured with (or without) 1 μg/mL phytohemagglutinin-P (PHA-P; Sigma Chemical) versus medium to control for T-cell and DC functionality. Prior to developing the ELISPOT plates after overnight incubation,15 cell-free supernatants were collected. Supernatants were analyzed immediately or after storage at –20°C for the presence of additional soluble factors using the Luminex assay (as described under “Luminex and ELISA analysis of cytokines and chemokines”). ELISPOT plates were counted using an AID ELISPOT reader (Cell Technology, Columbia, MD) using once optimized settings.
Results
HSV-2 proteins are expressed by infected immature macaque DCs
HSV-2–exposed immature macaque DCs were analyzed immediately or after reculture for the expression of viral proteins (Figure 1). Within 4 hours of reculture, we observed cytoplasmic expression of the major HSV-2 capsid protein (ICP5) and gD as well as the products of the env gene (Figure 1A). The HSV-2 single-strand DNA binding protein, ICP8, was detected after overnight reculture of HSV-2–exposed DCs (Figure 1A) but not earlier (not shown). ICP8 was predominantly expressed in a nuclear pattern (Figure 1A-B), with points of more intense staining sometimes at the periphery of the nucleus. No HSV-2 proteins were detected immediately following the virus pulse (not shown), and all IgG controls were negative (Figure 1A, lower row).
Monitoring ICP8 expression as a stringent measure of infection, flow cytometry was used to quantify the numbers of ICP8+ DCs (Figure 1C-D). Unlike medium-treated DCs (less than 1% ICP8+), ICP8 expression was evident after infection and this increased with time (Figure 1C). This result was confirmed in 15 experiments using cells from 7 different animals (Figure 1D), averaging 4.36% ICP8+ (range, 1.43%-8.11%) after 4 hours and 14.69% ICP8+ (range, 6.37%-34.5%) after overnight reculture. HSV-2–exposed human moDCs showed similar ICP8 expression (Figure 1D), typically in a nuclear localization (not shown). Thus, exposure of immature macaque DCs to HSV-2 resulted in the increased expression of HSV-2 proteins over time, indicative of viral infection—not uptake of virus particles.
HSV-2 infection of macaque DCs is cytotoxic
HSV infection leads to apoptosis of DCs from other species.26,27,37 Microscopic examination of HSV-2–infected macaque DCs suggested that these cells were unhealthy. FACS data revealed that many of the HSV-2–exposed DCs, especially the ICP8+ cells, were smaller in size, based on their forward scatter profiles (Figure 1C), further indicative of poor cell health. There was a considerable reduction in the numbers of trypan blue–negative viable cells recovered after overnight culture (48.6% uninfected versus 22.13% infected; Figure 2A).
Cells were also stained with annexin V/PI to identify apoptotic and necrotic cells. Increases in the percentages of early apoptotic (annexin V–positive PI-negative; 58.9% versus 9.49%) as well as late apoptotic/necrotic (annexin V–positive PI-positive; 28.6% versus 10.1%) cells were evident in the HSV-2–infected DCs (versus medium-treated controls; Figure 2B). There was a slight increase in the percentage of apoptotic cells after 4 hours of reculture, but this was greater after overnight incubation of HSV-2–infected macaque DCs (average of 45.72% versus 16.66% in HSV-2–infected versus uninfected cells; Figure 2C). The percentages of cells in late apoptosis/necrosis increased even further over time (average of 82.15% versus 30.61% in HSV-2–infected versus uninfected cells; Figure 2C). This was due to infection, because exposure to the same amount of UV-inactivated HSV-2 did not induce DC death (similar to medium-exposed controls). This mirrors results for human DCs (Figure 2C).
HSV-2 infection of macaque DCs. Immature macaque and human moDCs were exposed to HSV-2 for 1 hour at 37°C (5 pfu per cell). HSV-2–loaded cells were then washed, counted, and plated for 4 hours up to overnight (ON) reculture in medium containing GM-CSF and IL-4. (A) ICP5, gD, envelope (env) (4 hours), and ICP8 (ON; arrows) were detected in HSV-2–infected cells by immunofluorescence microscopy. HSV-2 protein staining is shown in green and the nuclei in blue (DAPI). A representative example of each staining for macaque moDCs is shown (5 of 5 experiments with ICP5 and gD, 2 of 2 experiments with env, 7 of 8 experiments with ICP8). In the cells from the one animal in which we did not detect ICP8 staining after overnight (ON) reculture, ICP8 expression was detected within 1 to 4 hours of exposure. The relative IgG controls are shown in the lower row for each stain. Uninfected DCs showed no staining with the anti-HSV Abs. Original magnification, × 60. (B) Higher magnification of the middle left ICP8-stained cell from panel A showing the DAPI staining, the ICP8 staining, and their overlay is shown to appreciate the predominant nuclear localization of ICP8. The green ICP8 stain completely masks the blue DAPI-stained nucleus. (C) ICP8 staining was monitored by flow cytometry after 4 hours or ON reculture of medium- (Med) versus HSV-2–exposed (HSV) DCs. At each time point, cells were fixed/permeabilized and bound anti-ICP8 Ab detected with AffiniPure Donkey Anti–mouse IgG and the Qdot 525 streptavidin conjugate. The numbers in each panel indicate the percentage of ICP8+ cells (fluorescence versus forward scatter [FSC]) for one representative experiment of macaque moDCs. The quadrants were drawn relative to the medium-treated DCs incubated with anti-ICP8 for each time point. Isotype IgG controls stained less than 1 log (under the horizontal gate) on both infected and uninfected samples. (D) The percentages of ICP8+ cells from several experiments (7 for 4 hours and 15 for ON recultured macaque moDCs [filled symbols] and 3 for ON recultured human moDCs [open symbols]; each symbol represents a donor) are summarized for medium-treated versus HSV-infected cells. The horizontal black bars indicate the mean values.
HSV-2 infection of macaque DCs. Immature macaque and human moDCs were exposed to HSV-2 for 1 hour at 37°C (5 pfu per cell). HSV-2–loaded cells were then washed, counted, and plated for 4 hours up to overnight (ON) reculture in medium containing GM-CSF and IL-4. (A) ICP5, gD, envelope (env) (4 hours), and ICP8 (ON; arrows) were detected in HSV-2–infected cells by immunofluorescence microscopy. HSV-2 protein staining is shown in green and the nuclei in blue (DAPI). A representative example of each staining for macaque moDCs is shown (5 of 5 experiments with ICP5 and gD, 2 of 2 experiments with env, 7 of 8 experiments with ICP8). In the cells from the one animal in which we did not detect ICP8 staining after overnight (ON) reculture, ICP8 expression was detected within 1 to 4 hours of exposure. The relative IgG controls are shown in the lower row for each stain. Uninfected DCs showed no staining with the anti-HSV Abs. Original magnification, × 60. (B) Higher magnification of the middle left ICP8-stained cell from panel A showing the DAPI staining, the ICP8 staining, and their overlay is shown to appreciate the predominant nuclear localization of ICP8. The green ICP8 stain completely masks the blue DAPI-stained nucleus. (C) ICP8 staining was monitored by flow cytometry after 4 hours or ON reculture of medium- (Med) versus HSV-2–exposed (HSV) DCs. At each time point, cells were fixed/permeabilized and bound anti-ICP8 Ab detected with AffiniPure Donkey Anti–mouse IgG and the Qdot 525 streptavidin conjugate. The numbers in each panel indicate the percentage of ICP8+ cells (fluorescence versus forward scatter [FSC]) for one representative experiment of macaque moDCs. The quadrants were drawn relative to the medium-treated DCs incubated with anti-ICP8 for each time point. Isotype IgG controls stained less than 1 log (under the horizontal gate) on both infected and uninfected samples. (D) The percentages of ICP8+ cells from several experiments (7 for 4 hours and 15 for ON recultured macaque moDCs [filled symbols] and 3 for ON recultured human moDCs [open symbols]; each symbol represents a donor) are summarized for medium-treated versus HSV-infected cells. The horizontal black bars indicate the mean values.
HSV-2 infection induces death of macaque immature moDCs. Immature moDCs were exposed to HSV-2 or medium (Figure 1) and cells assayed after 4 hours or ON reculture. (A) Cell viability was determined by trypan blue exclusion. The mean percentages of viable cells (± SEM), relative to the input of cells replated after the 1-hour exposure to medium versus HSV-2, are shown from 9 to 17 experiments. (B) Overnight recultured immature macaque moDCs (medium-treated versus HSV-2–infected) were stained with annexin V and PI and analyzed by flow cytometry. The percentage of early apoptotic (annexin V–positive PI-negative, lower right quadrant) and late apoptotic/necrotic cells (annexin V–positive PI-positive, upper right quadrant) are indicated for 1 representative experiment of 6 performed. (C) The percentages of annexin V–positive PI-negative (left panel) and annexin V–positive (both PI-negative and PI-positive, right panel) cells are shown after 4 hours of reculture (medium-treated versus HSV-2–infected cells) and ON reculture (for cells exposed to medium [Med], infectious HSV-2 [HSV], or UV-treated HSV-2 [UV]). The minimal amounts of apoptosis seen in UV-treated HSV-2–exposed DCs above the medium controls could represent death induced by residual infectious virus or toxic effects of the virus stocks. Data were obtained using immature moDCs derived from 5 different Indian macaques (*DCs from T613 tested on 2 different occasions), 1 Chinese macaque (K453), and 3 human leukopacks (Hu 1, Hu 2, Hu 3; open symbols). Each symbol marks the individual donors. The black bars indicate the mean values.
HSV-2 infection induces death of macaque immature moDCs. Immature moDCs were exposed to HSV-2 or medium (Figure 1) and cells assayed after 4 hours or ON reculture. (A) Cell viability was determined by trypan blue exclusion. The mean percentages of viable cells (± SEM), relative to the input of cells replated after the 1-hour exposure to medium versus HSV-2, are shown from 9 to 17 experiments. (B) Overnight recultured immature macaque moDCs (medium-treated versus HSV-2–infected) were stained with annexin V and PI and analyzed by flow cytometry. The percentage of early apoptotic (annexin V–positive PI-negative, lower right quadrant) and late apoptotic/necrotic cells (annexin V–positive PI-positive, upper right quadrant) are indicated for 1 representative experiment of 6 performed. (C) The percentages of annexin V–positive PI-negative (left panel) and annexin V–positive (both PI-negative and PI-positive, right panel) cells are shown after 4 hours of reculture (medium-treated versus HSV-2–infected cells) and ON reculture (for cells exposed to medium [Med], infectious HSV-2 [HSV], or UV-treated HSV-2 [UV]). The minimal amounts of apoptosis seen in UV-treated HSV-2–exposed DCs above the medium controls could represent death induced by residual infectious virus or toxic effects of the virus stocks. Data were obtained using immature moDCs derived from 5 different Indian macaques (*DCs from T613 tested on 2 different occasions), 1 Chinese macaque (K453), and 3 human leukopacks (Hu 1, Hu 2, Hu 3; open symbols). Each symbol marks the individual donors. The black bars indicate the mean values.
Changes in macaque DC surface marker expression induced by HSV-2 infection
Initially, we observed that HLA-DR and CD86 expression were reduced in the HSV-2–infected macaque DCs after overnight reculture but not after 4 hours of reculture (Figure 3A-B). More extensive examination revealed that HLA-DR, CD40, CD80, CD83, and CD86 were decreased on cells expressing the highest levels of ICP8 (defined as ICP8+; Figure 3C). These markers were largely unaffected or even slightly elevated on ICP8– cells within the infected macaque DC cultures (uninfected or low-level infection) compared with the medium-cultured controls. Interestingly, cells from 3 animals infected with only 1 pfu of HSV-2 per cell (average, 1.66% ICP8+) exhibited increased levels of several of these surface markers on the ICP8– cells, but the levels of costimulatory molecule expression were still lower in the ICP8+ fraction (Figure 3C). These cells were clearly far healthier than cells exposed to 5 pfu of HSV-2 per cell (microscopic examination and forward scatter profile; not shown). We confirmed that the down-modulation of the costimulatory molecules requires infectious HSV-2, that exposure to UV-treated HSV-2 tended to increase the expression of these markers (like ICP8– cells in the low-level infected cultures), and that these responses reflected those seen in human moDCs (Figure 3D).
HSV-2 infection induces membrane phenotypic changes in macaque DCs. Medium-versus HSV-2–treated (5 pfu per cell or 1 pfu per cell [C, lower row]) immature moDCs were sampled after 4 hours (A-B) or ON reculture and stained for ICP8 combined with Abs against different membrane markers (HLA-DR, CD40, CD80, CD83, CD86) before being assayed by flow cytometry. (A) Representative FACS plots of ICP8 (x-axes) versus HLA-DR (top row) or CD86 (bottom row) from one macaque experiment are shown. Numbers in each plot represent the mean fluorescence intensity (MFI) of the HLA-DR or CD86 expressed by the medium-treated versus HSV-2–infected DCs (gated on the total population). All IgG controls exhibited less than 1 log of staining. (B) MFIs of HLA-DR and CD86 are shown from 2 representative experiments (of 4; animal L534, ▪; animal T602, ▴) for which the complete 4 hours versus ON recultures were performed. MFIs from L534 are derived from panel A. (C) Extended phenotypic analysis of ON-recultured macaque cells exposed to 5 pfu per cell (top row) or 1 pfu per cell (bottom row) was performed and the MFIs of the additional markers calculated. Summarized are the MFIs of HLA-DR, CD40, CD80, CD83, and CD86 expressed by medium-treated cells versus the ICP8– and ICP8+ gated subsets within the HSV-2–infected cells (top row: MFI means ± SEM for 3 to 7 animals, except for the CD40 expression by medium-treated cells, which was performed on the cells from 1 donor; bottom row: MFI means ± SEM from 3 animals). The ICP8+ and ICP8– subsets within the HSV-2–infected cells were defined based on the gate set on medium-cultured DCs stained with the anti-ICP8 Ab. (D) Immature moDCs were exposed to live (HSV, macaque and human) or UV-treated (UV, macaque) HSV-2 (5 pfu per cell or the equivalent) or not (Med, macaque and human) and incubated overnight. The mean (± SEM) MFIs of the indicated markers on the total DC populations are shown for 4 macaque (•) and 5 to 6 human (▵) experiments.
HSV-2 infection induces membrane phenotypic changes in macaque DCs. Medium-versus HSV-2–treated (5 pfu per cell or 1 pfu per cell [C, lower row]) immature moDCs were sampled after 4 hours (A-B) or ON reculture and stained for ICP8 combined with Abs against different membrane markers (HLA-DR, CD40, CD80, CD83, CD86) before being assayed by flow cytometry. (A) Representative FACS plots of ICP8 (x-axes) versus HLA-DR (top row) or CD86 (bottom row) from one macaque experiment are shown. Numbers in each plot represent the mean fluorescence intensity (MFI) of the HLA-DR or CD86 expressed by the medium-treated versus HSV-2–infected DCs (gated on the total population). All IgG controls exhibited less than 1 log of staining. (B) MFIs of HLA-DR and CD86 are shown from 2 representative experiments (of 4; animal L534, ▪; animal T602, ▴) for which the complete 4 hours versus ON recultures were performed. MFIs from L534 are derived from panel A. (C) Extended phenotypic analysis of ON-recultured macaque cells exposed to 5 pfu per cell (top row) or 1 pfu per cell (bottom row) was performed and the MFIs of the additional markers calculated. Summarized are the MFIs of HLA-DR, CD40, CD80, CD83, and CD86 expressed by medium-treated cells versus the ICP8– and ICP8+ gated subsets within the HSV-2–infected cells (top row: MFI means ± SEM for 3 to 7 animals, except for the CD40 expression by medium-treated cells, which was performed on the cells from 1 donor; bottom row: MFI means ± SEM from 3 animals). The ICP8+ and ICP8– subsets within the HSV-2–infected cells were defined based on the gate set on medium-cultured DCs stained with the anti-ICP8 Ab. (D) Immature moDCs were exposed to live (HSV, macaque and human) or UV-treated (UV, macaque) HSV-2 (5 pfu per cell or the equivalent) or not (Med, macaque and human) and incubated overnight. The mean (± SEM) MFIs of the indicated markers on the total DC populations are shown for 4 macaque (•) and 5 to 6 human (▵) experiments.
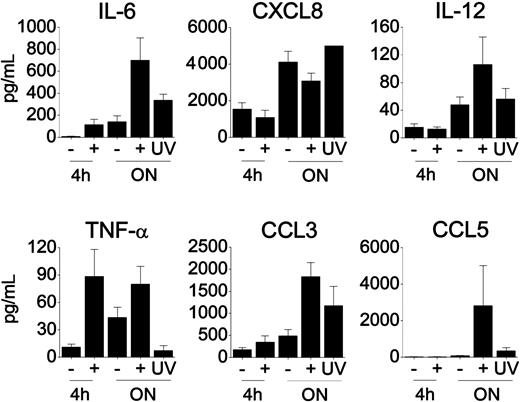
Modulation of macaque DC cytokine/chemokine profiles upon HSV-2 infection
As a functional measure of DC activity after exposure to infectious HSV-2 we evaluated the production of certain soluble factors over time. Infectious (not UV-treated) HSV-2 induced IL-6, tumor necrosis factor-α (TNF-α), macrophage inflammatory protein-1α (MIP-1α) (CCL3), and RANTES (regulated on activation normal T cells expressed and secreted) (CCL5) secretion by macaque DCs during the overnight reculture (Figure 4). UV-treated HSV-2 induced (at best) low-level IL-6, CCL3, and CCL5 release by macaque DCs. Some IL-6, TNF-α, and CCL3 were detected within 4 hours of infection, with the TNF-α response being maintained after overnight reculture. Contrastingly, HSV-2 induced little IL-12 p40 secretion by macaque DCs (Figure 4), and this was verified by an IL-12 ELISA that detects the p40 and p70 subunits (not shown). IL-8 (CXCL8) produced by macaque DCs was reduced upon HSV-2 infection. Unlike HSV-1 infection of human moDCs,38 we did not detect any increase in IFN-α release by HSV-2–infected macaque moDCs (not shown). Much like monkey DCs, HSV-2 infection induced human DCs to secrete more TNF-α (10.8 ± 1.8 versus 133.0 ± 39.9), negligible (overall less) IL-12 (111.8 ± 36.51 versus 35.4 ± 11.4), and consistently less CXCL8 (1191.7 ± 179.2 versus 727.1 ± 201.4; mean pg/mL ± SEM after overnight reculture, 3 uninfected versus HSV-2–infected DC donors). In 2 of the 3 human donors, infectious HSV-2 induced only low-level IL-6 release (about 1.2-fold more than medium-treated DCs), and this was evident only after 4 to 6 hours of reculture. HSV-2 did not induce CCL3 and CCL5 production by human DCs within this period. In 3 of 3 human DCs and 13 of 21 macaque DCs tested, MCP-1 (CCL2) was decreased after HSV-2 infection (not shown).
Decreased T-cell stimulatory capacity of HSV-2–infected macaque DCs
Because HSV-2 infection modulated the expression of costimulatory molecules and soluble factors by macaque DCs, we investigated whether this influenced their ability to stimulate an MLR.34 Because HSV-2 infection resulted in dramatic cell death after overnight reculture, we tested the immunostimulatory activity of HSV-2–infected versus uninfected DCs after 4 hours of reculture (when cells were still healthy; Figure 2).
Immature macaque DCs release cytokines and chemokines in response to HSV-2 infection. Cell-free supernatants were collected (after 4 hours or ON reculture) from infected (5 pfu per cell) versus uninfected (+ versus –) immature macaque moDC cultures. Cells from an additional 2 donors were also exposed to UV-treated HSV-2 (UV) and samples collected after overnight reculture. The presence of cytokines and chemokines was measured using the 22-plex Luminex assay (Upstate Biotechnology). The mean concentration of each cytokine/chemokine (mean pg/mL ± SEM) is shown from 3 to 17 experiments after 4 hours of reculture, from 15 to 21 experiments after ON reculture for uninfected versus infected DCs, and from 2 donors exposed to UV-treated HSV-2. Values for the CXCL8 responses from the 2 experiments comparing cells exposed to medium versus HSV-2 (live or UV treated) have been set at the 5000 pg/mL maximal value because the levels were out of range in the Luminex assay (with comparable MFIs for the differently treated cells). As a result, no error bars are shown for the UV-treated HSV-2 cell control.
Immature macaque DCs release cytokines and chemokines in response to HSV-2 infection. Cell-free supernatants were collected (after 4 hours or ON reculture) from infected (5 pfu per cell) versus uninfected (+ versus –) immature macaque moDC cultures. Cells from an additional 2 donors were also exposed to UV-treated HSV-2 (UV) and samples collected after overnight reculture. The presence of cytokines and chemokines was measured using the 22-plex Luminex assay (Upstate Biotechnology). The mean concentration of each cytokine/chemokine (mean pg/mL ± SEM) is shown from 3 to 17 experiments after 4 hours of reculture, from 15 to 21 experiments after ON reculture for uninfected versus infected DCs, and from 2 donors exposed to UV-treated HSV-2. Values for the CXCL8 responses from the 2 experiments comparing cells exposed to medium versus HSV-2 (live or UV treated) have been set at the 5000 pg/mL maximal value because the levels were out of range in the Luminex assay (with comparable MFIs for the differently treated cells). As a result, no error bars are shown for the UV-treated HSV-2 cell control.
HSV-2–infected macaque DCs induce lower T-cell proliferation. Immature macaque and human (A) moDCs were exposed or not to 5 pfu per cell HSV-2 (live versus UV treated) and recultured for 4 hours before being cultured with 2 × 105 human T cells in 96 well flat-bottomed trays (triplicates per condition) at the ratios indicated (normalized to viable DC numbers). After 5 days of culture, the plates were pulsed with 1 μCi (2.7 × 10–17 Bq) per well of 3H-TdR, and the amount of 3H-TdR incorporated (counts per minute [cpm]) in the last 8 hours of culture was measured. (A) The mean cpm (± SEM) is shown for one macaque and human representative experiment comparing DCs treated with medium (Med), UV-treated HSV-2 (UV), or infectious HSV-2 (HSV). The cpm values for DCs or T cells cultured alone were typically below 400 cpm. (B) The fold reduction (mean ± SEM, from 8 to 9 experiments comparing medium versus HSV-2–infected DCs and an additional 2 experiments comparing medium, UV-HSV, and infectious HSV-treated DCs) in the T-cell proliferation induced by HSV-2–infected macaque DCs at different DC–T cell ratios is indicated. The fold reduction was calculated by dividing the mean cpm of 3H-TdR uptake in cocultures of T cells with medium-treated DCs by the mean cpm of 3H-TdR incorporated by T cells cocultured with live or UV HSV-2–treated DCs. The values for the medium-treated DC–T cell cocultures are 1. (C) Medium-treated and HSV-2–infected macaque DCs were kept unfixed or an aliquot fixed with 1% PFA and used to stimulate human T cells at a ratio of 1 DC per 40 T cells. The fold reduction (mean ± SEM, 3 experiments using DCs from 3 different donors) in the T-cell proliferation induced by HSV-2–infected DCs was calculated (as in Figure 5B) for the unfixed and fixed DCs run in parallel.
HSV-2–infected macaque DCs induce lower T-cell proliferation. Immature macaque and human (A) moDCs were exposed or not to 5 pfu per cell HSV-2 (live versus UV treated) and recultured for 4 hours before being cultured with 2 × 105 human T cells in 96 well flat-bottomed trays (triplicates per condition) at the ratios indicated (normalized to viable DC numbers). After 5 days of culture, the plates were pulsed with 1 μCi (2.7 × 10–17 Bq) per well of 3H-TdR, and the amount of 3H-TdR incorporated (counts per minute [cpm]) in the last 8 hours of culture was measured. (A) The mean cpm (± SEM) is shown for one macaque and human representative experiment comparing DCs treated with medium (Med), UV-treated HSV-2 (UV), or infectious HSV-2 (HSV). The cpm values for DCs or T cells cultured alone were typically below 400 cpm. (B) The fold reduction (mean ± SEM, from 8 to 9 experiments comparing medium versus HSV-2–infected DCs and an additional 2 experiments comparing medium, UV-HSV, and infectious HSV-treated DCs) in the T-cell proliferation induced by HSV-2–infected macaque DCs at different DC–T cell ratios is indicated. The fold reduction was calculated by dividing the mean cpm of 3H-TdR uptake in cocultures of T cells with medium-treated DCs by the mean cpm of 3H-TdR incorporated by T cells cocultured with live or UV HSV-2–treated DCs. The values for the medium-treated DC–T cell cocultures are 1. (C) Medium-treated and HSV-2–infected macaque DCs were kept unfixed or an aliquot fixed with 1% PFA and used to stimulate human T cells at a ratio of 1 DC per 40 T cells. The fold reduction (mean ± SEM, 3 experiments using DCs from 3 different donors) in the T-cell proliferation induced by HSV-2–infected DCs was calculated (as in Figure 5B) for the unfixed and fixed DCs run in parallel.
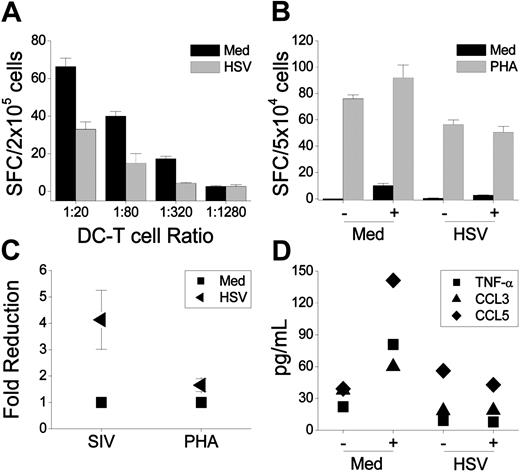
Although the same numbers of viable DCs were compared, HSV-2–infected immature DCs stimulated considerably less T-cell proliferation than medium- or UV-HSV-2–exposed DCs (Figure 5). Representative responses induced by macaque versus human DCs are shown (Figure 5A). Most assays were performed using fresh (unfixed) DCs (Figure 5A-B). Thus, the lower proliferative responses activated by infected DCs might simply have reflected the loss of DCs (and, hence, immunostimulatory activity) due to virus-induced DC death during the MLR cultures. Therefore, we compared PFA-fixed and unfixed HSV-2–infected and uninfected macaque DCs. The overall levels of T-cell proliferation were reduced when the DCs were PFA-fixed (not shown), as would be expected because the fixed DCs could not mature further in culture (upon T-cell contact). However, the difference between the magnitudes of the responses induced by HSV-2–infected and uninfected DCs persisted (Figure 5C).
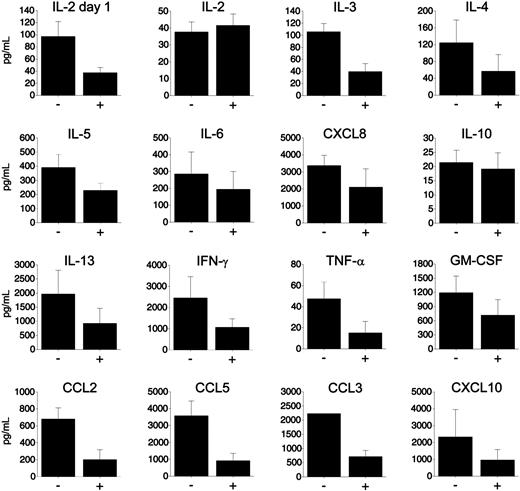
Luminex analyses revealed consistently lower responses in MLRs stimulated by HSV-2–infected macaque (Figure 6) and human (not shown) DCs. Although IL-2 levels were comparable after 5 days, examination after 1 day of culture confirmed that more IL-2 was produced earlier in the cultures stimulated by uninfected DCs, coinciding with greater T-cell proliferation (Figure 5). All other factors were highest in the 5-day cultures (not shown), and the levels of factors released in cultures stimulated by HSV-2–infected DCs were consistently lower than those induced by uninfected DCs (Figure 6). Comparable patterns (of lower magnitude) were seen in the supernatants collected from the cultures set up with PFA-fixed DCs (not shown). Comparing medium-, UV-treated HSV-2–, and live HSV-2–exposed DCs side by side, we verified that these decreased responses were dependent upon DC exposure to infectious HSV-2 (not shown).
HSV-2–infected immature macaque DCs induce poor SIV-specific responses
To extend our examination of the impact of HSV-2 infection on DC functions and to begin to understand how HSV-2 might impact SIV infection, we measured the capacity of HSV-2–infected moDCs to stimulate SIV-specific T cells. We achieved this by using DCs and T cells from healthy infected macaques and chemically inactivated AT-2 SIV as the antigen.15,31,34
Preliminary flow cytometric analyses using fluorescently labeled viruses39 confirmed that comparable numbers of HSV-2–infected DCs captured SIV compared with their uninfected counterparts (not shown; 30 ng p27 per 105 DCs as used for the antigen stimulation assays). Despite this, AT-2 SIV-loaded HSV-2–infected DCs induced lower levels of SIV-specific IFN-γ release compared with uninfected DCs (Figure 7A,C). The reduced stimulatory function of the HSV-infected DCs was less apparent (but detected) in PHA-stimulated DC–T cell mixtures (Figure 7B-C). Luminex analysis of the supernatants revealed that the low levels of SIV-specific TNF-α, CCL3, and CCL5 produced by cultures of healthy uninfected DCs and T cells were not detected in the HSV-2–infected DC–T cell mixtures (Figure 7D). PHA-induced TNF-α, CCL3, and CCL5 secretion was also lower in the HSV-2–infected DC–T cell cultures.
Reduced cytokine and chemokine responses in HSV-2–infected macaque DC-stimulated T-cell cocultures. MLR cultures were set up with uninfected (–) and HSV-2–infected (+) immature macaque moDCs (1 DC to 40 T cells), as described in Figure 5. Cell-free supernatants were collected and assayed (22-plex Luminex assay) prior to addition of 3H-TdR on day 5. Additional MLR cocultures were set up to assay IL-2 after 1 day of culture. The mean concentration (pg/mL) of each cytokine/chemokine (± SEM) is shown from 3 to 7 experiments using DCs and T cells from different donors. There is no error bar in the CCL3 graph because all values were above the highest limit of detection for the assay and have been set as this maximal value for each. Samples from the DC (5 × 104 cells per well) and T-cell (2 × 105 cells per well) controls cultured in medium were also measured. T cells produced negligible amounts of these factors when cultured alone. The amounts of factors released by the control DC cultures were close to or below the low detection limit for all except CXCL8. Even the 5 × 104 DCs per well released detectable amounts of CXCL8, but these were typically 1.5- to 2.2-fold less than that released in the DC–T cell cocultures (results from 4 experiments). Comparable results were obtained with human DCs from 2 separate donors, confirming that HSV-2–infected human DCs induced lower cytokine/chemokine responses in the allogeneic MLR.
Reduced cytokine and chemokine responses in HSV-2–infected macaque DC-stimulated T-cell cocultures. MLR cultures were set up with uninfected (–) and HSV-2–infected (+) immature macaque moDCs (1 DC to 40 T cells), as described in Figure 5. Cell-free supernatants were collected and assayed (22-plex Luminex assay) prior to addition of 3H-TdR on day 5. Additional MLR cocultures were set up to assay IL-2 after 1 day of culture. The mean concentration (pg/mL) of each cytokine/chemokine (± SEM) is shown from 3 to 7 experiments using DCs and T cells from different donors. There is no error bar in the CCL3 graph because all values were above the highest limit of detection for the assay and have been set as this maximal value for each. Samples from the DC (5 × 104 cells per well) and T-cell (2 × 105 cells per well) controls cultured in medium were also measured. T cells produced negligible amounts of these factors when cultured alone. The amounts of factors released by the control DC cultures were close to or below the low detection limit for all except CXCL8. Even the 5 × 104 DCs per well released detectable amounts of CXCL8, but these were typically 1.5- to 2.2-fold less than that released in the DC–T cell cocultures (results from 4 experiments). Comparable results were obtained with human DCs from 2 separate donors, confirming that HSV-2–infected human DCs induced lower cytokine/chemokine responses in the allogeneic MLR.
HSV-2 infection compromises the ability of immature DCs to stimulate SIV-specific responses. Immature moDCs from SHIV162P-infected macaques were infected with HSV-2 (5 pfu per cell) (HSV) or not (Med) before being recultured for 4 hours in the presence (+) or absence (–) of AT-2 SIV E11S (30 ng p27 per 105 DCs). The DCs were collected and viable cells recounted and plated in triplicate at varying doses with 5 × 104 to 2 × 105 autologous T cells. IFN-γ responses were monitored by ELISPOT (A-C) and other soluble factors measured in the supernatants using the Luminex assay (D). (A) The mean (± SEM) numbers of SIV-specific spot-forming cells (SFC) per 2 × 105 cells are plotted. The background levels of IFN-γ release in the absence of AT-2 SIV (0 SFCs for the medium-treated DC–T cell and 14 SFCs for the HSV-2–infected DC–T cell mixtures) have been subtracted to show the SIV-specific responses. Results from a representative animal are provided. (B) Representative PHA responses induced by the differently treated DC preparations (5 × 104 T cells with 2.5 × 103 DCs) are illustrated (mean ± SEM). Low-level SIV-specific responses (that are lowest in the HSV-2–infected DC cultures) are still apparent (compare the “+” versus “–” AT-2 SIV for Med- and HSV-treated DCs). (C) The fold reduction in the SIV-specific IFN-γ release for each experiment was calculated by dividing the mean SIV-specific IFN-γ release induced by medium-treated DCs by the mean SIV-specific IFN-γ release induced by HSV-treated DCs. The fold reduction in the IFN-γ release in the presence of PHA for each experiment was calculated by dividing the mean PHA-driven IFN-γ responses induced by medium-treated DCs by the mean responses induced by HSV-treated DCs. The mean fold lower (± SEM) SIV-specific (5 animals) and PHA-induced (3 animals) responses stimulated by HSV-2–infected DCs are shown. The values for the responses induced by medium-treated DCs are 1. (D) The pg/mL of TNF-α, CCL3, and CCL5 are graphed for cultures of 5 × 104 T cells with 2.5 × 103 DCs. PHA-induced TNF-α, CCL3, and CCL5 responses were also lower in cultures stimulated with HSV-2–infected DCs (16.19 versus 80.66, 18.78 versus 66.64, and 81.98 versus 123.84 pg/mL for infected versus uninfected DC-stimulated cultures, respectively). Data are shown from 1 representative experiment of 3 performed (3 different macaque donors).
HSV-2 infection compromises the ability of immature DCs to stimulate SIV-specific responses. Immature moDCs from SHIV162P-infected macaques were infected with HSV-2 (5 pfu per cell) (HSV) or not (Med) before being recultured for 4 hours in the presence (+) or absence (–) of AT-2 SIV E11S (30 ng p27 per 105 DCs). The DCs were collected and viable cells recounted and plated in triplicate at varying doses with 5 × 104 to 2 × 105 autologous T cells. IFN-γ responses were monitored by ELISPOT (A-C) and other soluble factors measured in the supernatants using the Luminex assay (D). (A) The mean (± SEM) numbers of SIV-specific spot-forming cells (SFC) per 2 × 105 cells are plotted. The background levels of IFN-γ release in the absence of AT-2 SIV (0 SFCs for the medium-treated DC–T cell and 14 SFCs for the HSV-2–infected DC–T cell mixtures) have been subtracted to show the SIV-specific responses. Results from a representative animal are provided. (B) Representative PHA responses induced by the differently treated DC preparations (5 × 104 T cells with 2.5 × 103 DCs) are illustrated (mean ± SEM). Low-level SIV-specific responses (that are lowest in the HSV-2–infected DC cultures) are still apparent (compare the “+” versus “–” AT-2 SIV for Med- and HSV-treated DCs). (C) The fold reduction in the SIV-specific IFN-γ release for each experiment was calculated by dividing the mean SIV-specific IFN-γ release induced by medium-treated DCs by the mean SIV-specific IFN-γ release induced by HSV-treated DCs. The fold reduction in the IFN-γ release in the presence of PHA for each experiment was calculated by dividing the mean PHA-driven IFN-γ responses induced by medium-treated DCs by the mean responses induced by HSV-treated DCs. The mean fold lower (± SEM) SIV-specific (5 animals) and PHA-induced (3 animals) responses stimulated by HSV-2–infected DCs are shown. The values for the responses induced by medium-treated DCs are 1. (D) The pg/mL of TNF-α, CCL3, and CCL5 are graphed for cultures of 5 × 104 T cells with 2.5 × 103 DCs. PHA-induced TNF-α, CCL3, and CCL5 responses were also lower in cultures stimulated with HSV-2–infected DCs (16.19 versus 80.66, 18.78 versus 66.64, and 81.98 versus 123.84 pg/mL for infected versus uninfected DC-stimulated cultures, respectively). Data are shown from 1 representative experiment of 3 performed (3 different macaque donors).
Discussion
Epidemiologic studies demonstrated that HSV-2 increases the frequency of HIV-1 transmission by 2- to 4-fold.19 Although mucosal epithelial cells (mainly keratinocytes) and sensory neurons are primary targets for HSV infection,40-42 DCs can become infected by HSV and their function down-modulated as a result.16,17,23,37 In addition to herpetic lesions affording HIV direct access to the blood and the activated leukocytes recruited to the lesions, we hypothesize that HSV-2 infection of DCs compromises their innate and adaptive activities to further exacerbate HIV transmission. Exploring ways to use the macaque system to study the spread of HIV in the face of sexually transmitted diseases (STDs), we provide the first evidence that HSV-2 infection of macaque DCs modifies their capacity to stimulate strong immunity, underscoring the potential for this animal model.
Similar to reports on murine37 and human18,23,27,38 DCs along with the human data herein, immature macaque DCs can be infected by HSV-2 (Figure 1). Discrepancies between the kinetics of infection and protein expression in each system probably reflect differences in the DCs used (bone marrow–derived DCs versus moDCs versus Langerhans cells in different species), the virus isolates examined (HSV-1 versus HSV-2), the different assays employed to monitor infection, and the time points after infection when the cells were analyzed. However, the predominantly nuclear localization of ICP8 in infected macaque DCs agrees with the fact that the single-strand DNA binding protein ICP8 is important for HSV-2 replication, accumulating in replication complexes within the nuclei.43,44
In all species, infectious HSV is cytotoxic and induces apoptosis of DCs (Figure 2).26,27,37,38 Inconsistencies in the numbers of infected macaque DCs (up to 35% ICP8+) versus the numbers of apoptotic (about 46%) or apoptotic/necrotic (about 82%) cells suggest that the methods employed to detect infection underestimated infected DC numbers. Alternatively, this might reflect bystander killing of noninfected DCs within the HSV-2–infected macaque DC cultures. HSV triggers apoptosis in immature human DCs by a caspase-dependent mechanism,26,27 but the mechanism of HSV-2–driven apoptosis of macaque DCs was not examined. TNF-α released in response to HSV by macaque (Figure 4) and human DCs26 could contribute to TNF-α–mediated apoptosis of infected DCs and bystander noninfected DCs that might also be mediated via a caspase-dependent mechanism.45
HSV infection of murine,35 human (Figures 3, 4, 5),16-18,23,27,38 and macaque DCs (Figures 3, 4, 5, 6, 7) also caused considerable modifications of their innate and adaptive functions. The earliest reports revealed that HSV infection of immature16,37 and mature17 DCs dampened their function by down-regulating expression of surface molecules important in DC–T-cell communication and DC immunostimulatory capacities. HSV infection even diminished the responses of immature human DCs to maturation stimuli.16 HSV-2 infection of macaque and human DCs similarly coincided with reduced HLA-DR, CD40, CD80, CD83, and CD86 expression. Notably, during early or low-level infection, there was less impact on the expression of these molecules. In fact, levels were often increased on cells expressing little HSV-2 protein (apparent in less aggressive infections after exposure to a low inoculum) or after exposure to UV-treated HSV-2.
Adding to human DC data,26 HSV-2–infected macaque DCs secreted increased levels of IL-6, TNF-α, CCL3, and CCL5 (Figure 4) but little IL-12 or IFN-α. Type I IFN released by HSV-1–infected human DCs might be important in the establishment of immune responses through the induction of maturation of bystander noninfected DCs.38 It is possible during the early stages of infection, when DCs are exposed to smaller amounts of infectious virus and/or noninfectious particles in the infecting inoculum, that at least low-level HSV-2–specific responses could be stimulated by the maturing DCs. IL-6 plays a significant role in limiting DC maturation.46 Even low-level IL-6 production combined with the paucity of type I IFN and IL-12 release by HSV-2–infected macaque (and human) DCs would favor the induction of less robust effector immune responses, allowing the onset of HSV-2 infection and potentially impacting responses against other antigens.
Predictably, HSV-2–infected macaque DCs possessed a generalized impairment of their immunostimulatory function (Figures 5, 6, 7), supporting evidence from other species (Figure 5A).16,23,37,38 This was evident at the level of T-cell proliferation and the magnitudes of cytokine and chemokine responses in DC–T cell cocultures. Decreased immunostimulatory activity was evident in macaque and human DCs within 4 hours of HSV-2 infection when membrane phenotypic changes were negligible but functional modifications were already apparent. This emphasizes the profound and rapid impact HSV-2 infection has on both macaque and human DCs. The most notable differences observed between human and macaque DCs were the kinetics of some of the cytokine responses and the lack of detectable CCL3 and CCL5 release by HSV-2–infected human DCs within the time frame examined. Importantly, however, DCs from both species manifested similarly impaired immunostimulatory function. Moreover, while the release of soluble factors was lower in HSV-2–infected (human and macaque) DC–T cell mixtures, there was no shift from T helper-1 (Th1) to Th2 type responses. Therefore, although responses were less robust, Th1 responses would still be able to be induced by HSV-2–infected DCs to ultimately control infection. This parallels the delayed or suboptimal responses described in the human system, which can be offset by uninfected DCs within an infected population cross-presenting HSV antigens.23,27,47 Innate activation of DCs through TLR stimulation by HSV also likely contributes to controlling HSV infection.48
Such compromised immunostimulatory function of macaque DCs also extended to SIV-specific responses (Figure 7). Even though HSV-2–infected macaque DCs captured SIV, their ability to stimulate SIV-specific IFN-γ, TNF-α, CCL3, and CCL5 release was diminished, much like the decreased antigen-specific responses induced by HSV-1–infected human DCs.23 This supports the hypothesis that HSV-2–infected immature DCs stimulate poor SIV-specific immune responses and may have important implications for HIV transmission.
This represents the first report demonstrating the modified immune functions of HSV-2–infected macaque DCs, validating the similarities between macaques and humans, and that HSV-2 infection compromises the capacity of DCs to stimulate SIV-specific responses. Whether this influences how HIV/SIV modifies immature moDCs12,13,49,50 remains to be determined and can now be explored in vivo in the macaque system. Being able to model HSV-2 infection in macaques, we are studying the impact of HSV-2 infection on transmission and testing how HSV-2 alters the efficacy of microbicide or vaccine strategies in preventing HIV spread. Identifying approaches that function effectively in the face of other infections, especially those that down-modulate immune function, will be critical in preventing the spread of HIV and other STDs.
Prepublished online as Blood First Edition Paper, April 21, 2005; DOI 10.1182/blood-2004-12-4899.
Supported by National Institutes of Health (NIH) grants R01 AI040877 and P01 HD041752; the Office of Population and Reproductive Health, Bureau for Global Health, U.S. Agency for International Development, under the terms of award no. HRN-A-00-99-00010; the Tulane National Primate Research Center (TNPRC) base grant RR00164; and the National Cancer Institute, NIH, under contract NO1-CO-12400. M.P. is an Elizabeth Glaser Scientist.
S.P., the primary researcher, performed research, analyzed data, and cowrote the paper; A.S. performed research under the primary researcher's guidance; J.B., R.B., and A.G. collaborated on animal care and acquisition of all blood samples for shipment to S.P.'s laboratory; G.M. performed research; J.D.L. contributed the (purified and characterized) SIV stocks used to monitor SIV capture and presentation by HSV-2–infected DCs and critically reviewed the paper; and M.P., the PI/Lab head, designed and coordinated the research and cowrote and edited the paper.
The publication costs of this article were defrayed in part by page charge payment. Therefore, and solely to indicate this fact, this article is hereby marked “advertisement” in accordance with 18 U.S.C. section 1734.
The authors thank Dr Anthony Cunningham, Lidija Bosnjak, and members of the laboratory of Dr Melissa Pope for critical discussions on the manuscript. Additional thanks go to Kanani Titchen and Jose Romero (in Dr David Phillips' laboratory at the Population Council) for additional technical support.
The opinions expressed herein are those of the authors and do not necessarily reflect the views of the U.S. Agency for International Development.

![Figure 1. HSV-2 infection of macaque DCs. Immature macaque and human moDCs were exposed to HSV-2 for 1 hour at 37°C (5 pfu per cell). HSV-2–loaded cells were then washed, counted, and plated for 4 hours up to overnight (ON) reculture in medium containing GM-CSF and IL-4. (A) ICP5, gD, envelope (env) (4 hours), and ICP8 (ON; arrows) were detected in HSV-2–infected cells by immunofluorescence microscopy. HSV-2 protein staining is shown in green and the nuclei in blue (DAPI). A representative example of each staining for macaque moDCs is shown (5 of 5 experiments with ICP5 and gD, 2 of 2 experiments with env, 7 of 8 experiments with ICP8). In the cells from the one animal in which we did not detect ICP8 staining after overnight (ON) reculture, ICP8 expression was detected within 1 to 4 hours of exposure. The relative IgG controls are shown in the lower row for each stain. Uninfected DCs showed no staining with the anti-HSV Abs. Original magnification, × 60. (B) Higher magnification of the middle left ICP8-stained cell from panel A showing the DAPI staining, the ICP8 staining, and their overlay is shown to appreciate the predominant nuclear localization of ICP8. The green ICP8 stain completely masks the blue DAPI-stained nucleus. (C) ICP8 staining was monitored by flow cytometry after 4 hours or ON reculture of medium- (Med) versus HSV-2–exposed (HSV) DCs. At each time point, cells were fixed/permeabilized and bound anti-ICP8 Ab detected with AffiniPure Donkey Anti–mouse IgG and the Qdot 525 streptavidin conjugate. The numbers in each panel indicate the percentage of ICP8+ cells (fluorescence versus forward scatter [FSC]) for one representative experiment of macaque moDCs. The quadrants were drawn relative to the medium-treated DCs incubated with anti-ICP8 for each time point. Isotype IgG controls stained less than 1 log (under the horizontal gate) on both infected and uninfected samples. (D) The percentages of ICP8+ cells from several experiments (7 for 4 hours and 15 for ON recultured macaque moDCs [filled symbols] and 3 for ON recultured human moDCs [open symbols]; each symbol represents a donor) are summarized for medium-treated versus HSV-infected cells. The horizontal black bars indicate the mean values.](https://ash.silverchair-cdn.com/ash/content_public/journal/blood/106/4/10.1182_blood-2004-12-4899/7/m_zh80160582670001.jpeg?Expires=1767709989&Signature=owEep2MH0bHyMvtZVv4FcmuevO1pHw4-Mixt2fUkalFnsh46ZchN71X1ypjl4NsgFOtqyO-zTxMayRLTc~824wfbhs3pexQ-Wt1xH2cJZ1EnMtvvCeT-IK0UAtGE-bMHevVjs4o2QZkh0RDhAHXV84LTEG-URK9LnCP6ry4Gjy1sMwlG39~Boh6VK9TBpARcFBi4--zXJ3KoCb2gfXEE2u3tQcfiVLuxniO-b5qfZjGt1kZy8~QUGxyqy~j7Ns9mEAa2OhSIBULqdPedjMURnoFMGNEyev8by9NVEJLDcFl-lyn9B0BRXpFmRUJQPI4pmoqQruN12c27Y7vfnrQe-Q__&Key-Pair-Id=APKAIE5G5CRDK6RD3PGA)
![Figure 2. HSV-2 infection induces death of macaque immature moDCs. Immature moDCs were exposed to HSV-2 or medium (Figure 1) and cells assayed after 4 hours or ON reculture. (A) Cell viability was determined by trypan blue exclusion. The mean percentages of viable cells (± SEM), relative to the input of cells replated after the 1-hour exposure to medium versus HSV-2, are shown from 9 to 17 experiments. (B) Overnight recultured immature macaque moDCs (medium-treated versus HSV-2–infected) were stained with annexin V and PI and analyzed by flow cytometry. The percentage of early apoptotic (annexin V–positive PI-negative, lower right quadrant) and late apoptotic/necrotic cells (annexin V–positive PI-positive, upper right quadrant) are indicated for 1 representative experiment of 6 performed. (C) The percentages of annexin V–positive PI-negative (left panel) and annexin V–positive (both PI-negative and PI-positive, right panel) cells are shown after 4 hours of reculture (medium-treated versus HSV-2–infected cells) and ON reculture (for cells exposed to medium [Med], infectious HSV-2 [HSV], or UV-treated HSV-2 [UV]). The minimal amounts of apoptosis seen in UV-treated HSV-2–exposed DCs above the medium controls could represent death induced by residual infectious virus or toxic effects of the virus stocks. Data were obtained using immature moDCs derived from 5 different Indian macaques (*DCs from T613 tested on 2 different occasions), 1 Chinese macaque (K453), and 3 human leukopacks (Hu 1, Hu 2, Hu 3; open symbols). Each symbol marks the individual donors. The black bars indicate the mean values.](https://ash.silverchair-cdn.com/ash/content_public/journal/blood/106/4/10.1182_blood-2004-12-4899/7/m_zh80160582670002.jpeg?Expires=1767709989&Signature=ZX3-xC6~ITP-oIUkWnqYsbtMXZPEWDH7IofuNA5zf0bStKs5kTtbIWWyZuOnAA7yI1fk31q3U6a7gVVp-Gi7mnuNnE6xbMUy85EKh5FnDLPzqE6i8XyVDh07xvKsauWYXrL3YJgQm6S3LW4cNj5hyF0Ri-49qnQ0eIvtQmyCTqZ9wmcyqSAKWxUzM6N8M8VGpVEdioX4N9t1pT3A1sUPxE4n83l~nLMoBz8e0EQLBat7omi0oWm7NU2agW9xBg86hJyGWt1WDucPBrJZ-ADa1ixDEuk6F8-lHpZ1qd9kOXmk-MrPOLUTNkHDP3MrqRubLSlPWByxQ2shkG8aWjOeMQ__&Key-Pair-Id=APKAIE5G5CRDK6RD3PGA)
![Figure 3. HSV-2 infection induces membrane phenotypic changes in macaque DCs. Medium-versus HSV-2–treated (5 pfu per cell or 1 pfu per cell [C, lower row]) immature moDCs were sampled after 4 hours (A-B) or ON reculture and stained for ICP8 combined with Abs against different membrane markers (HLA-DR, CD40, CD80, CD83, CD86) before being assayed by flow cytometry. (A) Representative FACS plots of ICP8 (x-axes) versus HLA-DR (top row) or CD86 (bottom row) from one macaque experiment are shown. Numbers in each plot represent the mean fluorescence intensity (MFI) of the HLA-DR or CD86 expressed by the medium-treated versus HSV-2–infected DCs (gated on the total population). All IgG controls exhibited less than 1 log of staining. (B) MFIs of HLA-DR and CD86 are shown from 2 representative experiments (of 4; animal L534, ▪; animal T602, ▴) for which the complete 4 hours versus ON recultures were performed. MFIs from L534 are derived from panel A. (C) Extended phenotypic analysis of ON-recultured macaque cells exposed to 5 pfu per cell (top row) or 1 pfu per cell (bottom row) was performed and the MFIs of the additional markers calculated. Summarized are the MFIs of HLA-DR, CD40, CD80, CD83, and CD86 expressed by medium-treated cells versus the ICP8– and ICP8+ gated subsets within the HSV-2–infected cells (top row: MFI means ± SEM for 3 to 7 animals, except for the CD40 expression by medium-treated cells, which was performed on the cells from 1 donor; bottom row: MFI means ± SEM from 3 animals). The ICP8+ and ICP8– subsets within the HSV-2–infected cells were defined based on the gate set on medium-cultured DCs stained with the anti-ICP8 Ab. (D) Immature moDCs were exposed to live (HSV, macaque and human) or UV-treated (UV, macaque) HSV-2 (5 pfu per cell or the equivalent) or not (Med, macaque and human) and incubated overnight. The mean (± SEM) MFIs of the indicated markers on the total DC populations are shown for 4 macaque (•) and 5 to 6 human (▵) experiments.](https://ash.silverchair-cdn.com/ash/content_public/journal/blood/106/4/10.1182_blood-2004-12-4899/7/m_zh80160582670003.jpeg?Expires=1767709989&Signature=XqHyIrDEj3BZo-8VeHMdd8cPGvTm6eItaLa2TFFNqM7bL7fvlQYjGRFDErp9NhPOIpv~~1~c00AIpW~yILzyBWZ9mRwlU9bdOUcVm8f7B1EeE54HpnDnxPPKMl~~KNOjiY5oFQ5P1x-l9BNiQUxrZ3-vGQn2pKYijOR-j9nOKgE5GxurTj7JR68xWDGfgVDLSAtILBtT8KyAD53SbhdR2aYCl0UaT5F5YIeaAl4vjfvGHPJ4yiQ7k9nS8bpwEZZapfOGtuHXu1y0z-1QI6~-po9hWn2W7XrXsSvdIJT8tacLLHipslXKPTg5~UuG1YvRjk5xjfVJSX2-84p1e91tMw__&Key-Pair-Id=APKAIE5G5CRDK6RD3PGA)
![Figure 5. HSV-2–infected macaque DCs induce lower T-cell proliferation. Immature macaque and human (A) moDCs were exposed or not to 5 pfu per cell HSV-2 (live versus UV treated) and recultured for 4 hours before being cultured with 2 × 105 human T cells in 96 well flat-bottomed trays (triplicates per condition) at the ratios indicated (normalized to viable DC numbers). After 5 days of culture, the plates were pulsed with 1 μCi (2.7 × 10–17 Bq) per well of 3H-TdR, and the amount of 3H-TdR incorporated (counts per minute [cpm]) in the last 8 hours of culture was measured. (A) The mean cpm (± SEM) is shown for one macaque and human representative experiment comparing DCs treated with medium (Med), UV-treated HSV-2 (UV), or infectious HSV-2 (HSV). The cpm values for DCs or T cells cultured alone were typically below 400 cpm. (B) The fold reduction (mean ± SEM, from 8 to 9 experiments comparing medium versus HSV-2–infected DCs and an additional 2 experiments comparing medium, UV-HSV, and infectious HSV-treated DCs) in the T-cell proliferation induced by HSV-2–infected macaque DCs at different DC–T cell ratios is indicated. The fold reduction was calculated by dividing the mean cpm of 3H-TdR uptake in cocultures of T cells with medium-treated DCs by the mean cpm of 3H-TdR incorporated by T cells cocultured with live or UV HSV-2–treated DCs. The values for the medium-treated DC–T cell cocultures are 1. (C) Medium-treated and HSV-2–infected macaque DCs were kept unfixed or an aliquot fixed with 1% PFA and used to stimulate human T cells at a ratio of 1 DC per 40 T cells. The fold reduction (mean ± SEM, 3 experiments using DCs from 3 different donors) in the T-cell proliferation induced by HSV-2–infected DCs was calculated (as in Figure 5B) for the unfixed and fixed DCs run in parallel.](https://ash.silverchair-cdn.com/ash/content_public/journal/blood/106/4/10.1182_blood-2004-12-4899/7/m_zh80160582670005.jpeg?Expires=1767709989&Signature=wxVkyPLxUUIzvwBVsf-621DJb8~2wjEK~nH-VEZ6g~XR0a38yPEonIKUvDn6rLr9I4LJmELzmIyaIK5u5vbmiDsKkk3mdsb8xZ1dQQguGX6yMRllpml95hgxQHOB4gU-rivVtcbuihZM1MgnRT199Wqz8K-r5TJoRwbokPJ0z25zYHY5CuvYHTvwooYRGaPNgpd-jlfJ-ebWvzN4118M9Ys~L3qCaj1ZKiu~IP2iWmzHGHwq0jntrw6oFCc1uoVkNhw3Yhq4cWynlZifhOFM1vg1gwWS3xJFPo0CWCJySEl43inlCEaIpEbQbg0mQwhyd7oE4~rUxikbL7C7fmX8Hg__&Key-Pair-Id=APKAIE5G5CRDK6RD3PGA)
This feature is available to Subscribers Only
Sign In or Create an Account Close Modal