The Bcr-Abl oncoprotein plays a major role in the development and progression of chronic myeloid leukemia (CML). Several studies have suggested that the expression levels of Bcr-Abl are elevated at disease progression to blast crisis and that this plays a significant role in the achievement of drug resistance. We have established cell lines expressing low and high levels of Bcr-Abl to study the molecular mechanisms involved in disease progression and drug resistance. It is now known that the endoplasmic reticulum (ER) can play a major role in the regulation of apoptosis. We therefore investigated whether Bcr-Abl expression modulates ER homeostasis and interferes with ER-mediated apoptotic pathways to promote survival. Bcr-Abl–expressing cells exhibit a decreased amount of free releasable calcium in the ER as well as a weaker capacitative calcium entry response, relative to parental cells. This effect is independent of Bcl-2, which is a known modulator of ER calcium homeostasis. The reduction in ER releasable calcium results in inhibition of the ER/mitochondria-coupling process and mitochondrial calcium uptake. This study demonstrates a novel downstream consequence of Bcr-Abl signaling. The ability to negate calcium-dependent apoptotic signaling is likely to be a major prosurvival mechanism in Bcr-Abl–expressing cells.
Introduction
Chronic myeloid leukemia (CML) is a clonal disorder of the hematopoietic system resulting in the excessive accumulation of immature and mature myeloid cells. The hallmark of CML is the presence of the Philadelphia (Ph) chromosome, which is present in more than 95% of cases and harbors the chimeric gene BCR-ABL. The resultant tyrosine kinase, Bcr-Abl, constitutively activates several signal transduction pathways1,2 and is believed to be the critical determinant in the pathogenesis of CML.
CML is a biphasic disease, starting as an indolent chronic phase followed by a much more aggressive and drug-resistant phase called blast crisis. A specific inhibitor of Bcr-Abl tyrosine kinase activity, imatinib (Gleevec, Glivec) has recently been introduced for the treatment of CML.3,4 Impressive responses have been seen with patients in chronic phase; however, resistance is common in advanced CML. Reported resistance mechanisms are primarily mutations and amplification of the BCR-ABL gene.5-8
A number of groups have suggested that the reason blast crisis (BC) CML is multidrug resistant is due to the antiapoptotic properties of the Bcr-Abl oncoprotein. It has already been shown that BCR-ABL can confer drug resistance when it is transfected into other leukemic cell lines.9 Several clinical and molecular studies have suggested that the expression levels of Bcr-Abl may play a major role in resistance to apoptosis.10-13 Down-regulation of Bcr-Abl transcript has been shown to increase drug sensitivity in cells derived from CML.14 Expression of Bcr-Abl at different levels in hematopoietic progenitor cells found that all cells became growth factor independent, but only the high Bcr-Abl–expressing cells were resistant to apoptosis induced by cytotoxic drugs.10 This is analogous to the 32D transfected cell lines that we have characterized.12,15,16
It has recently been established that the endoplasmic reticulum (ER) can play a critical role in the regulation of apoptosis. The ER can activate apoptosis independently of or upstream of the mitochondria.17,18 ER Ca2+ release leads to rapid calcium uptake and accumulation in mitochondria (ER/mitochondria coupling).19-23 Mitochondrial calcium overload can promote permeability transition pore (PTP) opening and cytochrome c release. Specific inhibition of ER-to-mitochondrion Ca2+ transport attenuates downstream biochemical events associated with apoptosis. Proapoptotic and antiapoptotic Bcl-2 family members located in the ER have also been shown to modulate ER calcium homeostasis, thereby influencing susceptibility to apoptosis.24-30
The influence of the Bcr-Abl oncoprotein on ER-initiated apoptotic signaling pathways has never been investigated. Here, we show that Bcr-Abl–expressing cells exhibit a decreased amount of free releasable calcium in the ER as well as a weaker capacitative calcium entry (CCE) response relative to parental 32D cells. This results in attenuation of the ER/mitochondria-coupling process and mitochondrial calcium overload. This effect is independent of Bcl-2 expression. This is the first evidence that Bcr-Abl expression affects ER homeostasis and calcium-dependent apoptotic signaling. This ability will undoubtedly contribute to the antiapoptotic repertoire of the Bcr-Abl oncogene.
Materials and methods
Materials
Etoposide (VP-16), thapsigargin (TG), propidium iodide (PI), streptolysin O, Hoechst 33258, and ruthenium red (RuRed) were obtained from Sigma-Aldrich (Dublin, Ireland). RuRed was taken up by cells on extended incubation, and therefore no permeabilization was undertaken. Calcium-Green-1 am (CaGreen-1), MitoTracker Green fm (MitoTracker Green), and rhodamine-2 am (Rhod-2) were from Molecular Probes (Leiden, The Netherlands). Caspase inhibitor zVAD.fmk was from Bachem (St Helens, United Kingdom) and calpain inhibitor III (MDL 28170) from Calbiochem (Darmstadt, Germany).
Cell culture
Bcr-Abl–transformed 32D cells (obtained from American Type Culture Collection, Manassas, VA) were cloned and cultured as described previously.16 K562, BV173, Jurkat, and HL60 cells were grown in RPMI 1640 with 10% FCS, 2 mM l-glutamine, 1% penicillin and streptomycin (P/S).
Assessment of cell viability
Cell viability was assessed by PI exclusion. Cells (0.5 × 106) were resuspended in PBS and 50 μg/mL PI prior to flow cytometric analysis (FACScan; Becton Dickinson, Oxford, United Kingdom).
TUNEL assay
Cells were fixed with 3% para-formaldehyde for 15 minutes on ice, washed, and transferred to ice-cold 70% ethanol. For the terminal deoxynucleotidyl transferase–mediated dUTP nick-end labeling (TUNEL) assay, the 3′OH DNA ends were labeled with FITC-conjugated dUTP (Roche, Mannheim, Germany) by terminal deoxynucleotidyl transferase (TdT; Promega, Southampton, United Kingdom).
Measurement of intracellular calcium
Cytosolic-free Ca2+ was assessed with the Ca2+-sensitive cell permeable dye CaGreen-1 and mitochondrial Ca2+ using Rhod-2. Cells (0.5 × 106) were resuspended in 1 mL RPMI and loaded with either 1 μM CaGreen-1 or Rhod-2 for 15 minutes at room temperature. Labeled cells were washed twice with calcium-free Krebs-Henseleit buffer (120 mM NaCl, 4.8 mM KCl, 1.2 mM MgSO4, 1.2 mM KH2PO4, 25 mM NaHCO3, 5 mM HEPES, pH 7.2, 10 mM glucose). Analysis was performed using flow cytometry (Becton Dickinson) on FL-l for CaGreen-1 and FL-2 for Rhod-2, respectively.
Measurement of ER calcium content and CCE
Cells were resuspended in calcium-free medium and loaded with 1 μM CaGreen-1 as described (see “Measurement of intracellular calcium”). Fluorescence was quantitated by flow cytometry on the FL-1 channel as a time function. TG at a concentration of 2 μM was added after recording the basal cytosolic Ca2+ concentration (steady state). The ER calcium content was determined as the difference between the base level of CaGreen-1 fluorescence and the level after TG addition using the statistical analysis performed by CellQuest software (Becton Dickinson) and shown as arbitrary fluorescence units. CCE was determined by the subsequent addition of 5 mM CaCl2 to the medium. Three regions corresponding to the steady-state cytosolic calcium level (R1), cytosolic calcium level after ER depletion by TG treatment (R2), and after capacitative calcium uptake (R3) and gated events were shown as histograms. The ER releasable calcium content and CCE were calculated as ΔF/F0 = 100((F–F0)/F0), where F0 is the median fluorescence of R1 (steady state) and F either the median fluorescence of R2 (ER content) or R3 (CCE). %ΔF/F0 ratios from all experiments are presented as mean ± SD (mean %ΔF/F0).
RNAi
C2 and C4 cells were transfected with siRNA (230 pmol) corresponding to a 21-nucleotide (nt) region from Bcl-2 (M16506, nt 64-86) in the presence of streptolysin O (100 U). A nonspecific siRNA was used as a negative control.31 ER calcium content was measured 18 to 24 hours after transfection and cells were cytospun for immunofluorescence analysis.
Immunofluorescence
siRNA-transfected cells were fixed and then permeabilized using 0.05% saponin, followed by incubation with anti–Bcl-2 (N-19) antibody (Santa Cruz Biotechnology, Santa Cruz, CA) at 1:500 and AlexaFluor-488 anti–rabbit IgG antibody at 1:200 (Invitrogen, Molecular Probes, Leiden, the Netherlands). Nuclei were counterstained with Hoechst 33258. Analysis was carried out using an Olympus BX51 fluorescence microscope equipped with an FITC filter (Olympus, Hamburg, Germany).
Results
Bcr-Abl–expressing cells have a decreased amount of free releasable calcium in the ER and exhibit decreased CCE
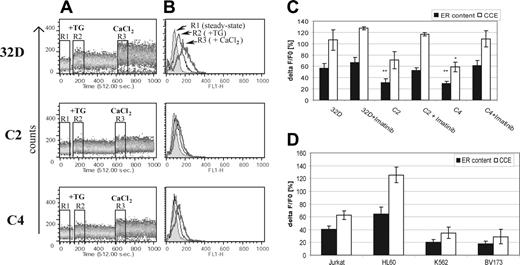
We had identified elevated expression of ER stress markers glucose-related protein (Grp78) and CAAT/enhancer-binding protein homologous protein (CHOP) in both high (C4) and low (C2) Bcr-Abl–expressing cells relative to parental 32D cells (Supplemental Figure S1, available on the Blood website by clicking the Supplemental Figures link at the top of the online article; refer also to the cell model described in our previous publications12,15,16,31 ). To determine whether this was also associated with altered calcium homeostasis, we measured the relative amounts of steady-state ER releasable calcium in 32D and Bcr-Abl–expressing cells (Figure 1). The ER calcium content was assessed by quantifying TG-induced ER-calcium release. Addition of 2 μM TG to the cells in a Ca2+-free medium led to an immediate emptying of ER Ca2+ stores, observed as an increase in the cytosolic calcium level. ER calcium store depletion is also known to be a direct signal for CCE, which replenishes empty stores. This response is proportional to the amount of calcium released from the ER. We therefore investigated the CCE by subsequently adding 5 mM CaCl2 to the medium. The further increase in the cytosolic calcium dye fluorescence indicated the level of calcium influx across the plasma membrane into the cytosol. The cytosolic calcium level was measured by flow cytometric analysis of cells loaded with a cytosolic calcium-sensitive fluorescent probe, CaGreen-1, and the level of fluorescence measured on the FL-1 channel was shown as a time function (Figure 1A). We found that in Bcr-Abl–expressing cells the amplitude of TG-induced Ca2+ release was markedly reduced compared with 32D cells. Accordingly, the CCE was much stronger in 32D cells, where the amount of calcium released from the ER was higher. Three regions corresponding to the steady-state cytosolic calcium level (R1), the calcium level after ER store depletion (R2), and after CCE to the cytosol (R3) were marked and shown as histograms (Figure 1B). Statistical analysis of data from triplicate experiments indicated that there is a 50% decrease in ER releasable calcium in C2 and C4 cells, followed by a similar decrease in CCE in these cells (Figure 1C).
Bcr-Abl–expressing cells exhibit a reduced amount of releasable calcium from ER stores and decreased CCE. (A) ER calcium release and CCE were measured in untreated 32D, C2, and C4 cells. Cells were loaded with 1 μM CaGreen-1 and the level of fluorescence was measured by flow cytometry on the FL-1 channel as a time function. Cells were treated with 2 μM TG to deplete ER calcium stores followed by 5 mM CaCl2 addition to the medium to assess the capacitative calcium uptake to the cytosol. Three regions were gated during the time-course analysis corresponding to the steady-state cytosolic calcium level (R1), cytosolic calcium level after ER depletion by TG treatment (R2), and after capacitative calcium uptake (R3). A representative result from 3 independent experiments is shown. (B) ER calcium release and CCE in 32D, C2, and C4 cells shown as histograms corresponding to the 3 selected regions. R1 region is the filled histogram, R2 is black-line histogram, and R3 is the gray-line histogram. (C) Ca2+ pool released from the ER (▪) and taken up from the extracellular medium (□, CCE) in 32D, C2, and C4 cells and (D) Jurkat, HL60, K562, and BV173 cells. In the case of 32D, C2, and C4 cells, imatinib treatment at 10 μM was performed for 4 hours prior to analysis (32D, C2, C4 + imatinib). The fluorescence values were obtained by statistical analysis using CellQuest software, and the ER releasable calcium content and CCE were calculated as ΔF/F0 = 100((F–F0)/F0), where F0 is the median fluorescence of R1 (steady state) and F either the median fluorescence of R2 (ER content) or R3 (CCE). %ΔF/F0 ratios from all experiments presented as mean %ΔF/F0 ± SD. *P < .01; **P < .001 versus 32D cells (Student t test).
Bcr-Abl–expressing cells exhibit a reduced amount of releasable calcium from ER stores and decreased CCE. (A) ER calcium release and CCE were measured in untreated 32D, C2, and C4 cells. Cells were loaded with 1 μM CaGreen-1 and the level of fluorescence was measured by flow cytometry on the FL-1 channel as a time function. Cells were treated with 2 μM TG to deplete ER calcium stores followed by 5 mM CaCl2 addition to the medium to assess the capacitative calcium uptake to the cytosol. Three regions were gated during the time-course analysis corresponding to the steady-state cytosolic calcium level (R1), cytosolic calcium level after ER depletion by TG treatment (R2), and after capacitative calcium uptake (R3). A representative result from 3 independent experiments is shown. (B) ER calcium release and CCE in 32D, C2, and C4 cells shown as histograms corresponding to the 3 selected regions. R1 region is the filled histogram, R2 is black-line histogram, and R3 is the gray-line histogram. (C) Ca2+ pool released from the ER (▪) and taken up from the extracellular medium (□, CCE) in 32D, C2, and C4 cells and (D) Jurkat, HL60, K562, and BV173 cells. In the case of 32D, C2, and C4 cells, imatinib treatment at 10 μM was performed for 4 hours prior to analysis (32D, C2, C4 + imatinib). The fluorescence values were obtained by statistical analysis using CellQuest software, and the ER releasable calcium content and CCE were calculated as ΔF/F0 = 100((F–F0)/F0), where F0 is the median fluorescence of R1 (steady state) and F either the median fluorescence of R2 (ER content) or R3 (CCE). %ΔF/F0 ratios from all experiments presented as mean %ΔF/F0 ± SD. *P < .01; **P < .001 versus 32D cells (Student t test).
Lower ER releasable calcium is associated with Bcr-Abl expression and activity
To demonstrate that this reduced ER calcium content was a consequence of Bcr-Abl activity, we treated C4 and C2 cells with imatinib, a specific inhibitor of Bcr-Abl tyrosine kinase activity. We have previously shown that a concentration of 10 μM reduces cellular phosphotyrosine in C4 cells to basal (32D) levels.15 Treatment for a short period (4 hours) does not affect viability as demonstrated by lack of PI uptake (not shown). Imatinib treatment resulted in an increase in ER releasable calcium and stronger CCE in both cell lines, indicating that Bcr-Abl activity is required for their reduction in ER releasable calcium. Importantly, in 32D cells, imatinib did not alter the releasable calcium content or CCE, indicating that the drug is specifically influencing Bcr-Abl (Figure 1C).
To further investigate this association between Bcr-Abl expression and low ER releasable calcium, we examined the ER calcium content in 2 Bcr-Abl+ and 2 Bcr-Abl– human hematopoietic cell lines. As shown in Figure 1D, the Bcr-Abl–expressing cell lines K562 and BV173 had comparable levels of releasable calcium in the ER to the 2 Bcr-Abl–expressing mouse cell lines, C2 and C4. The ER of Jurkat and HL60 cells contained higher amounts of releasable calcium. The CCE was also stronger in the Bcr-Abl– cell lines than in K562 and BV173 cells. These data indicate that low releasable amounts of calcium in the ER and a weak CCE is not only a feature of C2 and C4 cells, but also of 2 Bcr-Abl+ human cell lines. We were, however, unable to elevate the ER calcium levels of K562 and BV173 to those of HL60 and Jurkat cells with imatinib. Although these cell lines are not directly comparable, due to different origins and lineages, these data suggest that, whereas 32D cells rely solely on the presence of Bcr-Abl to lower their ER calcium levels, the established human cell lines may have acquired additional survival mechanisms that contribute to the reduction in ER calcium. The attainment of low ER calcium may be a particularly important feature for cells transformed by Bcr-Abl.
Reduced releasable ER calcium is independent of Bcl-2 expression
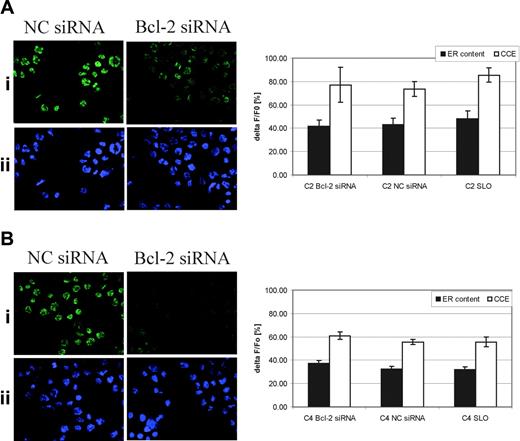
Decreased levels of releasable calcium in the ER are associated with cell survival (for a review, see Szabadkai et al32 ) and are analogous to the reported effects of Bcl-2, which reduces ER releasable calcium and confers drug resistance. We therefore examined whether Bcl-2 may be acting downstream of Bcr-Abl and be responsible for the reduced releasable calcium. Bcl-2 transcript was down-regulated by siRNA expression in Bcr-Abl–expressing cell lines, and down-regulation of protein expression was confirmed by fluorescence microscopy (Figure 2A-B). The depletion of Bcl-2 had no effect on ER releasable calcium or CCE (Figure 2A-B).
VP-16 treatment induces elevation of cytosolic and mitochondrial Ca2+ levels in 32D hematopoietic cells
We have previously reported that high levels of Bcr-Abl (as in C4 cells) can confer drug resistance.16 Inhibition of Bcr-Abl activity using imatinib can reverse the phenotype indicating that Bcr-Abl activity is associated with drug resistance and this phenotype is dose dependent. Cells expressing lower amounts of Bcr-Abl (C2 cells) are IL-3 independent, but their drug sensitivity is analogous to parental cells (Keeshan et al16 and subsequent data sections).
A previous study has reported that apoptotic stimuli can be classified into 3 categories depending on the apoptotic pathways they engage—calcium/ER, BH3/mitochondria, or both.22 VP-16 was reported to engage both the ER and mitochondrial pathways. To investigate whether the lower steady-state ER calcium content observed in Bcr-Abl–expressing cells affects calcium changes associated with drug-induced ER/mitochondria coupling, levels of cytosolic and mitochondrial calcium were assessed following VP-16 treatment. Cells were treated with VP-16 at 7.5 μg/mL and the percentage of cell death was quantified by measuring PI uptake by flow cytometry.
Effect of Bcl-2 down-regulation on levels of ER releasable calcium and CCE in Bcr-Abl–expressing cells. (A) C2 and (B) C4 cells were transfected with either a nonspecific (NC siRNA) or Bcl-2 siRNA and Bcl-2 down-regulation was shown by immunofluorescence. Cells were incubated with anti–Bcl-2 (N-19) antibody followed by an incubation with AlexaFluor-488 anti–rabbit IgG antibody (i). Nuclei were stained with Hoechst 33258 (ii). Analysis was carried out using an Olympus BX51 fluorescence microscope equipped with an FITC filter using an Olympus UPlan Fl 40 ×/0.75 objective lens. Pictures were taken using a DP70 digital microscope camera (Olympus). Olympus DP-Soft 823 version 3.2 image acquisition software was used to acquire images, which were then processed in Adobe Photoshop 7.0 software (Adobe Systems, San Jose, CA). The figure was prepared with Adobe Illustrator 10.0.3 software. ER releasable calcium was measured as described in “Materials and methods” and Figure 1 and data are shown as ΔF/F0 (%). As a negative control, cells were treated with streptolysin O only (SLO) and ER calcium content and CCE were also measured. Data of 3 independent experiments are shown (mean ± SD).
Effect of Bcl-2 down-regulation on levels of ER releasable calcium and CCE in Bcr-Abl–expressing cells. (A) C2 and (B) C4 cells were transfected with either a nonspecific (NC siRNA) or Bcl-2 siRNA and Bcl-2 down-regulation was shown by immunofluorescence. Cells were incubated with anti–Bcl-2 (N-19) antibody followed by an incubation with AlexaFluor-488 anti–rabbit IgG antibody (i). Nuclei were stained with Hoechst 33258 (ii). Analysis was carried out using an Olympus BX51 fluorescence microscope equipped with an FITC filter using an Olympus UPlan Fl 40 ×/0.75 objective lens. Pictures were taken using a DP70 digital microscope camera (Olympus). Olympus DP-Soft 823 version 3.2 image acquisition software was used to acquire images, which were then processed in Adobe Photoshop 7.0 software (Adobe Systems, San Jose, CA). The figure was prepared with Adobe Illustrator 10.0.3 software. ER releasable calcium was measured as described in “Materials and methods” and Figure 1 and data are shown as ΔF/F0 (%). As a negative control, cells were treated with streptolysin O only (SLO) and ER calcium content and CCE were also measured. Data of 3 independent experiments are shown (mean ± SD).
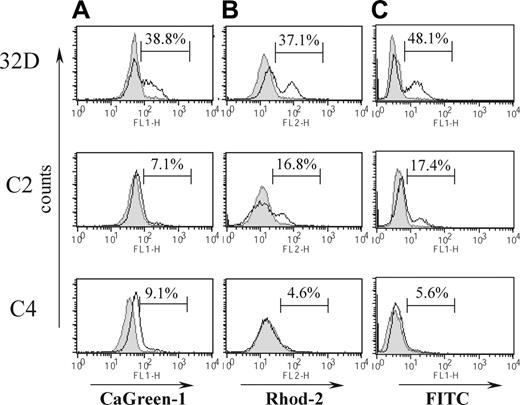
To exclude the possibility that changes in internal calcium levels were a consequence of late apoptotic-cell membrane permeabilization, we analyzed cells after 14 hours of VP-16 treatment. At this time, the percentage of PI+ cells did not exceed 15%. Cells were loaded with the cytosolic Ca2+ probe CaGreen-1 or the mitochondrial probe Rhod-2 and analyzed by flow cytometry; representative histograms are shown in Figures 3A and 3B, respectively. In 32D cells loaded with CaGreen-1, a population with a higher cytosolic calcium level was apparent (38.8%; n = 3; 34.4% ± 5.5%) in contrast to C2 and C4 cells, where the increase in cytosolic calcium was much smaller, almost undetectable (7.1% for C2 cells, n = 3; 5.4% ± 2% and 9.1% for C4 cells, n = 3; 5.1% ± 3.6%; Figure 3A). After staining with Rhod-2, the level of mitochondrial calcium following VP-16 treatment was also analyzed (Figure 3B). Exposure of 32D cells to VP-16 resulted in a significant increase in the mitochondrial Ca2+ concentration and 37.1% (n = 3; 34.8% ± 3.5%) of cells possessed mitochondria with elevated calcium. In C2 cells, mitochondrial, calcium-dependent changes were weaker (16.8%; n = 3; 17.5% ± 3.4%) and almost undetectable in C4 cells (4.6%). A strongly elevated mitochondrial calcium level is a known inducer of PTP opening, releasing proapoptotic factors from mitochondria to the cytosol. Because we detected a pronounced mitochondrial calcium overload in 32D cells, this suggested that a different pathway of cell-death initiation might occur in these cells. We then analyzed DNA nicking by TUNEL assay as an early indicator of apoptosis 14 hours after inducing cell death (Figure 3C). We found that 48.1% of 32D cells (n = 3; 39.8% ± 9.7%) had nicked DNA, whereas only 17.4% of C2 cells (n = 3; 15.8% ± 2.8%) were considered as TUNEL+. This experiment suggested that 32D cells may be able to activate an early calcium-dependent apoptotic response caused by substantial ER calcium release, whereas this is diminished in both the high and low Bcr-Abl–expressing cell lines.
Because mitochondrial Ca2+ elevation is a critical parameter in assessing a potential role for ER/mitochondrial coupling and apoptosis, we undertook a number of approaches to verify the specificity of the Rhod-2 probe for mitochondrial calcium (colocalization with MitoTracker Green, and use of a mitochondrial uncoupling agent to dissipate fluorescence; Figure S2) and also to verify the quantitative differences in mitochondrial Ca2+ accumulation identified by flow cytometry. TILL photonic imaging (Till Photonics, Gräfelfing, Germany) and quantification of calcium content in individual cells indicated that 32D cells exhibit significantly higher (almost 3-fold higher) mitochondrial calcium following drug treatment compared with Bcr-Abl–expressing cells (Figure S2B).
Effects of Bcr-Abl expression on apoptosis and intracellular calcium changes after VP-16 treatment. (A) Cytosolic and (B) mitochondrial calcium level measured by flow cytometry. Cells were treated with VP-16 for 14 hours, loaded with 1 μM CaGreen-1 or 1 μM Rhod-2, and analyzed on the FL-1 or FL-2 channel, respectively. Untreated cells are shown as filled histograms, and VP-16–treated cells as black-line histograms. (C) Percentage of apoptotic cells quantified by TUNEL assay. Cells were incubated with VP-16 for 14 hours, fixed, incubated with TdT/dUTPs-FITC, and analyzed on the FL-1 channel. Cells with higher fluorescence correspond to cells with fragmented DNA. Untreated cells are shown as filled histograms and VP-16–treated cells as black-line histograms. Representative histograms are shown.
Effects of Bcr-Abl expression on apoptosis and intracellular calcium changes after VP-16 treatment. (A) Cytosolic and (B) mitochondrial calcium level measured by flow cytometry. Cells were treated with VP-16 for 14 hours, loaded with 1 μM CaGreen-1 or 1 μM Rhod-2, and analyzed on the FL-1 or FL-2 channel, respectively. Untreated cells are shown as filled histograms, and VP-16–treated cells as black-line histograms. (C) Percentage of apoptotic cells quantified by TUNEL assay. Cells were incubated with VP-16 for 14 hours, fixed, incubated with TdT/dUTPs-FITC, and analyzed on the FL-1 channel. Cells with higher fluorescence correspond to cells with fragmented DNA. Untreated cells are shown as filled histograms and VP-16–treated cells as black-line histograms. Representative histograms are shown.
In addition, flow cytometric analysis indicated that 32D cells exhibit a rise in cytosolic calcium following drug treatment and that this is not a significant feature of Bcr-Abl–expressing cells (Figure 3), again possibly due to their lower releasable ER calcium. Comparison of calcium-dependent calpain activity in 32D and C2 cells provided further biochemical evidence for higher cytosolic calcium in 32D cells following drug treatment (Figure S3).
Differential effects of protease inhibitors indicate distinct pathways for drug sensitivity in 32D and C2 cells
We have already shown that both C2 and C4 cells have lower releasable calcium in the ER; however, C2 cells still show minor alterations in cytosolic or mitochondrial calcium, which become much more significant as the duration of drug treatment is increased. Such calcium elevations are never detected in the high Bcr-Abl–expressing C4 cells. It is therefore possible that these calcium changes are a secondary, caspase-dependent event in C2 cells because they are drug sensitive and are therefore initiating at least one apoptotic pathway, whereas the C4 cells are drug resistant. We already know that the classic (caspase-dependent) pathway is defective in C4 cells (reported in our previous publication12 ) but is active in C2 and 32D cells. We therefore examined whether the mitochondrial calcium accumulation in 32D and C2 cells is caspase dependent.
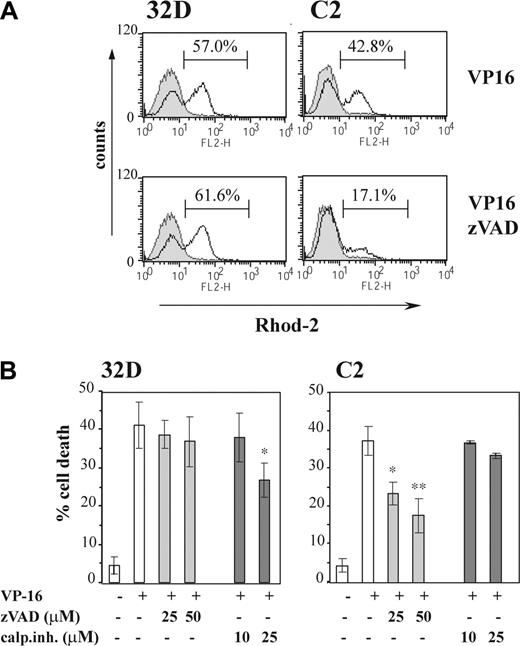
32D and C2 cells were pretreated with 50 μM zVAD, a potent inhibitor of multiple caspases. Cells were then treated with VP-16 for 18 hours and loaded with Rhod-2 and the mitochondrial calcium level was analyzed by flow cytometry. For this experiment the 18-hour time point was chosen to achieve appreciable Ca2+ accumulation in C2 cells (which is a slightly later event than in 32D), and examine whether or not this can be inhibited with zVAD. We found that 35% of 32D cells were positive for PI without zVAD pretreatment and 40% with pretreatment. C2 cells were 40% PI+ without zVAD and 14% with treatment (not shown; refer to combined data in Figure 4B). As Figure 4A shows, after 18 hours of VP-16 treatment, a population with strongly elevated mitochondrial calcium level was present in 32D cells and, to a lesser extent, in C2 cells. A total of 57% of 32D cells (n = 3; 52.6% ± 4.6%) possessed mitochondria overloaded with calcium. The caspase inhibitor had no effect on the mitochondrial uptake in 32D cells (61.6%), but significantly prevented the mitochondrial calcium overload observed in C2 cells from 42.8% (n = 3; 40.6% ± 5.2%) to 17.1% (n = 3; 15.1% ± 2.9%). These results confirm that the mitochondrial calcium uptake observed in 32D cells occurs independently of caspase activation, in contrast to C2 cells, where it is a secondary event and a consequence of caspase-dependent, downstream events. These data suggest that 32D cells may be able to activate a caspase-independent, calcium-dependent apoptotic pathway, in contrast to the Bcr-Abl–expressing cell lines C2 and C4 where this pathway is blocked.
Data using protease inhibitors also indicated that 32D cells could not be rescued from cell death by caspase inhibition, whereas C2 cells are protected (Figure 4B). Calpain inhibitors are also slightly protective for 32D cells, whereas they have no effect on C2 cells. These data further suggest 32D cells possess a distinct pathway for apoptosis initiation, which can still cause apoptosis when caspases are inhibited. Calpain activity may partially contribute to the death process because inhibitors marginally reduce cell death. In contrast, C2 cells depend on a pathway in which caspase initiation is a critical feature because they can be rescued by zVAD. C4 cells (high Bcr-Abl expression) do not initiate either pathway and are drug resistant.
Differential effects of the protease inhibitors zVAD and calpain inhibitor III on mitochondrial calcium content and cell death in 32D and C2 cells. (A) 32D and C2 cells were treated with VP-16 or preincubated for 1 hour with 50 μM zVAD followed by VP-16. After 18 hours of incubation cells were loaded with 1 μM Rhod-2 and analyzed by flow cytometry on the FL-2 channel. Untreated cells are presented as the filled histograms and treated as the open histograms. Histograms representative of 3 independent experiments are shown. (B) Cells were treated with VP-16 or preincubated for 1 hour with various concentrations of zVAD or calpain inhibitor III (as indicated). After 18 hours, cell viability was analyzed by PI uptake on the FL-2 channel. Percentage of PI+ (% cell death) cells is shown as the mean ± SD of 3 independent experiments. *P < .05; **P < .005 versus VP-16–treated group (Student t test).
Differential effects of the protease inhibitors zVAD and calpain inhibitor III on mitochondrial calcium content and cell death in 32D and C2 cells. (A) 32D and C2 cells were treated with VP-16 or preincubated for 1 hour with 50 μM zVAD followed by VP-16. After 18 hours of incubation cells were loaded with 1 μM Rhod-2 and analyzed by flow cytometry on the FL-2 channel. Untreated cells are presented as the filled histograms and treated as the open histograms. Histograms representative of 3 independent experiments are shown. (B) Cells were treated with VP-16 or preincubated for 1 hour with various concentrations of zVAD or calpain inhibitor III (as indicated). After 18 hours, cell viability was analyzed by PI uptake on the FL-2 channel. Percentage of PI+ (% cell death) cells is shown as the mean ± SD of 3 independent experiments. *P < .05; **P < .005 versus VP-16–treated group (Student t test).
Pretreatment with RuRed prevents mitochondrial calcium uptake and protects 32D cells from apoptosis induced by VP-16
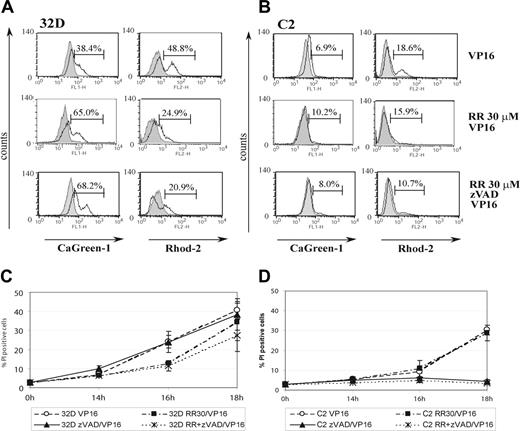
To investigate directly if the ER/mitochondrial calcium-coupling process is an important proapoptotic event in VP-16–treated 32D cells, we studied the effect of an inhibitor of the mitochondrial Ca2+ uniporter RuRed on the cytosolic and mitochondrial calcium levels and sensitivity to apoptosis. Because this uniporter is the primary channel for the uptake of calcium into mitochondria, its inhibition would be expected to induce a rise in the cytosolic calcium levels and a decrease in mitochondrial calcium levels. Representative histograms obtained by flow cytometry are presented in Figure 5. We initially confirmed that RuRed at 30 μM for 18 hours had no toxic effects, as determined by PI viability assay (not shown). Because we already know that caspases are activated during VP-16–induced apoptosis, we preincubated all cells for 1 hour with 30 μM RuRed in the presence or absence of 50 μM zVAD to protect them from caspase-dependent effects, followed by VP-16 treatment. RuRed strongly influenced the cytosolic and mitochondrial calcium changes observed in 32D cells after 16 hours of VP-16 treatment (Figure 5A). In VP-16–treated 32D cells the amount of cells with elevated mitochondrial calcium decreased from 48.8% (n = 6; 45.9% ± 7.1%) to 24.9% (n = 3; 29.2% ± 4.4%) and 20.9% (n = 5; 26% ± 3.9%) after pretreatment with 30 μM RuRed and RuRed plus zVAD, respectively. In RuRed-treated cells calcium uptake by the mitochondria was inhibited, and therefore also resulted in an elevated cytosolic calcium level in comparison to that observed in cells treated with VP-16 alone. The percentage of cells characterized by an increase of cytosolic level above normal values rose from 38.4% (n = 3; 39.6% ± 4.6%) to 65% (n = 3; 55.3% ± 9.5%) after pretreatment with 30 μM RuRed and to 68.2% (n = 4: 56% ± 6.4%). In C2 cells after 16 hours of VP-16 treatment, major changes in cytosolic or mitochondrial calcium were not visible and RuRed did not influence calcium levels (Figure 5B).
Pretreatment with RuRed prevents mitochondrial calcium uptake and protects 32D cells from apoptosis induced by VP-16. (A) 32D and (B) C2 cells were pretreated with either 30 μM RuRed (RR), or RR plus zVAD as indicated, followed by VP-16 treatment. After 16 hours cells were loaded with CaGreen-1 (for cytoplasmic Ca2+) or Rhod-2 (for mitochondrial Ca2+) and analyzed by flow cytometry. Representative histograms, corresponding to untreated cells (filled histograms), and VP-16 treated (open histograms) are shown. (C) 32D and (D) C2 cells were preincubated with 30 μM RR alone or with 50 μM zVAD as indicated for 1 hour followed by 7.5 μg/mL VP-16 treatment. Cell death was quantified at the time points indicated by measuring PI uptake by flow cytometry. Percent of dead cells is shown as the mean ± SD of 3 independent experiments. Four groups of treatment are shown: ○, treatment with VP-16; ♦, pretreatment with 50 μM zVAD followed by VP-16 treatment; ▪, pretreatment with 30 μM RR followed by VP-16 treatment; and X, pretreatment with 30 μM RR and 50 μM zVAD followed by VP-16 treatment.
Pretreatment with RuRed prevents mitochondrial calcium uptake and protects 32D cells from apoptosis induced by VP-16. (A) 32D and (B) C2 cells were pretreated with either 30 μM RuRed (RR), or RR plus zVAD as indicated, followed by VP-16 treatment. After 16 hours cells were loaded with CaGreen-1 (for cytoplasmic Ca2+) or Rhod-2 (for mitochondrial Ca2+) and analyzed by flow cytometry. Representative histograms, corresponding to untreated cells (filled histograms), and VP-16 treated (open histograms) are shown. (C) 32D and (D) C2 cells were preincubated with 30 μM RR alone or with 50 μM zVAD as indicated for 1 hour followed by 7.5 μg/mL VP-16 treatment. Cell death was quantified at the time points indicated by measuring PI uptake by flow cytometry. Percent of dead cells is shown as the mean ± SD of 3 independent experiments. Four groups of treatment are shown: ○, treatment with VP-16; ♦, pretreatment with 50 μM zVAD followed by VP-16 treatment; ▪, pretreatment with 30 μM RR followed by VP-16 treatment; and X, pretreatment with 30 μM RR and 50 μM zVAD followed by VP-16 treatment.
We then investigated whether the protective effect of RuRed on mitochondrial overload increases the viability of 32D cells incubated with VP-16. Cell death/PI permeability was assessed at 3 time points to assess the influence of inhibitors on the kinetics of cell death (Figure 5C). These experiments indicated that RuRed significantly reduced cell death in 32D cells at 16 hours and, in fact, the kinetics of cell death then resembled C2 cells. A combination of RuRed plus zVAD further protected 32D cells, suggesting that the caspase pathway is also active as death proceeds. zVAD alone is, however, unable to protect 32D cells from apoptosis (as noted in Figure 3), demonstrating the predominance of a caspase-independent pathway. In contrast, in C2 cells RuRed is not protective, suggesting that ER/mitochondrial coupling is irrelevant, whereas zVAD is strongly protective (Figure 5D).
These data indicate that a calcium-dependent apoptotic pathway is the primary initiator of apoptosis in 32D cells. In contrast, in C2 cells, Bcr-Abl reduces the amount of releasable ER calcium and therefore this Ca2+/ER pathway does not play a role. In both cells, other caspase-dependent pathways can be activated, and the cells are drug sensitive. Together, these results demonstrate that calcium signals, which contribute to the apoptotic process activated in 32D cells by VP-16, are not activated in Bcr-Abl–expressing cells. Furthermore, our combined data suggest that the high-expressing cells (C4 cells) have the capacity to inhibit at least 2 apoptosis-initiating pathways (Ca2+/ER pathway and caspase dependent; Figure 6), and have therefore obtained drug resistance.
Discussion
The data presented in this study suggest a novel function for the Bcr-Abl oncoprotein in the regulation of apoptotic pathways. We have demonstrated that a calcium-dependent, ER-initiated apoptotic pathway is inactive when Bcr-Abl is expressed and that this is likely to contribute to the prosurvival mechanisms associated with this oncogene.
We have detected significantly decreased ER calcium levels in both mouse and human Bcr-Abl–expressing cells and a weaker CCE response. This reduced CCE is analogous to the effect of other treatments that reduce ER Ca2+ levels, such as overexpression of Bcl-2 or prolonged culture in a low-calcium buffer.24,28 It has been suggested that sufficient and probably sustained ER calcium release, supported by CCE, has to occur to induce a calcium overload and loss of mitochondrial membrane potential (MMP) in the mitochondria.23 Because C2 and C4 cells released 2-fold less calcium from the ER, the corresponding CCE in these cells was also weaker. This reduction in both ER Ca2+ levels and CCE may severely compromise all calcium-dependent apoptotic signaling in Bcr-Abl–expressing cells. Imatinib treatment could restore ER calcium levels, indicating dependence on Bcr-Abl activity, whereas Bcl-2 down-regulation had no effect.
To assess the calcium-dependent apoptotic response, we treated cells with VP-16, a chemotherapeutic drug that has been reported to induce both classic BH3/mitochondria-dependent and ER calcium–dependent apoptotic pathways.22 C4 cells displayed a highly drug-resistant phenotype, as previously described.16 Their ER calcium status was similar to C2 cells, but due to complete drug resistance (ie, more than one pathway blocked), they could not contribute further to the analysis. Subsequent studies compared 32D to C2 cells. After apoptosis induction, at early time points, when the percentage of cell death did not exceed 15%, cytosolic calcium changes were not observed in C2 cells and mitochondrial uptake was weak. In contrast, a strong elevation of cytosolic Ca2+ and mitochondrial Ca2+ uptake was evident in 32D cells.
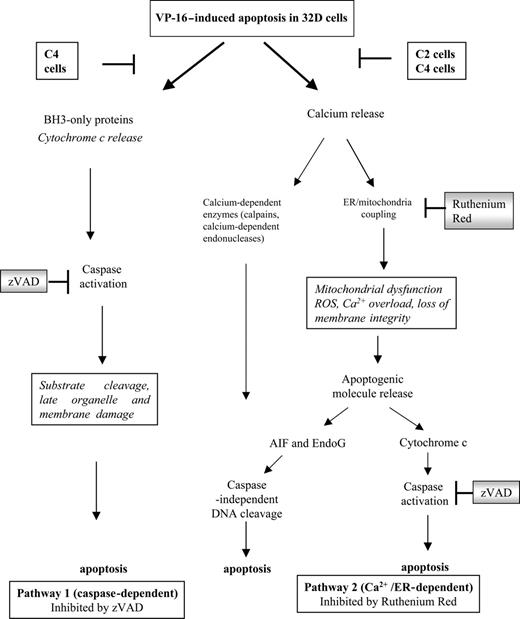
Available apoptosis pathways in 32D myeloid hematopoietic cells. In this model we propose that 32D cells have at least 2 pathways by which death can be initiated. Pathway 1 is often referred to as “classical” and involves BH3 protein activation leading to cytochrome c release and caspase activation. Calcium release from damaged organelles is a late event and is prevented by caspase inhibitors. Pathway 2 is initiated by calcium release, leading to activation of calcium-dependent cytosolic enzymes and ER/mitochondrial Ca2+ coupling. This has been proposed to generate reactive oxygen species (ROS) and lead to irreversible mitochondria damage. This process is likely to release apoptogenic factors including cytochrome c and, therefore, caspase activation, but also apoptosis-inducing factor (AIF) and endonuclease G (EndoG), which will subsequently lead to caspase-independent DNA fragmentation. Although late caspase activation will occur, inhibition cannot prevent the calcium accumulation or rescue the cells from apoptosis as other pathways contribute to cell death (activation of calcium-dependent cytosolic proteases, endonucleases, ER/mitochondrial coupling leading to mitochondrial dysfunction and release of apoptogenic factors). Evidence from our study suggests that Bcr-Abl–expressing cells are deficient in the calcium-dependent pathway. Cells expressing high levels of Bcr-Abl (C4 cells) are also resistant to the classical/BH3 pathway. This dual resistance may make a major contribution to treatment failure in BC CML, where elevated expression of Bcr-Abl is a common feature.
Available apoptosis pathways in 32D myeloid hematopoietic cells. In this model we propose that 32D cells have at least 2 pathways by which death can be initiated. Pathway 1 is often referred to as “classical” and involves BH3 protein activation leading to cytochrome c release and caspase activation. Calcium release from damaged organelles is a late event and is prevented by caspase inhibitors. Pathway 2 is initiated by calcium release, leading to activation of calcium-dependent cytosolic enzymes and ER/mitochondrial Ca2+ coupling. This has been proposed to generate reactive oxygen species (ROS) and lead to irreversible mitochondria damage. This process is likely to release apoptogenic factors including cytochrome c and, therefore, caspase activation, but also apoptosis-inducing factor (AIF) and endonuclease G (EndoG), which will subsequently lead to caspase-independent DNA fragmentation. Although late caspase activation will occur, inhibition cannot prevent the calcium accumulation or rescue the cells from apoptosis as other pathways contribute to cell death (activation of calcium-dependent cytosolic proteases, endonucleases, ER/mitochondrial coupling leading to mitochondrial dysfunction and release of apoptogenic factors). Evidence from our study suggests that Bcr-Abl–expressing cells are deficient in the calcium-dependent pathway. Cells expressing high levels of Bcr-Abl (C4 cells) are also resistant to the classical/BH3 pathway. This dual resistance may make a major contribution to treatment failure in BC CML, where elevated expression of Bcr-Abl is a common feature.
Analysis of calpain activity in 32D and C2 cells further suggested that only 32D cells had sufficient cytosolic calcium for an early activation of these calcium-dependent proteases (Figure S3). In addition, differential effects of protease inhibitors indicated disparate death pathways in 32D and C2 cells. In 32D cells mitochondrial calcium uptake is an early event, part of a Ca2+-dependent signaling pathway, which is not inhibited by zVAD. In contrast, in Bcr-Abl–expressing C2 cells, it is a consequence of downstream events that are reversed by caspase inhibition. The source of the later caspase-dependent calcium rise in C2 cells may be (1) general loss of membrane integrity, to which caspases contribute, (2) caspase-dependent cleavage of a calcium channel in the plasma membrane,33,34 or (3) caspase-mediated cleavage of Bap31 to Bap 20 leading to ER calcium release.35
If ER/mitochondrial Ca2+ coupling is important for drug sensitivity, then direct inhibition of this pathway should also inhibit the zVAD-independent cell-death pathway in 32D cells. We therefore examined the effect of a mitochondrial Ca2+ uptake inhibitor, RuRed, on the apoptotic events induced by VP-16. RuRed has been previously shown to inhibit mitochondrial calcium uptake and protect from Ca2+-dependent cell death.22 RuRed pretreatment protected 32D cells from mitochondrial calcium uptake and diverted calcium to the cytosol. This significantly reduced apoptosis in these cells (at 16 hours) until other proteolytic enzymes became active at 18 hours. Addition of zVAD inhibited the caspase-dependent pathway and reduced apoptosis at 18 hours compared to RuRed only. zVAD has less effect on 32D cells than on C2 cells because other enzymes such as calpains will also contribute to cell death as shown in Figure 4 and Figure S3. These experiments confirmed that ER/mitochondria coupling is a critical part of VP-16–induced apoptosis in 32D cells, whereas it does not play a role in Bcr-Abl–expressing cells.
To our knowledge this is the first report that has shown a link between the Bcr-Abl oncoprotein and ER Ca2+ homeostasis. The mechanism by which Bcr-Abl reduces ER Ca2+ levels is unknown. ER calcium levels are strongly influenced by Bcl-2 family members, although the mechanism by which this is achieved is unknown, and the literature still contains some conflicting data.36 Studies have demonstrated that Bcl-2 overexpression reduces the steady-state ER Ca2+ concentration and limits mitochondrial calcium uptake.37,38 Depletion of Bcl-2 in Bcr-Abl–expressing cells did not, however, increase ER calcium levels, suggesting that it is not responsible for the decrease in ER releasable calcium. It remains possible that other ER-localized Bcl-2 family members contribute to this effect. Bax and Bak promote Ca2+ uptake into the mitochondria,21,39 and knockouts are resistant to a wide range of apoptotic stimuli, exhibit decreased ER calcium, and ineffective calcium delivery to the mitochondria.22
It is also possible that Bcr-Abl exerts a direct effect at the ER. This protein is a cytosolic tyrosine kinase; however, c-Abl has been reported to also be present in the ER40 and to be associated with a proapoptotic ER protein Aph-2.41 It is conceivable, therefore, that a portion of Bcr-Abl is also present at the ER and exerts a direct influence on ER homeostasis. Bcr-Abl expression can, however, influence various signaling pathways, including Ras and phosphatidylinositol 3-kinase. Of interest in this regard is a study indicating that the Ras-related G protein (R-Ras) alters Ca2+ homeostasis by increasing the calcium leak across the ER membrane.42 It is also possible that downstream signaling might influence existing channels that regulate calcium release, for example, the inositol-1,4,5-triphosphate receptor. In addition, ER calcium homeostasis can also be influenced by the cellular redox state and Bcr-Abl has been reported to increase ROS production.43
We propose that the drug resistance observed during the progression of CML is a consequence of the acquisition of various antiapoptotic functions of Bcr-Abl (Figure 6). Low and high Bcr-Abl–expressing cells show impaired ER homeostasis and are unable to activate ER calcium-mediated apoptotic pathways. However, low Bcr-Abl–expressing (C2) cells can still activate a classic BH3/mitochondrial pathway leading to caspase activation. In high Bcr-Abl–expressing, drug-resistant cells (C4) the classic BH3/mitochondrial pathway is also inhibited, as indicated by several previous studies including our own.12,44,45
There is still uncertainty regarding the molecular mechanisms of CML progression to blast crisis and the nature of drug resistance mechanisms occurring at this stage. This study provides a new insight into the repertoire of survival mechanisms that can be used by Bcr-Abl. It is clear that if we intend to combat drug resistance with combination regimens, we need a better understanding of available apoptosis pathways in cancer cells and classes of drugs that can adequately target them.
Prepublished online as Blood First Edition Paper, February 9, 2006; DOI 10.1182/blood-2005-04-1523.
Supported by grants from the Irish Cancer Society, the Higher Education Authority of Ireland, Health Research Board, and the Cork Cancer Research Centre.
K.P. and S.V. designed and performed research, analyzed data, and wrote the paper; T.G.C. and G.C.O'S. contributed reagents, expertise, and analytic tools; and S.L.M. conceived of and designed research, analyzed data, and wrote and edited the paper.
K.P. and S.V. contributed equally to this work.
The online version of this article contains a data supplement.
The publication costs of this article were defrayed in part by page charge payment. Therefore, and solely to indicate this fact, this article is hereby marked “advertisement” in accordance with 18 U.S.C. section 1734.
We thank Dr Karen Keeshan for early contributions to this study and Dr John Mackrill for valuable advice. We also thank mgr Bogusz Kulawiak for the help with TILL photonics. We are grateful to Lisa O'Connor for help with the siRNA transfection experiments. Imatinib was kindly provided by Novartis (Basel, Switzerland).

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal