Abstract
Background: Primary systemic amyloidosis (AL) is an incurable plasma cell disorder. Lenalidomide, especially in conjunction with dexamethasone, has been shown to be highly active in patients with multiple myeloma.
Methods: We studied the toxicity and efficacy of lenalidomide in patients with symptomatic AL. Patients received single agent lenalidomide. If progression by 3 months or no evidence of hematologic response after 3 cycles, dexamethasone was added. Originally, twenty-three patients (Cohort 1) were enrolled according to study design. Because of a significant early drop out rate and notable activity of the regimen, the trial was modified to include an additional 15 patients (Cohort 2). Baseline characteristics and adverse events are available for all enrolled patients, but at the time of this writing, response data are available for Cohort 1 patients due to short follow-up of Cohort 2, but will be updated by the time of the meeting.
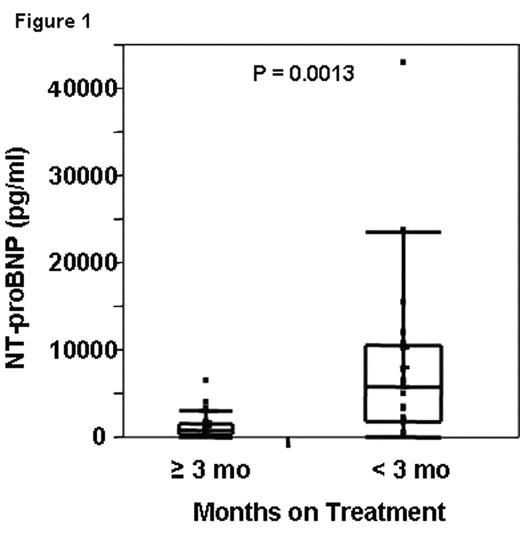
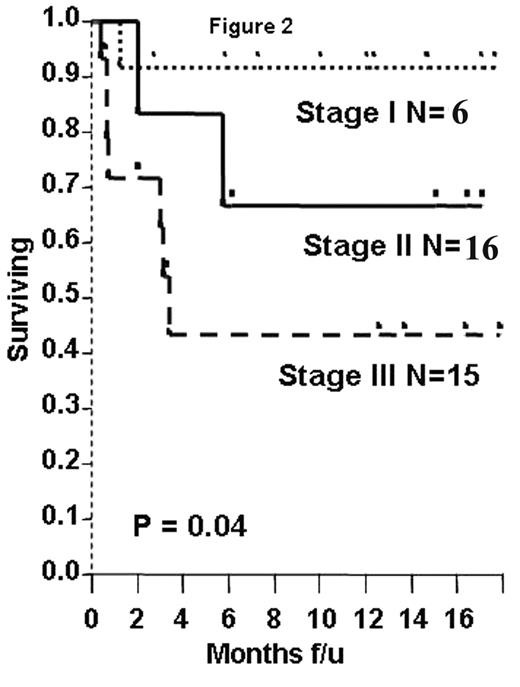
Results: Median age was 64 years, with 69% male. Twenty-three were previously treated. Organ involvement was cardiac (67%), renal (64%), hepatic (17%), nerve (17%). Thirty-three, twenty-two, and forty-four percent of patients were cardiac biomarker stage 1, 2, and, 3 respectively. Of the 37 patients, one was a cancel, and 6 have not yet made it through 3 months of protocol treatment and event monitoring. The respective median follow-ups for Cohorts 1 and 2 are 17 and 3.4 months. Of the remaining, 30 patients, within the first 3 cycles of therapy fifteen patients discontinued treatment: 7 early deaths and 8 adverse events or other causes. Three additional patients died 0.5 to 2 months after stopping treatment. The best predictor for early withdrawal and/or death was baseline NT-proBNP and cardiac biomarker staging system (cut-offs for serum troponin T <0.035 ng/ml and NT-proBNP <332 pg/ml--Stage I neither above cut-off; Stage III, both above cut-off; and Stage II, one above cut-off).
Of the twenty 22 patients assessed for response ten patients responded to treatment for an overall response rate of 45%, including 23% organ responders. Among the patients with organ responses, there were four renal responses, two cardiac responses and two liver responses. All but one of the responders had dexamethasone added to their treatment program. The most common grade 3−4 adverse events at least possibly attributable to lenalidomide were neutropenia (38%), thrombocytopenia (21%), fatigue (15%), and rash (12%).
Conclusions: Lenalidomide and dexamethasone has significant activity in patients with AL with an overall response rate of 45%. Baseline NT−proBNP may be an effective eligibility screening strategy for subsequent trials.
Disclosures: Clinical trial using lenalidomide and dexamethasone for amyloidosis.; Trial supported in part by Celgene.; Trial payed for in part by Celgene.
Author notes
Corresponding author

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal