Deregulated MYC expression has been implicated in the etiology of many human cancers, including hematopoietic malignancies. To explore the impact of widespread constitutive MYC expression in the hematopoietic compartment, we have used a vector containing regulatory elements of the Vav gene to generate transgenic mice. VavP-MYC mice are highly tumor-prone and the level of MYC was found to influence both the kinetics and nature of the malignancies that developed. Whereas aggressive T-cell lymphomas rapidly overwhelmed the highest-expressing line, late-onset monocytic tumors greatly predominated in 2 low-expressing lines. These monocytic tumors most likely arise from abnormal macrophage colony-stimulating factor (M-CSF)–dependent progenitor cells having enhanced self-generative capacity. There appears to be a sharp threshold for MYC-induced T-cell lymphomagenesis because merely doubling the MYC level in a low-expressing line by breeding homozygous transgenic animals switched the phenotype from primarily monocytic tumors to exclusively T-cell tumors. Even the low level of MYC, however, clearly affected T-cell cycling, size, and sensitivity to apoptosis, and coexpression of a BCL2 transgene promoted efficient T-cell lymphomagenesis. The implication is that MYC level affects the spontaneous acquisition of synergistic oncogenic mutations.
Introduction
MYC is a transcription factor that activates or represses a wide spectrum of target genes.1,2 In response to growth factors or cytokines, MYC levels are upregulated in the G1 phase of the cell cycle to promote cell growth and transition into S phase.3 Deregulated expression of MYC can also inhibit differentiation, induce genomic instability, and sensitize cells to apoptosis.4,5 The apoptotic response is proposed to be a safety mechanism that deletes cells with deregulated, potentially oncogenic MYC.6
The role of MYC in human cancer was first established in Burkitt lymphoma, where chromosome translocation links MYC to immunoglobulin (Ig) loci and subjugates its expression to Ig enhancers.7 MYC has now been implicated in the etiology of a wide variety of hematologic8,9 and other malignancies.3,10 The deregulating mutations impact either on MYC itself, as for example in translocations or amplifications, or on upstream regulatory pathways. The stability of MYC protein is tightly regulated, and the level of MYC may also be important during tumorigenesis, because secondary mutations that increase MYC stability have been observed for the translocated MYC allele in Burkitt lymphoma.11-13
Valuable insights into the mechanism of MYC-induced tumorigenesis have come from studies of transgenic mice, particularly the Eμ-Myc mouse,14,15 which models the major chromosome translocation found in Burkitt lymphoma. In these mice, Myc is expressed under the control of the IgH enhancer (Eμ), and every mouse succumbs to a clonal pre-B or B-cell lymphoma, preceded by a benign proliferative phase during which expansion of the B lymphoid compartment is held within tolerable limits by apoptosis.16,17 Lymphomagenesis in Eμ-Myc mice is markedly accelerated by the introduction of antiapoptotic mutations, as first demonstrated by overexpression of the prosurvival gene BCL2.18 Furthermore, tumors arising spontaneously in Eμ-Myc mice carry mutations in the p19Arf-Mdm2-p53 pathway that disable p53-induced apoptosis.19,20 The demonstration in transgenic mice that suppression of MYC can cause tumor regression21 is encouraging the development of therapeutic strategies that target MYC or its upstream regulators.22
In order to assess the oncogenic impact of MYC in a wider range of hematopoietic cell types, we have generated several lines of transgenic mice in which MYC is expressed in a vector containing regulatory elements of the mouse Vav gene (VavP). The VavP transgene directs expression in every nucleated hematopoietic cell examined, including both differentiated and progenitor cells.17,23,24 We have recently described the line with the highest expression (VavP-MYC17) in which the mice develop early-onset T lymphomas.25 Here we characterize 3 further lines of VavP-MYC mice, and show that MYC provokes very different tumor phenotypes depending on the level of its expression. Furthermore, we show that MYC has marked effects on the development of megakaryocytes and macrophages.
Materials and methods
VavP-MYC mice
All experiments were carried out in accordance with the National Health and Medical Research Council (Canberra, Australia) Code of Practice for the Care and Use of Animals for Scientific Purposes (2004) and approved by the Melbourne Health Research Directorate Animal Ethics Committee.
All mice were of the C57BL/6J (B6) background and bred in the facilities of this institute. Transgene construction and the generation of VavP-MYC mice by pronuclear injection were as previously described.25 Three lines were bred from primary VavP-MYC mice: MYC17 (with 11 copies of the transgene), MYC10 (7 copies), and MYC12 (3 copies). Transgenic offspring were identified by polymerase chain reaction (PCR) of DNA prepared from tail biopsies or blood using primers to the vector SV40 polyadenylation signal (forward: gccgcagacatgataagatacatt; reverse: tcggctcgcgaggttttac).
Mice homozygous for the VavP-MYC10 locus (MYC10hom mice) were generated by crossing MYC10 mice. Quantitative slot-blotting of tail biopsy DNA was used to distinguish wild-type, MYC10, and MYC10hom littermates. Tail DNA was quantified using a fluorometer after staining with Hoechst 33258 and then 6 μg of denatured DNA was slot-blotted onto nylon membranes and fixed by ultraviolet (UV) irradiation. The 32P-labeled probe was prepared by random primer labeling of a 323-bp fragment of the VavP-MYC transgene (generated by PCR with the primers gcggccgcagacatgataa and actagtggatccgctggcg). The membrane was prehybridized and hybridized at 65°C in modified Church and Gilbert buffer (7% SDS, 10 mM EDTA, 0.5 M sodium phosphate [pH 7.2], and 100 μg/mL sonicated and denatured salmon sperm DNA) and washed at 65°C in 0.5 × standard saline citrate (SSC; 1× = 150 mM sodium chloride and 15 m sodium citrate, pH 7.0)/0.1% SDS.
Bitransgenic MYC10-BCL2.69 mice were generated by in vitro fertilization26 of eggs from approximately 4-week-old VavP-MYC10 mice using sperm from VavP-BCL2.69 mice. The VavP-MYC transgene was detected by PCR using a forward primer to the MYC cDNA (ccaagcagaggagcaaagctcatt) and the reverse SV40 polyadenylation signal primer. VavP-BCL2.69 mice were identified by their high blood-leukocyte count27 determined with a Coulter Z2 particle count and size analyzer (Beckman-Coulter, Miami, FL).
Platelet numbers and size were determined with a hematology analyzer (Advia 120; Bayer, Tarrytown, NY). Tumor transplantation tests and histologic analyses were performed as previously described.25 Sections were stained with hematoxylin and eosin (H&E), mounted in Safety Mount No. 4 (Fronine, Sydney, Australia), and observed using an Optiphot microscope (Nikon, Tokyo, Japan) with Plan Apo × 40 (NA 0.95) or Plan Apo × 100 (NA 1.35, oil) objective lenses. Images were captured with a Nikon DS camera head (DS-5M; Nikon, Melville, NY) and control unit (DS-L1) using integral software.
Expression analysis
Western blots were performed using 40 μg of protein extract per lane as previously described25 but using a different rabbit MYC antibody (no. 11917; Abcam, Cambridge, United Kingdom). For reverse transcriptase (RT)–PCR, total cellular RNA was prepared from cell suspensions using the QIAshredder, RNeasy mini kit, and RNase-Free DNase set (Qiagen, Hilden, Germany) as described by the manufacturer. For each RNA sample, 4 25-μL OneStep RT-PCR reactions (Qiagen) were carried out using serially 5-fold less RNA (100 ng, 20 ng, 4 ng, and 0.8 ng). Reverse transcription was at 50°C for 30 minutes, followed by heat inactivation at 95°C for 15 minutes. PCR products were synthesized using 35 cycles of 94°C for 30 seconds, 64°C for 30 seconds, 72°C for 60 seconds, and resolved on 2% agarose gels. Transgene-expressed human MYC RNA was detected as a 379-bp PCR product synthesized using a forward primer (accagaaagttaactggcctgtacg) spanning VavP vector exonic sequences either side of the SV40 late intron and a reverse primer (accatctccagctggtcgg) in MYC cDNA. Endogenous β-Actin RNA was detected as a 688-bp product synthesized using a forward primer in exon 1 (ctttgcagctccttcgttgc) and a reverse primer in exon 4 (ctctttgatgtcacgcacgatttc).
Flow cytometry and cell-cycle analysis
Flow cytometry and sorting was performed as previously described.25 For cell-cycle analyses, 2.5 × 105 sorted thymocytes were stained with propidium iodide (100 μg/mL) in 400 μL of hypotonic buffer (10 mM Tris-HCl [pH 8], 1 mM NaCl, 0.1% [vol/vol] NP-40, and 10 μg/mL RNase A) at 4°C for 60 minutes. Flow cytometric data were analyzed with the ModFit LT 3.0 program (Verity Software House, Topsham, ME) to determine the proportions of cells in the G0/G1, S, and G2/M phases of the cell cycle.
Thymocyte survival assays
Thymocytes (1 × 106) were cultured in 24-well plates in 1 mL Dulbecco modified Eagle medium (DMEM) supplemented with 10% fetal calf serum (FCS), 100 μM L-asparagine and 50 μM 2-mercaptoethanol. Following 0, 1, 2, and 3 days in culture, replicate wells were harvested and the cells transferred to tubes containing 20 × 103 10-μm-diameter latex beads (Beckman-Coulter) and surface-stained with biotin–anti-CD4/streptavidin-PE and FITC–anti-CD8. The cell and latex-bead mixture was washed and resuspended in 200 μL balanced salt solution containing 2% FCS and 2 μg/mL propidium iodide. For flow cytometry, the latex beads were detected in a forward–versus–side light scatter gate and data were acquired until 4 × 103 beads had been counted. The number of viable (unstained by propidium iodide) CD4+ CD8+, CD4+ CD8–, and CD4– CD8+ thymocytes acquired per sample was determined, and the proportion relative to day 0 in culture calculated.
Colony-forming assays
Marrow cells (2.5 × 104) were cultured for 7 days in 1 mL DMEM containing 20% newborn calf serum and 0.3% agar and cytokines, as previously described.28 Cytokines were purified recombinant murine molecules (PeproTech, Rocky Hill, NJ) or recombinant human granulocyte (G)–CSF (Amgen, Thousand Oaks, CA), used at the following concentrations: G-CSF, M-CSF, granulocyte-macrophage (GM)–CSF, interleukin-3 (IL-3), and leukemia inhibitory factor (LIF) at 10 ng/mL; stem-cell factor (SCF) and IL-6 at 100 ng/mL; erythropoietin (EPO) at 2 IU/mL; thrombopoietin (TPO) at 50 ng/mL; interferon γ (IFNγ) at 2000 U/mL; and FMS-like tyrosine kinase 3 ligand (Flt3L) at 500 ng/mL. Differential colony counts were performed on fixed whole-mount preparations stained for acetylcholinesterase and with Luxol fast blue and hematoxylin. For recloning studies, individual 7-day colonies were resuspended in 8 mL of agar medium and then cultured for 7 days in duplicate dishes.
Results
VavP-MYC mouse lines differ in transgene expression levels
Three lines of mice were generated by pronuclear microinjection of human MYC cDNA in a Vav enhancer/promoter vector (VavP-MYC17, VavP-MYC10, and VavP-MYC12 mice; hereafter referred to as MYC17, MYC10, and MYC12 mice), and a fourth line was produced by breeding mice homozygous for the MYC10 locus (MYC10hom mice). Analysis of MYC protein levels in thymocytes from young healthy mice (Figure 1A) indicated that the expression level varied significantly between these lines (MYC17 > MYC10hom > MYC10 > MYC12). As reported for Eμ-Myc transgenic mice,29 expression of endogenous MYC was effectively repressed, even in the line with the lowest expression (Figure 1A), presumably due to a negative feedback mechanism.
MYC17 mice have been shown previously by Western blotting to express the transgene in the B, T, and myeloid lineages, with expression being highest in the T lineage.25 In the lower-expressing transgenic lines protein expression was not readily detected outside the T lineage, but RT-PCR analysis of tissues and cells from MYC10 mice (Figure 1B) indicated that the transgene was expressed in thymocytes, splenocytes, bone marrow cells, and in sorted populations of pre-B cells (B220+ IgM–), B cells (B220+ IgM+), monocytes (Mac1+ Gr1–), and granulocytes (Mac1+ Gr1+). RNA levels were somewhat lower in B cells (and, correspondingly, in the spleen) than in the other cell types.
VavP-MYC lines with a range of transgene expression levels. (A) Differing expression levels of transgenic MYC protein in MYC17, MYC10, MYC10hom (homozygous for the transgene locus), and MYC12 mice. Western blot of thymocyte lysates prepared from healthy young transgenic and wild-type mice were probed with anti-MYC antibody. Note that expression of endogenous Myc (a slower-migrating, broad, phosphorylated band) is repressed in the transgenic samples. The same filter probed with anti–β-actin is shown as a loading control. (B) Widespread expression of transgenic MYC RNA in MYC10 mice. Semiquantitative RT-PCR analysis of RNA prepared from thymocytes, splenocytes, bone marrow cells, and sorted pre-B cells (B220+ IgM–), B cells (B220+ IgM+), monocytes (Mac1+ Gr1–), and granulocytes (Mac1+ Gr1+) from 8-week-old MYC10 mice. Analyses of RNA with the reverse transcription reaction omitted (–RT) confirms specificity of PCR primers for transgene-expressed MYC RNA. RT-PCR reactions from serial 5-fold dilutions of input RNA are shown. Analysis of endogenous β-Actin RNA is shown as a control.
VavP-MYC lines with a range of transgene expression levels. (A) Differing expression levels of transgenic MYC protein in MYC17, MYC10, MYC10hom (homozygous for the transgene locus), and MYC12 mice. Western blot of thymocyte lysates prepared from healthy young transgenic and wild-type mice were probed with anti-MYC antibody. Note that expression of endogenous Myc (a slower-migrating, broad, phosphorylated band) is repressed in the transgenic samples. The same filter probed with anti–β-actin is shown as a loading control. (B) Widespread expression of transgenic MYC RNA in MYC10 mice. Semiquantitative RT-PCR analysis of RNA prepared from thymocytes, splenocytes, bone marrow cells, and sorted pre-B cells (B220+ IgM–), B cells (B220+ IgM+), monocytes (Mac1+ Gr1–), and granulocytes (Mac1+ Gr1+) from 8-week-old MYC10 mice. Analyses of RNA with the reverse transcription reaction omitted (–RT) confirms specificity of PCR primers for transgene-expressed MYC RNA. RT-PCR reactions from serial 5-fold dilutions of input RNA are shown. Analysis of endogenous β-Actin RNA is shown as a control.
Rate of tumorigenesis and tumor type correlates with transgene expression levels
All mice in each of the 4 lineages succumbed to tumors within the first 18 months of life and, significantly, median survival was inversely proportional to the level of MYC. The line with the lowest expression, MYC12, had a median survival of 58 weeks compared with 41 weeks for MYC10 mice, 13 weeks for MYC10hom mice, and 8 weeks for MYC17 mice (Figure 2).
In marked contrast to the MYC17 mice, which succumbed to very early onset T-cell lymphoma,25 all MYC12 mice autopsied (n = 24) and 93% of the MYC10 mice (n = 112) developed tumors with the histologic appearance of histiocytic sarcoma (Figure 3A).30 All tumors analyzed by flow cytometry (10 from MYC10 mice and 5 from MYC12 mice) expressed the myeloid markers F4/80 and Mac1 but not the granulocyte marker Gr1 (Figure 3B). They were also negative for B-lineage markers, including B220 and CD19; T-lineage markers, including Thy1 and CD3; and the erythroid marker Ter119 (Figure 3B; not shown). The histiocytic sarcomas were therefore tumors in the monocyte or macrophage lineage.
In the MYC10 mice, the macrophage tumors were widely disseminated throughout the hematopoietic tissues, with large deposits in 1 or more lymph nodes and also in the spleen, bone marrow, and thymus (90%, 80%, 60%, and 40% of mice, respectively). Tumor infiltration of the liver, lung, and kidney was frequent (80%, 70%, and 40% of mice, respectively). In addition, 40% of the mice had deposits that had spread locally through the dermis, hypodermis, muscle, and fat of the skin. In the MYC12 mice, similar cutaneous histiocytic tumors were the primary feature (80% of cases) and deposits elsewhere (in hematopoietic tissues, liver, lung, and kidney) were variable and generally less extensive than in MYC10 mice. None of the histiocytic tumors was accompanied by monocytic leukemia, but granulocytosis (presumably as a response to tumor15 ) was sometimes noted. Of the 5 MYC10 tumors tested, all were transplantable.
A small proportion of MYC10 mice (7%) succumbed to lymphoblastic lymphoma rather than histiocytic sarcoma. Significantly, these tumors were earlier in onset and all 5 were T-cell lymphomas; they expressed the T-lineage markers Thy1, TCRβ, and CD3, and lacked CD19, Ter119, and Mac1 (not shown). Four were CD4+ CD8+ pre–T-cell lymphomas, and 1 was a CD4+ CD8– mature T-cell lymphoma. These tumors resembled the T-cell lymphomas seen in the high expressing (MYC17) line,25 which developed mostly CD4+ CD8– and a significant minority of CD4+ CD8+ lymphomas.
The observations that most of the tumors arising in the 2 lowest-expressing lines were slow-developing macrophage tumors, and that the few early-onset tumors were T-cell lymphomas, as in the high-expressing (MYC17) line,25 suggested that there might be a threshold level of MYC expression required for efficient T-cell lymphomagenesis. We tested this directly by ascertaining the tumor susceptibility of MYC10hom mice, which expressed approximately twice as much MYC in thymocytes as MYC10 mice (see Figure 1A). Significantly, MYC10hom mice had a greatly shortened lifespan compared with MYC10 mice (median of 13 weeks rather than 41 weeks) and all succumbed to T-cell lymphoma. The 21 tumors analyzed histologically were all lymphoblastic in morphology (Figure 3C), and by flow cytometric analysis 20 were CD4+ CD8+ pre–T-cell lymphomas (Figure 3D), and 1 was a CD4+ CD8– mature T-cell lymphoma. The tumors presented as a grossly enlarged thymus (apart from the mature T-cell lymphoma) and the involvement of other organs was variable, except for the few leukemic mice (15%), which had extensive tumor infiltration of many organs. In occasional mice (15%), patches of histiocytic sarcoma were also apparent histologically.
Preneoplastic changes primarily affect megakaryocytes and macrophage precursors
In contrast to the marked preneoplastic phenotype of MYC17 mice,25 that of MYC10 and MYC10hom mice were relatively mild. Flow cytometry indicated a generally modest increase in the frequency of all cell types analyzed in the spleen (pre-B cells, B cells, CD4+ CD8– and CD4– CD8+ T cells, monocytes, granulocytes, and erythroid cells), and the increase was greater for MYC10hom than MYC10 mice (Table 1). Cell numbers were near normal in the bone marrow, thymus, and blood (not shown) but many cell types were slightly enlarged, including pre-B cells, B cells, CD4+ CD8– and CD4– CD8+ T cells, and monocytes (Figure 4B; not shown).
Tumorigenesis in VavP-MYC mice.MYC10 (n = 53), MYC10hom (n = 29), MYC12 (n = 19), MYC17 (n = 23; data from Smith et al25), and wild-type (B6) (n = 25) mice were monitored for tumors and killed when sick. The percentage of mice surviving at weekly intervals is shown. Log-rank analysis comparing wild-type versus MYC12, MYC12 versus MYC10, MYC10 versus MYC10hom, and MYC10hom versus MYC17 showed that in all cases survival was significantly different (P < .001).
Tumorigenesis in VavP-MYC mice.MYC10 (n = 53), MYC10hom (n = 29), MYC12 (n = 19), MYC17 (n = 23; data from Smith et al25), and wild-type (B6) (n = 25) mice were monitored for tumors and killed when sick. The percentage of mice surviving at weekly intervals is shown. Log-rank analysis comparing wild-type versus MYC12, MYC12 versus MYC10, MYC10 versus MYC10hom, and MYC10hom versus MYC17 showed that in all cases survival was significantly different (P < .001).
Distinct tumor types in MYC10 and MYC10hommice. (A) High-power view of a MYC10 thymic macrophage tumor, comprising large cells with bulky, variably vacuolated cytoplasm and a vesicular, sometimes pleomorphic nucleus, characteristics of histiocytic sarcoma. H&E-stained section; scale bar equals 20 μm. (B) Flow cytometric analysis of typical MYC10 macrophage (Mac1+ F4/80+ Gr1– CD19– Thy1–) tumor. The percentage of cells in each quadrant is indicated. (C) High-power view of a thymic MYC10hom pre–T-cell lymphoma with lymphoblastic morphology, comprising smaller cells with indistinct cell borders. (D) Flow cytometric analysis of typical MYC10hom pre–T-cell (Thy1+ TCRβ+ CD4+ CD8+ CD19–) lymphoma.
Distinct tumor types in MYC10 and MYC10hommice. (A) High-power view of a MYC10 thymic macrophage tumor, comprising large cells with bulky, variably vacuolated cytoplasm and a vesicular, sometimes pleomorphic nucleus, characteristics of histiocytic sarcoma. H&E-stained section; scale bar equals 20 μm. (B) Flow cytometric analysis of typical MYC10 macrophage (Mac1+ F4/80+ Gr1– CD19– Thy1–) tumor. The percentage of cells in each quadrant is indicated. (C) High-power view of a thymic MYC10hom pre–T-cell lymphoma with lymphoblastic morphology, comprising smaller cells with indistinct cell borders. (D) Flow cytometric analysis of typical MYC10hom pre–T-cell (Thy1+ TCRβ+ CD4+ CD8+ CD19–) lymphoma.
Altered composition of the spleen in healthy young MYC10 and MYC10hom mice
. | Cell no. per spleen, × 106* . | . | . | . | . | ||||
|---|---|---|---|---|---|---|---|---|---|
| Cell type . | Wild-type . | MYC10 . | Ratio†(P) . | MYC10hom . | Ratio†(P) . | ||||
| Total | 180 ± 20 | 240 ± 20 | 1.3 (.03) | 310 ± 40 | 1.7 (.01) | ||||
| Pre-B | 2.9 ± 0.3 | 4.1 ± 0.5 | 1.4 (.06) | 7.2 ± 0.1 | 2.5 (< .01) | ||||
| B | 110 ± 10 | 140 ± 10 | 1.3 (.03) | 190 ± 20 | 1.7 (.01) | ||||
| CD4+ CD8- T | 31 ± 4 | 42 ± 2 | 1.4 (.03) | 44 ± 2 | 1.4 (.02) | ||||
| CD4- CD8+ T | 18 ± 2 | 24 ± 2 | 1.3 (.06) | 24 ± 2 | 1.3 (.07) | ||||
| Erythroid | 0.9 ± 0.3 | 2.8 ± 0.6 | 3.1 (.02) | 9.5 ± 3.2 | 10.6 (.03) | ||||
| Monocyte | 5.2 ± 0.8 | 6.4 ± 0.4 | 1.2 (.13) | 8.6 ± 1.1 | 1.7 (.03) | ||||
| Granulocyte | 4.0 ± 0.2 | 4.5 ± 0.6 | 1.1 (.23) | 10 ± 3 | 2.5 (.04) | ||||
. | Cell no. per spleen, × 106* . | . | . | . | . | ||||
|---|---|---|---|---|---|---|---|---|---|
| Cell type . | Wild-type . | MYC10 . | Ratio†(P) . | MYC10hom . | Ratio†(P) . | ||||
| Total | 180 ± 20 | 240 ± 20 | 1.3 (.03) | 310 ± 40 | 1.7 (.01) | ||||
| Pre-B | 2.9 ± 0.3 | 4.1 ± 0.5 | 1.4 (.06) | 7.2 ± 0.1 | 2.5 (< .01) | ||||
| B | 110 ± 10 | 140 ± 10 | 1.3 (.03) | 190 ± 20 | 1.7 (.01) | ||||
| CD4+ CD8- T | 31 ± 4 | 42 ± 2 | 1.4 (.03) | 44 ± 2 | 1.4 (.02) | ||||
| CD4- CD8+ T | 18 ± 2 | 24 ± 2 | 1.3 (.06) | 24 ± 2 | 1.3 (.07) | ||||
| Erythroid | 0.9 ± 0.3 | 2.8 ± 0.6 | 3.1 (.02) | 9.5 ± 3.2 | 10.6 (.03) | ||||
| Monocyte | 5.2 ± 0.8 | 6.4 ± 0.4 | 1.2 (.13) | 8.6 ± 1.1 | 1.7 (.03) | ||||
| Granulocyte | 4.0 ± 0.2 | 4.5 ± 0.6 | 1.1 (.23) | 10 ± 3 | 2.5 (.04) | ||||
Eight-week-old mice were analyzed. Total nucleated counts were determined with a Coulter counter and subpopulations by flow cytometry. Cell types examined were pre-B (B220+ IgM-), B (B220+ IgM+), erythroid (Ter119+), granulocyte (Gr1+ Mac1+), monocyte (Gr1- Mac1+), and T-cell subpopulations (based on CD4/CD8 expression). P for a t test.
Mean ± SEM; n = 3.
Ratio of MYC10 or MYC10hom to wild-type control.
Megakaryocytopoiesis was strikingly abnormal in VavP-MYC mice. In MYC17 mice, there were substantially more megakaryocytes in the spleen at 2 weeks (Smith et al25 ; Table 2) and in the bone marrow by 4 weeks (Table 2). Megakaryocyte numbers were also significantly increased in the spleen and bone marrow of MYC10hom mice, but not MYC10 mice (Table 2). Notably, the megakaryocytes were consistently larger (Table 2) and they had a more complex nucleus. Further histologic examination revealed that in MYC17, MYC10hom, and to a lesser extent, MYC10 mice, many megakaryocytes in the bone marrow contained other leukocytes, almost all of them intact granulocytes (Figure 5A; Table 2), presumably as a result of emperipolesis (“Discussion”). Concomitant with megakaryocyte abnormalities, MYC10hom and MYC17 mice produced a reduced number of circulating platelets (approximately 2-fold and 5-fold fewer, respectively) and those present were larger, but there was little change in MYC10 mice (Table 2).
Megakaryocyte and platelet abnormalities in healthy young VavP-MYC mice
. | Bone marrow megakaryocytes . | . | . | Spleen megakaryocytes . | Platelets§ . | . | |||
|---|---|---|---|---|---|---|---|---|---|
. | No.* . | Size, mm† . | Internalized cells, %‡ . | No.* . | × 107/mL . | Vol, fL . | |||
| Wild-type, 2-4 wk | 4.7 ± 0.5 | 23 ± 2 | 2 | 3.6 ± 0.4 | 104 ± 13 | 5.3 ± 0.1 | |||
| MYC17, 2-4 wk∥ | 7.2 ± 0.5 | 46 ± 2 | 57 | 7.1 ± 0.6 | 20 ± 3 | 8.4 ± 0.6 | |||
| Wild-type, 5-7 wk | 6.2 ± 0.6 | 29 ± 1 | 0 | 1.9 ± 0.3 | 130 ± 7 | 5.8 ± 0.1 | |||
| MYC10het, 5-7 wk | 7.1 ± 0.4¶ | 38 ± 1∥ | 20∥ | 2.0 ± 0.3¶ | 113 ± 6¶ | 6.2 ± 0.1∥ | |||
| MYC10hom, 5-7 wk∥ | 9.5 ± 0.6 | 50 ± 2 | 58 | 3.4 ± 0.4 | 49 ± 3 | 7.1 ± 0.1 | |||
. | Bone marrow megakaryocytes . | . | . | Spleen megakaryocytes . | Platelets§ . | . | |||
|---|---|---|---|---|---|---|---|---|---|
. | No.* . | Size, mm† . | Internalized cells, %‡ . | No.* . | × 107/mL . | Vol, fL . | |||
| Wild-type, 2-4 wk | 4.7 ± 0.5 | 23 ± 2 | 2 | 3.6 ± 0.4 | 104 ± 13 | 5.3 ± 0.1 | |||
| MYC17, 2-4 wk∥ | 7.2 ± 0.5 | 46 ± 2 | 57 | 7.1 ± 0.6 | 20 ± 3 | 8.4 ± 0.6 | |||
| Wild-type, 5-7 wk | 6.2 ± 0.6 | 29 ± 1 | 0 | 1.9 ± 0.3 | 130 ± 7 | 5.8 ± 0.1 | |||
| MYC10het, 5-7 wk | 7.1 ± 0.4¶ | 38 ± 1∥ | 20∥ | 2.0 ± 0.3¶ | 113 ± 6¶ | 6.2 ± 0.1∥ | |||
| MYC10hom, 5-7 wk∥ | 9.5 ± 0.6 | 50 ± 2 | 58 | 3.4 ± 0.4 | 49 ± 3 | 7.1 ± 0.1 | |||
Mean frequency in tissue sections (± SEM) per high-power (× 400) microscope field. For each tissue, 20 to 48 fields were counted from 3 to 6 mice of each genotype and age. For MYC17 mice, the spleen was analyzed at 2 weeks and bone marrow was analyzed at 4 weeks. Note: the overall increase in splenic megakaryocytes in MYC mice is greater than suggested by these field counts because the average spleen size is larger than wild-type controls (3-fold for MYC17, approximately 1.3-fold for MYC10het, and 1.7-fold for MYC10hom mice). P for a t test.
Mean megakaryocyte diameter in tissue sections (± SEM); n = 60.
The percentage of megakaryocytes (n = 60) in tissue sections containing internalized cells (mainly intact granulocytes). The mean number of internalized cells per megakaryocyte section containing such cells was 2.6 ± 0.4 for MYC17, 1.2 ± 0.1 for MYC10het, and 2.1 ± 0.2 for MYC10hom.
Mean platelet counts (± SEM) and mean platelet volume (± SEM) in blood samples (n = 4-14).
P ≤ .001.
P ≥ .05.
To ascertain myeloid and megakaryocyte progenitor- and preprogenitor-cell numbers, bone marrow cells from MYC17 and MYC10 mice were cultured in semisolid medium in the presence of 12 different cytokines or combinations thereof. The frequency of different colony types that grow in such assays reflects the size and composition of the progenitor-cell compartment. In control cultures containing no added stimulus, no colonies developed, indicating that enforced expression of MYC had not conferred cytokine independence. In the presence of exogenous cytokines, colony number and composition were normal, with 1 remarkable exception: Cultures stimulated by M-CSF developed prominent, abnormally large, compact macrophage colonies (80% of colonies in MYC17 cultures and 50% of colonies in MYC10 cultures; Figure 5B). Furthermore, the total number of macrophage colonies was significantly increased in the bone marrow of MYC17 and, to a lesser degree, MYC10 mice (Table 3). The macrophage nature of these colonies was confirmed by flow cytometry; the cells were all strongly positive for the macrophage marker Mac1 but negative for T- and B-cell markers (CD3 and B220, respectively).
Macrophage colonies are increased in response to M-CSF in clonal cultures of VavP-MYC bone marrow cells
. | Mean no. colonies, ± SD . | . | . | . | . | . | |||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Stimulus . | Blast . | Granulocyte . | Granulocyte-macrophage . | Macrophage . | Eosinophil . | Megakaryocyte . | |||||
| Wild-type, 2 wk; n = 3 | |||||||||||
| GM-CSF | 0 | 15 ± 4 | 7 ± 4 | 17 ± 4 | 2 ± 1 | 0 | |||||
| M-CSF | 0 | 0.7 ± 0.6 | 3 ± 1 | 50 ± 17* | 0 | 0 | |||||
| IL-3 | 8 ± 2 | 12 ± 4 | 8 ± 3 | 11 ± 12 | 2 ± 2 | 5 ± 2 | |||||
| SCF + IL-3 + EPO | 8 ± 3 | 18 ± 6 | 12 ± 11 | 11 ± 12 | 2 ± 2 | 24 ± 5 | |||||
| MYC17, 2 wk; n = 3 | |||||||||||
| GM-CSF | 0 | 10 ± 2 | 4 ± 0 | 19 ± 8 | 2 ± 1 | 0 | |||||
| M-CSF | 0 | 0 | 2 ± 2 | 158 ± 48* | 0 | 0 | |||||
| IL-3 | 5 ± 2 | 9 ± 3 | 3 ± 3 | 13 ± 6 | 3 ± 3 | 6 ± 2 | |||||
| SCF + IL-3 + EPO | 8 ± 3 | 7 ± 3 | 3 ± 2 | 13 ± 15 | 0.7 ± 1.2 | 11 ± 4 | |||||
| Wild-type, 8 wk; n = 4 | |||||||||||
| GM-CSF | 0 | 16 ± 5 | 5 ± 2 | 31 ± 3 | 4 ± 2 | 0 | |||||
| M-CSF | 0 | 3 ± 1 | 6 ± 4 | 49 ± 8* | 0 | 0 | |||||
| IL-3 | 7 ± 4 | 19 ± 6 | 11 ± 3 | 15 ± 3 | 2 ± 1 | 7 ± 4 | |||||
| SCF + IL-3 + EPO | 11 ± 6 | 21 ± 2 | 13 ± 3 | 17 ± 3 | 2 ± 1 | 28 ± 4 | |||||
| MYC10, 8 wk; n = 4 | |||||||||||
| GM-CSF | 0 | 19 ± 6 | 5 ± 2 | 29 ± 15 | 3 ± 3 | 0 | |||||
| M-CSF | 0 | 3 ± 1 | 2 ± 1 | 109 ± 32* | 0 | 0 | |||||
| IL-3 | 6 ± 1 | 16 ± 5 | 10 ± 3 | 22 ± 11 | 2 ± 1 | 6 ± 3 | |||||
| SCF + IL-3 + EPO | 9 ± 4 | 20 ± 8 | 11 ± 6 | 15 ± 9 | 1 ± 2 | 23 ± 8 | |||||
. | Mean no. colonies, ± SD . | . | . | . | . | . | |||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Stimulus . | Blast . | Granulocyte . | Granulocyte-macrophage . | Macrophage . | Eosinophil . | Megakaryocyte . | |||||
| Wild-type, 2 wk; n = 3 | |||||||||||
| GM-CSF | 0 | 15 ± 4 | 7 ± 4 | 17 ± 4 | 2 ± 1 | 0 | |||||
| M-CSF | 0 | 0.7 ± 0.6 | 3 ± 1 | 50 ± 17* | 0 | 0 | |||||
| IL-3 | 8 ± 2 | 12 ± 4 | 8 ± 3 | 11 ± 12 | 2 ± 2 | 5 ± 2 | |||||
| SCF + IL-3 + EPO | 8 ± 3 | 18 ± 6 | 12 ± 11 | 11 ± 12 | 2 ± 2 | 24 ± 5 | |||||
| MYC17, 2 wk; n = 3 | |||||||||||
| GM-CSF | 0 | 10 ± 2 | 4 ± 0 | 19 ± 8 | 2 ± 1 | 0 | |||||
| M-CSF | 0 | 0 | 2 ± 2 | 158 ± 48* | 0 | 0 | |||||
| IL-3 | 5 ± 2 | 9 ± 3 | 3 ± 3 | 13 ± 6 | 3 ± 3 | 6 ± 2 | |||||
| SCF + IL-3 + EPO | 8 ± 3 | 7 ± 3 | 3 ± 2 | 13 ± 15 | 0.7 ± 1.2 | 11 ± 4 | |||||
| Wild-type, 8 wk; n = 4 | |||||||||||
| GM-CSF | 0 | 16 ± 5 | 5 ± 2 | 31 ± 3 | 4 ± 2 | 0 | |||||
| M-CSF | 0 | 3 ± 1 | 6 ± 4 | 49 ± 8* | 0 | 0 | |||||
| IL-3 | 7 ± 4 | 19 ± 6 | 11 ± 3 | 15 ± 3 | 2 ± 1 | 7 ± 4 | |||||
| SCF + IL-3 + EPO | 11 ± 6 | 21 ± 2 | 13 ± 3 | 17 ± 3 | 2 ± 1 | 28 ± 4 | |||||
| MYC10, 8 wk; n = 4 | |||||||||||
| GM-CSF | 0 | 19 ± 6 | 5 ± 2 | 29 ± 15 | 3 ± 3 | 0 | |||||
| M-CSF | 0 | 3 ± 1 | 2 ± 1 | 109 ± 32* | 0 | 0 | |||||
| IL-3 | 6 ± 1 | 16 ± 5 | 10 ± 3 | 22 ± 11 | 2 ± 1 | 6 ± 3 | |||||
| SCF + IL-3 + EPO | 9 ± 4 | 20 ± 8 | 11 ± 6 | 15 ± 9 | 1 ± 2 | 23 ± 8 | |||||
Bone marrow cells from healthy young mice of the age and genotype indicated were cultured in agar and stimulated with cytokines (GM-CSF, G-CSF, M-CSF, IL-3, EPO, SCF, G-CSF + SCF, TPO, FIt3L + LIF, SCF + IL-3 + EPO, IFNγ, IL-6) for 7 days. The resultant colonies were stained, examined for composition, and counted. The results with 4 different stimuli are shown as examples.
Significant difference from wild-type controls was that the frequency of M-CSF-dependent macrophage colonies was increased for both MYC17 and MYC10 mice.
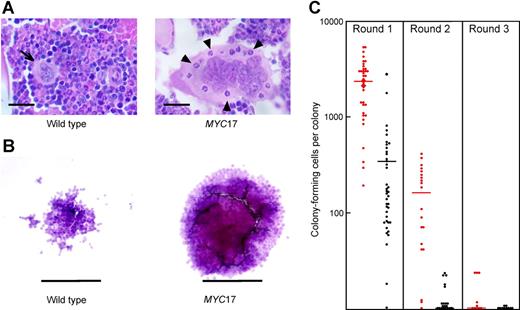
Titration experiments established that the quantitative responsiveness to M-CSF of MYC-expressing progenitor cells, including those capable of producing large colonies, was comparable to that of normal M-CSF–dependent progenitors (not shown). Recloning experiments were then undertaken to determine how many cells in the abnormally large colonies retained clonogenic potential (Figure 5C). Colonies grown in M-CSF were resuspended and recultured with M-CSF in semisolid medium. As expected, primary colonies from wild-type mice contained very few clonogenic cells: on average, each produced only 300 small colonies or clusters, and when recloning was repeated sequentially for a second and third cycle almost no colonies developed. In marked contrast, giant colonies from MYC10 mice contained an average of 2000 cells able to form compact secondary colonies, although these were smaller than the primary colonies. In subsequent rounds of recloning, however, there was an abrupt fall to an average of 150 clonogenic cells per colony in the second cycle, and only 10 per colony in the third cycle. These data imply that the MYC10 progenitor cells forming giant colonies lacked capacity for full self-renewal, but did have a significantly extended capacity for self-generative divisions.
VavP-MYC transgene expression has multiple effects on thymocytes. (A) Proliferation is increased. Sorted thymocyte subpopulations from healthy young mice were permeabilized, stained for DNA content with propidium iodide (PI), and analyzed by flow cytometry. Representative cell-cycle profiles for CD4+ CD8– T cells show an increased proportion of cells in S phase in VavP-MYC transgenic mice, and the cell-cycle distributions of all the thymocyte subpopulations analyzed is shown in Table 4. (B) Cell size is increased. Forward light-scatter analysis of gated thymocyte subpopulations from healthy young mice (2-week-old MYC17 and 6-week-old MYC10, MYC10hom, and wild-type mice). Representative examples from 3 mice or more examined are shown; wild-type thymocyte size was comparable at 2 and 6 weeks. Gray filled indicates wild-type; black dashed line, MYC10; black solid line, MYC10hom; and gray solid line, MYC17. (C) Sensitivity to spontaneous apoptosis is increased. Thymocytes from healthy young mice (2- and 6-week-old wild-type, 2-week-old MYC17, and 6-week-old MYC10 and MYC10hom mice) were cultured in vitro in simple medium. The percentage of gated thymocyte subpopulations remaining viable (not stained with propidium iodide) after 1 to 3 days was determined by flow cytometry (assays were performed on 2 or more independent cultures from each of 2 or more mice of each genotype; mean percentages ± SEM are for replicate mice).
VavP-MYC transgene expression has multiple effects on thymocytes. (A) Proliferation is increased. Sorted thymocyte subpopulations from healthy young mice were permeabilized, stained for DNA content with propidium iodide (PI), and analyzed by flow cytometry. Representative cell-cycle profiles for CD4+ CD8– T cells show an increased proportion of cells in S phase in VavP-MYC transgenic mice, and the cell-cycle distributions of all the thymocyte subpopulations analyzed is shown in Table 4. (B) Cell size is increased. Forward light-scatter analysis of gated thymocyte subpopulations from healthy young mice (2-week-old MYC17 and 6-week-old MYC10, MYC10hom, and wild-type mice). Representative examples from 3 mice or more examined are shown; wild-type thymocyte size was comparable at 2 and 6 weeks. Gray filled indicates wild-type; black dashed line, MYC10; black solid line, MYC10hom; and gray solid line, MYC17. (C) Sensitivity to spontaneous apoptosis is increased. Thymocytes from healthy young mice (2- and 6-week-old wild-type, 2-week-old MYC17, and 6-week-old MYC10 and MYC10hom mice) were cultured in vitro in simple medium. The percentage of gated thymocyte subpopulations remaining viable (not stained with propidium iodide) after 1 to 3 days was determined by flow cytometry (assays were performed on 2 or more independent cultures from each of 2 or more mice of each genotype; mean percentages ± SEM are for replicate mice).
MYC expression increases thymocyte proliferation, size, and sensitivity to apoptosis
As efficient T-cell lymphomagenesis appeared to require a threshold level of transgenic MYC expression, we surveyed the impact of the various levels of MYC expression on thymocytes from healthy young mice.
Cells from MYC17 mice were analyzed at approximately 2 weeks, an age when they reliably lack tumor cells, as judged by flow cytometry, histology, or transplantation (not shown). As expected from the susceptibility of this line to early T lymphomas,25 the T cells in young MYC17 mice displayed multiple changes. Cell-cycle analysis showed that more CD4+ CD8+ pre-T cells and especially CD4+ CD8– thymocytes were in S phase than in control wild-type littermates (Figure 4A; Table 4). (The increased cycling was not as marked for CD4– CD8+ thymocytes, probably because a striking 40% of these cells were already in S phase in 2-week-old control mice.) Another notable difference was that pre-T, CD4+ CD8–, and CD4-CD8+ thymocytes from MYC17 mice were substantially larger (Figure 4B; Smith et al25 ), and much more susceptible to spontaneous apoptosis when cultured without cytokine support (Figure 4C).
Increased thymocyte proliferation in healthy young VavP-MYC mice
. | 2 weeks old . | . | . | . | 6 weeks old . | . | . | . | . | . | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| . | Wild-type; n = 4 . | . | MYC17; n = 2 . | . | Wild-type; n = 5 . | . | MYC10; n = 5 . | . | MYC10hom; n = 5 . | . | ||||||||
. | S phase . | G2/M phase . | S phase . | G2/M phase . | S phase . | G2/M phase . | S phase . | G2/M phase . | S phase . | G2/M phase . | ||||||||
| CD4+ CD8+* | 11.6 ± 0.7 | 0.8 ± 0.1 | 15.9 ± 0.6 | 1.4 ± 0.2 | 7.6 ± 1 | 1.6 ± 0.3 | 9.2 ± 0.3 | 1.7 ± 0.3 | 10.3 ± 0.6 | 1.9 ± 0.4 | ||||||||
| P | — | — | .01 | .01 | — | — | .01 | > .05 | .001 | > .05 | ||||||||
| CD4+ CD8-* | 9.7 ± 0.4 | 0.8 ± 0.1 | 28.8 ± 0.8 | 1.3 ± 0.4 | 3.1 ± 0.9 | 1.1 ± 0.2 | 7.1 ± 0.2 | 1.3 ± 0.2 | 15.0 ± 0.5 | 1.5 ± 0.2 | ||||||||
| P | — | — | < .001 | > .05 | — | — | .001 | > .05 | < .001 | > .05 | ||||||||
| CD4- CD8+* | 40.1 ± 1.6 | 0.9 ± 0.3 | 37.6 ± 0.5 | 0.8 ± 0.1 | 14.7 ± 2.4 | 1.3 ± 0.3 | 21.9 ± 1.2 | 1.5 ± 0.3 | 26.7 ± 1.1 | 1.6 ± 0.4 | ||||||||
| P | — | — | > .05 | > .05 | — | — | .01 | > .05 | < .001 | > .05 | ||||||||
. | 2 weeks old . | . | . | . | 6 weeks old . | . | . | . | . | . | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| . | Wild-type; n = 4 . | . | MYC17; n = 2 . | . | Wild-type; n = 5 . | . | MYC10; n = 5 . | . | MYC10hom; n = 5 . | . | ||||||||
. | S phase . | G2/M phase . | S phase . | G2/M phase . | S phase . | G2/M phase . | S phase . | G2/M phase . | S phase . | G2/M phase . | ||||||||
| CD4+ CD8+* | 11.6 ± 0.7 | 0.8 ± 0.1 | 15.9 ± 0.6 | 1.4 ± 0.2 | 7.6 ± 1 | 1.6 ± 0.3 | 9.2 ± 0.3 | 1.7 ± 0.3 | 10.3 ± 0.6 | 1.9 ± 0.4 | ||||||||
| P | — | — | .01 | .01 | — | — | .01 | > .05 | .001 | > .05 | ||||||||
| CD4+ CD8-* | 9.7 ± 0.4 | 0.8 ± 0.1 | 28.8 ± 0.8 | 1.3 ± 0.4 | 3.1 ± 0.9 | 1.1 ± 0.2 | 7.1 ± 0.2 | 1.3 ± 0.2 | 15.0 ± 0.5 | 1.5 ± 0.2 | ||||||||
| P | — | — | < .001 | > .05 | — | — | .001 | > .05 | < .001 | > .05 | ||||||||
| CD4- CD8+* | 40.1 ± 1.6 | 0.9 ± 0.3 | 37.6 ± 0.5 | 0.8 ± 0.1 | 14.7 ± 2.4 | 1.3 ± 0.3 | 21.9 ± 1.2 | 1.5 ± 0.3 | 26.7 ± 1.1 | 1.6 ± 0.4 | ||||||||
| P | — | — | > .05 | > .05 | — | — | .01 | > .05 | < .001 | > .05 | ||||||||
P indicates P value calculated by t test; —, not applicable.
Values in these rows are expressed as % ± SEM.
Thymocytes from MYC10 and MYC10hom mice were similarly compared with those from wild-type littermate control mice at the oldest age (approximately 6 weeks) reliably lacking detectable tumor cells. The impact of the transgene was generally less than in MYC17 mice but nonetheless clear, even in MYC10 mice, which rarely develop T-cell tumors. The number of pre-T, CD4+ CD8–, and CD4– CD8+ thymocytes in S phase increased, and the increase was more pronounced in MYC10hom mice (Figure 4A; Table 4). The effect on CD4+ CD8– cells was most notable, with MYC10 mice having approximately twice as many, and MYC10hom mice approximately 5 times as many of these cells in S phase as controls. The size of thymocytes within all 3 subpopulations was also increased (Figure 4B), and again the impact was greater for MYC10hom mice. Finally, the 3 thymocyte subpopulations analyzed were all more sensitive to spontaneous apoptosis (Figure 4C), pre-T thymocytes being the least and CD4– CD8+ thymocytes the most sensitive, particularly for MYC10hom cells.
Abnormal hematopoiesis in VavP-MYC mice. (A) An abnormally large megakaryocyte in the bone marrow of a 4-week-old MYC17 mouse (right panel) containing internalized granulocytes (some indicated by arrowheads) and for comparison, a normal megakaryocyte (left panel; indicated by arrow) in the bone marrow of a wild-type littermate. H&E-stained sections; scale bar equals 20 μm. (B) Abnormally large macrophage colonies developed in M-CSF–stimulated cultures of MYC17 (right panel) and MYC10 (not shown) bone marrow cells when compared with cultures of wild-type cells (left panel); scale bar equals 125 μm. (C) Primary M-CSF–stimulated macrophage colonies from 8-week-old MYC17 (red) and wild-type (B6) mice were recultured in M-CSF (Round 1) and the colonies that developed were counted. Recloning was performed for 2 more successive rounds using colonies from the first and second rounds. Bars indicate the mean.
Abnormal hematopoiesis in VavP-MYC mice. (A) An abnormally large megakaryocyte in the bone marrow of a 4-week-old MYC17 mouse (right panel) containing internalized granulocytes (some indicated by arrowheads) and for comparison, a normal megakaryocyte (left panel; indicated by arrow) in the bone marrow of a wild-type littermate. H&E-stained sections; scale bar equals 20 μm. (B) Abnormally large macrophage colonies developed in M-CSF–stimulated cultures of MYC17 (right panel) and MYC10 (not shown) bone marrow cells when compared with cultures of wild-type cells (left panel); scale bar equals 125 μm. (C) Primary M-CSF–stimulated macrophage colonies from 8-week-old MYC17 (red) and wild-type (B6) mice were recultured in M-CSF (Round 1) and the colonies that developed were counted. Recloning was performed for 2 more successive rounds using colonies from the first and second rounds. Bars indicate the mean.
Overall, these data indicate that constitutive expression of MYC in T cells increases cell cycling, size, and sensitivity to apoptosis even when expression is below a critical threshold required for efficient progression to malignancy, and that the impact of MYC is proportional to its level of expression.
Overexpression of BCL2 facilitates T-cell lymphomagenesis in MYC10 mice
BCL2 synergizes with Myc in transforming B-lymphoid cells.18 We sought to ascertain whether this was also true of T cells by crossing VavP-MYC10 and VavP-BCL2 mice (strain 6924,27 ). VavP-BCL2 mice accumulate excessive numbers of both B- and T-lymphoid cells, including IgH class-switched B cells, and develop germinal center hyperplasia; a large number (37% to 50%, depending on the line) succumb to follicular lymphoma by 18 months of age.24,27 Significantly, bitransgenic VavP-MYC10-BCL2 mice had a much shorter lifespan (median, 13 weeks; n = 28) than mice expressing either transgene alone. Furthermore, BCL2 dramatically increased the rate of T-cell lymphomagenesis: of the sick mice autopsied, 47% (8 of 17) developed T-cell lymphomas and both of those tested were transplantable. The phenotype of these T-cell tumors varied: in 4 tumors, most cells were CD4+ CD8+, 1 tumor was CD4+ CD8–, 2 tumors had a mixture of these cell types, and another was CD4– CD8–. Tumor-cell morphology varied in appearance from classic lymphoblastic to a more immunoblastic type (not shown), as observed for MYC17 T-cell lymphomas.25 In all cases, the sick mice were highly leukemic and large tumor deposits were routinely seen throughout the hematopoietic system and in the liver, lung, and kidney.
In the remaining sick MYC10-BCL2 mice (9 of 17), no T-cell tumors were detected by flow cytometry or histologic analysis. Instead, their lymphoid organs were enlarged by a very large excess of small IgH class-switched B cells (mostly CD19+ IgM– IgD+ κ light-chain+). These cells did not appear to be neoplastic and the 3 cases tested were not transplantable. These class-switched B cells presumably reflect an exaggeration of the germinal center hyperplasia seen in VavP-BCL2 mice.27 An expansion of this population may be a constant feature of MYC10-BCL2 mice, since it was also noted in some mice that developed T-cell tumors.
Discussion
Our comparison of 4 lines of transgenic mice expressing different levels of MYC in a range of hematopoietic cell types has revealed notable preneoplastic changes and marked effects of MYC levels on tumor phenotype.
Preneoplastic changes
The line with the highest expression, MYC17, had a dramatic preneoplastic phenotype, including a 3-fold enlarged spleen (with excess B-lymphoid and myeloid cells), a transient 10-fold increase in circulating leukocytes (both B-lymphoid and myeloid), and a substantial increase in bone marrow monocytes.25 In contrast, the lower expression levels in MYC10 mice and MYC10hom mice only modestly perturbed hematopoietic homeostasis (Table 1; not shown).
Megakaryocyte and platelet abnormalities were a notable feature of healthy young VavP-MYC mice, although the frequency and proliferative capacity of megakaryocyte progenitors in response to cytokines including SCF, IL-3, EPO, and TPO was normal (Table 3). Megakaryocytes were more abundant and larger (Figure 5A; Table 2) and their nuclei were abnormally large and complex, which suggests that MYC had promoted extra rounds of endoreplication, resulting in even greater polyploidy than is normal for this cell type.31 Myc-driven endoreplication has previously been reported for mammalian keratinocytes32 and some Drosophila cell types.33 Since platelets in MYC17 and MYC10hom mice were larger and less numerous than in normal littermates, MYC may also have retarded megakaryocyte maturation. Inhibition of differentiation by deregulated Myc has also been observed in other cell lineages.4,16 Another prominent feature of bone marrow megakaryocytes was that the cytoplasm frequently contained multiple granulocytes, presumably due to increased emperipolesis. In this poorly understood process, megakaryocytes internalize other cells within demarcation membranes, a network that is continuous with extracellular fluid (see Bobik and Dabrowski,34 McGarry et al,35 and references therein). The “engulfed” cells therefore remain intact and able to exit. As in platelet α-granule disorders and myelofibrosis,35-37 the reduced platelet production in VavP-MYC mice may result in the abnormal accumulation of granulocyte-attracting platelet proteins within megakaryocytes and augmented emperipolesis.
The bone marrow of young healthy VavP-MYC mice contained normal numbers of immature progenitor cells, as judged by the frequency of blast colonies that developed in semisolid agar cultures (Table 3). Committed megakaryocyte and myeloid progenitors were also normal in frequency and proliferative capacity, except for M-CSF–responsive macrophage progenitors, which were highly abnormal, generating large compact “cannonball” colonies (Figure 5B; Table 3). Comparable colonies were not seen with the other 2 cytokines able to stimulate macrophage colony formation (GM-CSF and IL-3; Table 3), suggesting that MYC may have led to the expansion of a specific subset of macrophage progenitors. The abnormal macrophage progenitors were not present in spleen, but were already present at birth in the neonatal liver of at least the MYC17 mice (not shown). Recloning experiments (Figure 5C) indicated that they were not capable of full self-generation, although they did have greatly extended capacity for generating macrophage progenitors in response to M-CSF (but not GM-CSF). This feature was particularly marked in the case of the high MYC-expressing (MYC17) cells, which may account for the large excess of monocyte/macrophages found in the bone marrow of healthy young MYC17 mice.25
Tumor phenotype varies with MYC level
MYC17 mice all succumb to T-cell tumors,25 most being mature CD4+ CD8– lymphomas, which are rare in mouse models. The very rapid development of these tumors (Figure 2) masks the tumorigenic potential conferred by MYC on other hematopoietic cell types in these mice, since several other tumor classes, including pro–B-cell, progenitor-cell, and macrophage tumors are obtained in the absence of mature lymphocytes in the Rag1-null background.25
In the lines with much lower levels of VavP-MYC transgene expression (MYC10 and MYC12), morbidity is primarily due to slow-developing disseminated histiocytic monocyte/macrophage tumors (Figures 2, 3A-B). This suggests that the macrophage lineage is particularly susceptible to MYC-induced transformation. It is tempting to speculate that these tumors arise from the abnormal macrophage progenitors seen in preneoplastic mice. The VavP-MYC macrophage tumors resemble human histiocytic sarcoma (which, like the VavP-MYC tumors, can present in the skin) and its disseminated form, malignant histiocytosis.38 Since tumors develop in MYC10 and MYC12 mice within an experimentally acceptable latent period and the penetrance is high, these lines may be valuable models for these rare but generally incurable human neoplasms. MYC involvement in these in these neoplasms has not been described, and our data indicate that such an analysis may be worthwhile.
The 2-fold increase in MYC obtained by generating mice homozygous for the MYC10 locus led to a remarkable difference in pathogenic outcome. The median survival was 13 weeks rather than 41 weeks (Figure 2) and the MYC10hom mice succumbed to lymphoblastic T-cell lymphomas (Figure 3C-D) rather than histiocytic monocyte/macrophage tumors. Curiously, although the preneoplastic impact of MYC on T cells (proliferation, growth, and apoptosis) in MYC10hom mice was greatest in the CD4– CD8+ and, in particular, the CD4+ CD8– thymocyte population (Figure 4; Table 4), the tumors were almost all CD4+ CD8+ pre–T-cell lymphomas, as is typical in other MYC transgenic lines that are prone to T-cell tumors.21,39 In contrast, most high-expressing MYC17 mice succumb to mature CD4+ CD8– tumors,25 suggesting that malignant transformation of this cell type requires higher levels of MYC.
Strikingly, crossing a BCL2 transgene into MYC10 mice greatly enhanced their susceptibility to T-cell lymphomas (50% versus 7%). This suggests that the malignant transformation of MYC-driven T cells depends, at least in part, on accumulating antiapoptotic mutation(s) and that, in conjunction with an antiapoptotic mutation, even the modest proliferative and cell-growth effects of low-level MYC expression (Figure 4; Table 4) can drive efficient T-cell lymphomagenesis. The differing levels of MYC in VavP-MYC mice all induce apoptosis in cultured T cells (Figure 4C), and the mechanism is likely to be similar. However, the observations that MYC10 mice only efficiently developed T-cell tumors when an antiapoptotic mutation was enforced and that all MYC10hom and MYC17 mice rapidly developed T-cell lymphomas suggest that the odds of spontaneously developing cooperating antiapoptotic mutations are proportional to the level of MYC.
The mechanism of Myc-induced apoptosis is still poorly understood,6 but involves resetting the life/death rheostat regulated by the Bcl2 family.20,40 Myc up-regulates the tumor suppressor p19Arf, which in turn leads to increased p5341 and transcriptional activation of 2 proapoptotic Bcl2 relatives, the BH3-only proteins Puma and Noxa.42,43 Myc also elevates the p53-independent expression of another BH3-only protein, Bim,20 and, in parallel, the levels of antiapoptotic Bcl2 and BclxL proteins diminish.20,44 Thus, Myc increases the ratio of proapoptotic to antiapoptotic proteins, lowering the threshold for induction of apoptosis. To progress to malignancy, cells with constitutive MYC expression must overcome their predilection to apoptosis, and this can be achieved experimentally in Eμ-Myc mice either by imposing expression of Bcl218 or BclxL,45 or by loss of p53,46 p19Arf,19,46 Bim,20 or Puma.47 Pertinently, a high proportion of spontaneous Eμ-Myc lymphomas19 and Burkitt lymphomas48 exhibit mutations inactivating the p19Arf/Mdm2/p53 axis. Furthermore, 2 MYC point mutants isolated from Burkitt lymphomas are defective at inducing Bim (while still retaining ability to stimulate proliferation and p53 activation),49 and loss of BIM is a common feature of human mantle-cell lymphoma.50
Implications
Our studies have demonstrated a remarkable difference in pathogenic phenotype that is dependent on the level of MYC expression. More specifically, the results suggest that macrophages are susceptible to malignant transformation at even low levels of MYC, and that there is a sharp threshold level of MYC that must be surpassed for efficient T-cell lymphomagenesis.
Once this threshold is surpassed, as in MYC10hom mice, the acquisition of T-cell–relevant synergistic mutations appears to be greatly facilitated. This may simply be the consequence of the increased number of cell divisions. Alternatively, higher levels of MYC expression could have more direct effects, such as altered expression of pathologic target genes due to occupation of low affinity and aberrant promoter binding sites.1,2 Higher levels of MYC may also promote genomic instability.5 Fine-tuning the level of deregulated MYC expression may be an important mechanism in the progression of human cancers. For example, mutations in MYC that are selected during the progression of Burkitt lymphoma not only abrogate the induction of Bim49 but also increase the stability of MYC.11-13
Since the tumor phenotype of MYC10 mice is so sensitive to MYC levels, this line should be a uniquely responsive indicator strain to examine the lymphomagenic impact of mutations in genes that may influence MYC stability (for example, GSK3B, PIN1, PP2A, FBW7, and SKP2, which regulate MYC phosphorylation and subsequent degradation11,12 ) or activity (for example, the MAD family of antagonists51 ). Furthermore, in MYC10hom mice, agents that decrease the level of MYC even 2-fold should dramatically reduce the incidence of T-cell lymphomas. Hence, these mice may prove to be a valuable strain in which to test therapeutics targeted at pathways regulating MYC stability or activity.22
In summary, these VavP-MYC transgenic mice represent valuable new tools to study the mechanisms of leukemogenesis, to search for and test putative cooperating mutations, and to investigate the impact of chemotherapeutics on tumors of the T lymphoid and monocyte/macrophage lineages. They also provide the means to compare MYC targets at different expression levels and in multiple hematopoietic cell types.
Prepublished online as Blood First Edition Paper, March 14, 2006; DOI 10.1182/blood-2006-01-0172.
Supported by the National Health and Medical Research Council (NHMRC; Canberra, program grant 257502), the National Cancer Institute (grant CA43540), and the Leukemia and Lymphoma Society (Specialized Center of Research grant 7015).
D.P.S. and D.M. designed and performed research, analyzed data and wrote the paper; M.L.B. designed and performed research and analyzed data; A.W.H. designed research and analyzed data; and S.C. designed research, analyzed data, and wrote the paper.
An Inside Blood analysis of this article appears at the front of this issue.
The publication costs of this article were defrayed in part by page charge payment. Therefore, and solely to indicate this fact, this article is hereby marked “advertisement” in accordance with 18 U.S.C. section 1734.
We thank J. Adams for helpful discussions, A. Strasser for his gift of antibodies, and A. Wiegmans for technical assistance.





This feature is available to Subscribers Only
Sign In or Create an Account Close Modal