Casein kinase 2 (CK2) is a ubiquitous cellular serine-threonine kinase that regulates relevant biologic processes, many of which are dysregulated in malignant plasma cells. Here we investigated its role in multiple myeloma (MM). Analysis of MM cell lines and highly purified malignant plasma cells in patients with MM revealed higher protein and CK2 activity levels than in controls (normal in vitro-generated polyclonal plasma cells and B lymphocytes). The inhibition of CK2 with specific synthetic compounds or by means of RNA interference caused a cytotoxic effect on MM plasma cells that could not be overcome by IL-6 or IGF-I and that was associated with the activation of extrinsic and intrinsic caspase cascades. CK2 blockage lowered the sensitivity threshold of MM plasma cells to the cytotoxic effect of melphalan. CK2 inhibition also resulted in impaired IL-6-dependent STAT3 activation and in decreased basal and TNF-α-dependent IκBα degradation and NF-κB-driven transcription. Our data show that CK2 was involved in the pathophysiology of MM, suggesting that it might play a crucial role in controlling survival and sensitivity to chemotherapeutics of malignant plasma cells.
Introduction
Multiple myeloma (MM) is one of the most frequent hematologic malignancies arising from end-stage B lymphocytes.1,2 The growth of MM cells strictly depends on the surrounding bone marrow (BM) microenvironment1 through a number of paracrine and autocrine intercellular loops. For instance, several soluble mediators—such as IL-6, TNF-α, IL-1β, IGF-I, TGF-β, and VEGF— released by the BM stroma or by malignant plasma cells regulate plasma cell survival and proliferation, adhesiveness, migration, neoangiogenesis, and other features in the BM milieu.3 Several studies have identified genetic and biochemical abnormalities and play a relevant role in MM pathogenesis, but the mechanisms of MM cell growth are still largely unknown.4 In particular, a key point is the onset of resistance to drug-induced apoptosis underlying the poor outcome for patients with MM, whose molecular determinants are still elusive.5
CK2 (formerly known as casein-kinase II) is a constitutively active serine-threonine protein kinase with a tetrameric structure consisting of 2 catalytic (α or α′ or both) and 2 regulatory (β) subunits. CK2 may counteract apoptosis and sustain the cell cycle,6-8 and its expression and activity are frequently enhanced in tumors.9-11 Increases in CK2 level and activity in cancer cells do not simply reflect the proliferative status of transformed cells because its expression pattern in tumor samples does not parallel that of the proliferation marker Ki67 and it is found to be highly expressed in nonproliferative dysplastic areas.12 CK2 controls molecular pathways that are central for B-cell biology and MM cell growth, such as NF-κB signaling. CK2 can phosphorylate IκBα in the carboxy-terminal PEST domain, leading to changes in its stability,13,14 and in the amino-terminal serine residues 32 and 36, which are generally targeted by the kinase IKKβ.15 Moreover, CK2-dependent phosphorylation after TNF-α and IL-1 stimulation is necessary to lend full transcriptional potential to NF-κB RelA/p65.16,17
Several stimuli, such as IL-6, TNF-α, and IGF-I, may sustain MM cell growth, survival, and adhesion, in part through NF-κB activation.18,19 Other stimuli, such as IL-1, may also mediate bone disease and plasma cell proliferation by activating NF-κB.20 Whether CK2 takes part in these processes by modulating NF-κB in MM is still unknown. Interestingly, CK2 also positively regulates c-myc protein stability.21 C-myc is crucial for cell proliferation, is frequently rearranged in B-cell malignancies and MM,22 and functionally interacts with CK2 in vivo.23 Because NF-κB drives the transcription of c-myc,24,25 NF-κB and CK2 may cross-talk in regulating this common target molecule, but whether this cooperation occurs in human cancer is unknown.
Moreover, IL-6 and IGF-I stimulate other important signaling pathways for MM cell growth, such as the JAK-STAT, the PI3K-AKT, and the MAPK cascades. Although CK2 has been evoked to regulate cell survival upon growth factors,26,27 its role downstream from these signaling pathways in MM and other B-cell malignancies is largely unknown. In addition, though CK2 activity has been shown to be elevated in solid tumors,9,10 no definitive studies are available regarding whether this kinase plays a role in the pathogenesis of B-cell malignancies and MM. In this work, we investigated CK2 levels and kinase activity in MM cells and the cellular and molecular consequences of hampering its function in these cells. In addition, we analyzed the effects of CK2 in regulating IL-6 and NF-κB signaling in MM cells. Our data suggest that increased CK2 function is instrumental to MM cell survival and protection from apoptosis, and they show that this kinase controls the IL-6/STAT3 and NF-κB pathways in these cells.
Materials and methods
Patients and cell cultures
Informed consent was obtained from patients in accordance with the Declaration of Helsinki. Sources of samples are listed in Table 1. Mononuclear cells from PB and BM were separated as in previous studies.28 CD138+ MM cells were purified by immunomagnetic sorting (Miltenyi Biotech, Bergish Gladbach, Germany) according to the manufacturer's protocol. MM cell lines OPM2, U266, and RPMI 8226 were purchased from the American Type Culture Collection (Rockville, MD); all cell lines were maintained in RPMI 1640 medium supplemented with 10% FCS, L-glutamine, penicillin, and streptomycin (Gibco Laboratories, Grand Island, NY) under controlled atmosphere at 37°C in the presence of 5% CO2. HEK293 cells were grown in DMEM with 10% FCS and other supplements. Plasma cells were generated in vitro, as previously described.29 B cells were enriched from PBMCs, as previously reported,30 and by positive selection with anti-CD19 MACS microbeads and were plated at 1.5 × 105/mL in the presence of 3.75 × 104/mL mitomycin C (Sigma-Aldrich, Milan, Italy)-treated CD40L-transfectant L-cells (a generous gift from Dr John Gordon, MRC Centre for Immune Regulation, Birmingham, United Kingdom) with various combinations of IL-2 (20 U/mL), IL-4 (50 ng/mL), IL-10 (50 ng/mL), and IL-12 (2 ng/mL) (PeproTech, Rocky Hill, NJ). On day 4, B cells were reseeded at 3 × 105/mL without CD40L transfectants and with IL-2 (20 U/mL), IL-10 (50 ng/mL), IL-12 (2 ng/mL), and IL-6 (5 ng/mL). On day 6, cells were double stained with FITC-conjugated anti-CD20 and PE-conjugated anti-CD38 (Becton Dickinson, San Jose, CA), and CD20-/CD38++ cells were sorted with a BD FACSAria (Becton Dickinson).
Clinical features of patients with MM
Patient no. . | Age at diagnosis, y/sex . | Paraprotein type . | Plasma cells in BM at diagnosis, % . | Stage (other organs involved) . | Source of samples . |
|---|---|---|---|---|---|
| 1 | 62/F | IgG λ | 85 | IIIA (–) | PB |
| 2 | 62/F | IgG λ | 80 | IIIA (SNC, lung) | BM |
| 3 | 65/F | IgG λ | 40 | IIIA (osteolysis) | BM |
| 4 | 56/M | IgG κ | 99 | IIIA (–) | BM |
| 5 | 70/M | IgG λ | 3* | IIIA (pleural effusion, testis, osteolysis) | PE |
| 6 | 57/M | IgA κ | 25 | IIIA (pleural effusion, osteolysis) | PE |
| 7 | 68/M | IgG λ | 30 | IIIB (osteolysis, CRF, anemia) | BM |
Patient no. . | Age at diagnosis, y/sex . | Paraprotein type . | Plasma cells in BM at diagnosis, % . | Stage (other organs involved) . | Source of samples . |
|---|---|---|---|---|---|
| 1 | 62/F | IgG λ | 85 | IIIA (–) | PB |
| 2 | 62/F | IgG λ | 80 | IIIA (SNC, lung) | BM |
| 3 | 65/F | IgG λ | 40 | IIIA (osteolysis) | BM |
| 4 | 56/M | IgG κ | 99 | IIIA (–) | BM |
| 5 | 70/M | IgG λ | 3* | IIIA (pleural effusion, testis, osteolysis) | PE |
| 6 | 57/M | IgA κ | 25 | IIIA (pleural effusion, osteolysis) | PE |
| 7 | 68/M | IgG λ | 30 | IIIB (osteolysis, CRF, anemia) | BM |
Stage was determined according to the Durie-Salmon staging system
PE indicates pleural effusion; CRF, chronic renal failure
Patient with an initial presentation of extramedullary myeloma (testis)
Chemicals and cytokines
TBB (4,5,6,7 tetrabrome benzotriazole) was synthesized as in previous studies,31 and IQA ([5-oxo-5,6-dihydroindole (1,2-a) quinazolin-7-yl]-acetic acid), formerly termed CGP029482,32 was kindly provided by Dr J. Schoepfer (Novartis, Basel, Switzerland). The TBB-derivative K27 (2-amino-4,5,6,7-tetrabromo-1H-benzimidazole), also termed 2A,33 was synthesized and kindly provided by Dr Z. Kazimierczuk (Warsaw, Poland). The remarkable specificity of TBB and IQA toward only CK2 out of a panel of more than 30 protein kinases (with the partial exception of DYRK1A) has been assessed elsewhere.32,34 K27/2a is closely related to DMAT/2C,33,35 whose specificity is similar to that of TBB. All these compounds are cell permeable.7,32,33 Melphalan, MG132 (carbobenzoxyl-leucinyl-leucinyl-leucinal), and the cytokines TNF-α, IL-6, and IGF-I were purchased from Sigma-Aldrich (Milan, Italy).
CK2 activity in cell lysates
Evaluation of growth and apoptosis
MM cell growth was monitored using the MTT (3-[4,5-dimethylthiazole-2-yl]-2,5-diphenyltetrazolium bromide)-based assay, according to the manufacturer's protocol (Roche, Monza, Italy) and as described elsewhere.7 Apoptosis was assessed by annexin V/propidium iodide staining (BD PharMingen, San Francisco, CA) according to the manufacturer's instructions using a FACSCalibur cytofluorometer with the CellQuest analytic software (Becton Dickinson).
Western blot and immunoprecipitation
Whole cell extracts were prepared by lysis with 20 mM Tris, 150 mM NaCl, 2 mM EDTA, 2 mM EGTA, 0.5% Triton X-100 supplemented with complete protease inhibitor cocktail (Complete Mini; Roche, Basel, Switzerland), 1 mM dithiothreitol (DTT; Amersham Biosciences, Little Chalfont, Buckinghamshire, United Kingdom), 1 mM phenyl-methyl-sulfonyl fluoride (PMSF; Sigma-Aldrich), 10 μM sodium fluoride (Sigma-Aldrich), 1 μM okadaic acid, and 1 mM sodium orthovanadate (Calbiochem, San Diego, CA). Extraction of cytosolic fractions was carried out as described elsewhere.37 Immunoprecipitation was performed according to standard protocols.
Proteins were subjected to SDS-PAGE, transferred to nitrocellulose membranes, and immunoblotted with the following primary antibodies: CK2 α-subunit rabbit antiserum raised against the [376-391] region of human protein38 ; anti-PARP and anti-SMAC/DIABLO, anti-STAT3, phospho-Tyr702-STAT3, and phospho-Ser727-STAT3 (Cell Signaling, Beverly, MA); anti-caspase-3, anti-caspase-9, and anti-caspase-8 (Calbiochem-Merck Biosciences, Bad Soden, Germany); anti-cytochrome c (Becton Dickinson, Milan, Italy); anti-IκBα (C-15) and p105/50 (C-19) (Santa Cruz Biotechnology, Santa-Cruz, CA); anti-p65 (CT) (Upstate Biotechnology, Lake Placid, NY); anti-phospho-Ser32 and -Ser36; and IκBα (Ab-1; OP142; Calbiochem). Detection was performed by ECL chemiluminescence reaction according to the manufacturer's instructions (Amersham Biosciences). When indicated, densitometry was performed with ImageJ 1.34 S software (Apple, Cupertino, CA).
RNA interference, plasmids, transfection, and luciferase assay
For RNA interference, 25 to 35 × 106 U266 MM cells in log phase of growth were transfected by electroporation with 600 pmol siGlo-Red or 600 pmol siGlo-Red plus control siRNA pool or CK2α-specific siRNA pool (SmartPool; Dharmacon, Lafayette, CO) using a Bio-Rad GenePulser II electroporator (Bio-Rad, Hercules, CA) set at 960 μF and 250 V. Seventy-two to 96 hours after transfection, siGlo-positive cells were FACS sorted and stained with FITC-conjugated annexin V (BD PharMingen), apoptotic cells were scored, and mean fluorescence intensity was determined; moreover, cell lysates were prepared to assess CK2α and IκBα protein expression levels. In experiments with CK2α wild-type and mutant that bore a mutation of 2 residues (Val66 and Ile174), the respective expression vectors were kindly provided by Dr M. Salvi and Dr S. Sarno (Department of Biological Chemistry, University of Padova, Padova, Italy) and were transiently transfected along with a green fluorescent protein (pEGFP)-expressing vector (Clontech, Palo Alto, CA) in HEK293 cells using lipofectamine (Invitrogen, Carlsbad, CA) according to the manufacturer's instructions. For luciferase assay experiments, MM cell lines OPM2 and RPMI 8226 at 3 × 105 cells/well were transfected with an NF-κB luciferase reporter plasmid (Stratagene, La Jolla, CA) using the FuGENE transfection reagent (Roche Diagnostics, Almere, The Netherlands) according to the manufacturer's instructions and as described elsewhere.39 The Dual Luciferase Reporter System (Promega, Madison, WI) was used according to the supplier's protocol. Experiments were performed in triplicate or quadruplicate.
Confocal microscopy
MM cells seeded on polylysine-coated glass slides for 10 minutes were then fixed in 4% paraformaldehyde for 10 minutes at RT and permeabilized in PBS plus 0.1% Triton X-100 and sodium citrate (10 minutes at RT) before proceeding to blocking and staining. Specimens were mounted in Vectashield medium (Vector Laboratories, Peterborough, England) and analyzed using a dual-photon confocal microscope equipped with a 60×/1.4 numeric aperture (NA) or 100×/1.3 NA objective and fluorescence filters set for excitation at 488 nM and 595 nM (Bio-Rad, Milan, Italy), and images were acquired with Lasersharp 2000 software (Bio-Rad, Milan, Italy). Secondary conjugated antibodies Alexa Fluor 488 goat antirabbit (A-11008), Alexa Fluor 594 goat antirabbit (A-11012), and Alexa Fluor 594 donkey antigoat (A-11058) were purchased from Molecular Probes Europe (Leiden, The Netherlands).
Statistical analysis
Data were evaluated for their statistical significance with the 2-tail paired Student t test. Values were considered statistically significant at P values below .05.
Results
CK2 activity and expression in MM cells
We investigated CK2 activity and protein levels in MM cell lines (OPM2, U266, and RPMI 8226) and purified CD138+ plasma cells from MM patients (whose clinical features are summarized in Table 1). As controls, we used in vitro-generated normal polyclonal plasma cells/plasmablasts29 and purified normal CD19+ B lymphocytes from peripheral blood, spleen, or tonsil.
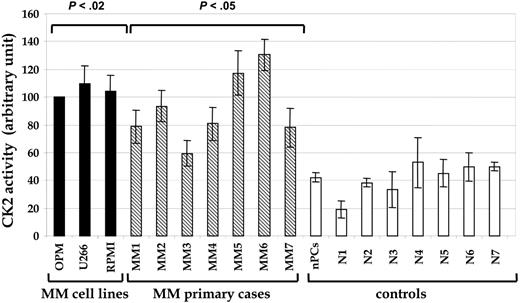
CK2 kinase activity in MM cells. Graph shows the levels of CK2-specific kinase activity in MM cell lines (▪), CD138+ purified plasma cells from 7 MM patients ( ), in vitro-generated normal plasma cells (nPCs), and purified CD19+ B lymphocytes from healthy donors (□). Samples N1 to N3 were from peripheral blood; samples N4 and N5 were from tonsil; samples N6 and N7 were from spleen. Data are mean ± SD.
), in vitro-generated normal plasma cells (nPCs), and purified CD19+ B lymphocytes from healthy donors (□). Samples N1 to N3 were from peripheral blood; samples N4 and N5 were from tonsil; samples N6 and N7 were from spleen. Data are mean ± SD.
CK2 kinase activity in MM cells. Graph shows the levels of CK2-specific kinase activity in MM cell lines (▪), CD138+ purified plasma cells from 7 MM patients ( ), in vitro-generated normal plasma cells (nPCs), and purified CD19+ B lymphocytes from healthy donors (□). Samples N1 to N3 were from peripheral blood; samples N4 and N5 were from tonsil; samples N6 and N7 were from spleen. Data are mean ± SD.
), in vitro-generated normal plasma cells (nPCs), and purified CD19+ B lymphocytes from healthy donors (□). Samples N1 to N3 were from peripheral blood; samples N4 and N5 were from tonsil; samples N6 and N7 were from spleen. Data are mean ± SD.
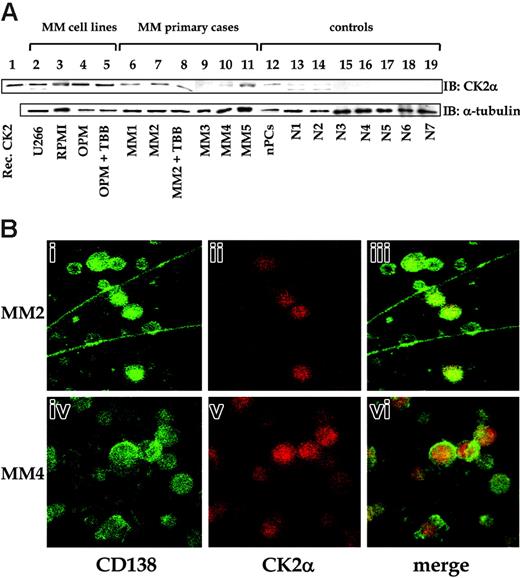
An in vitro kinase assay on whole cell extracts using a CK2-specific peptide40 as substrate revealed that CK2 activity was considerably higher in tumor cells than in normal plasma cells and lymphocytes (Figure 1); the activity was higher in cell lines (P < .02) and in cells from 6 of 7 patients (P < .05, with an increase of up to 5-fold). We then examined CK2 catalytic α subunit levels. They paralleled the enzymatic activity and were higher in MM cell lines and in samples from MM patients (4 of 5 examined) than in normal plasma cells and lymphocytes (Figure 2A, top panel). Comparable results were obtained by Western blot experiments for the noncatalytic β subunit of CK2, but the α′ catalytic subunit was not significantly detected in any sample (not shown). We also investigated the phospho-radiolabeling of endogenous proteins on incubation of cell extracts with γ33P-ATP and Mg++, and we observed significantly higher levels of protein phosphorylation in MM cells, with a pattern different from that found in whole cell extracts from normal plasma cells and lymphocytes. Remarkably, the intensity of the bands corresponding to some of these phosphorylated proteins was significantly reduced with the addition of the selective CK2 inhibitor TBB (data not shown).
To confirm CK2 expression levels in MM cells as observed on Western blot, we performed immunofluorescence experiments on freshly isolated bone marrow cells from MM patients in whom more than 20% of plasma cell infiltrate was in the bone marrow. As seen in 2 representative patients (Figure 2B), we observed more intense CK2 staining in CD138+ plasma cells than in the remaining cell population, thus indicating that this kinase exhibits a preferential pattern of expression in malignant MM cells. Altogether, these results suggest that CK2 protein levels and enzymatic activity are higher in MM cells than in normal plasma cells, consistent with a role for CK2 in the biology of the malignant plasma cell.
CK2 blockage causes apoptosis in MM cells
To study the role of CK2 in MM cells, we used 3 synthetic CK2 inhibitors (2 of which are structurally unrelated) previously described as strong and specific. An MTT-based assay was used to perform cell proliferation/viability analysis.7,31-35 As shown in Figure 3A, MM cells cultured for 48 hours in the presence of increasing concentrations of the compounds K27, TBB, and IQA displayed a dose-dependent growth arrest.
To prove that these inhibitors specifically affected CK2 kinase activity inside MM cells, we also performed kinase assays against a CK2-specific peptide substrate using K27 or TBB-treated MM cell lysates. As shown in Figure 3A, CK2 kinase activity was efficiently hampered in MM cells by K27 and TBB in a dose-dependent manner.
Moreover, the specificity of the CK2 inhibitors in causing growth inhibition was also determined by assessing their effects in cells transiently transfected with a CK2α mutant that bore a mutation of 2 residues (Val66 and Ile174); these mutations are crucial for the binding of TBB and TBB derivatives to CK2, and they confer resistance to TBB in vitro33 and in vivo41 without affecting all the other enzymatic and biochemical features of the native protein. Therefore, we transiently transfected HEK293 cells with GFP and either CK2α wild-type or CK2α V66A, I174A mutant coding vectors. After 48 hours, cells were GFP sorted and cultured for 8 hours with TBB (20 μM); then cells were analyzed by FACS for viability. The viability of HEK293 cells transfected with only GFP decreased by an average of 25% upon CK2 blockage, whereas cells transfected with CK2 V66A, I174A mutant were resistant to TBB-induced cell death (data not shown).
CK2α expression in MM cells. (A) Top panel: Western blot for CK2α. Bottom panel: Corresponding Western blot for α-tubulin as a loading control (different sets of experiments are shown). Analyses were performed on the same samples as in Figure 1, corresponding to MM cell lines (lanes 2-5, U266, RPMI, or OPM, as indicated), CD138+ purified plasma cells from 5 MM patients (lanes 6-11), normal plasma cells (lane 12, indicated as nPCs), normal CD19+ B lymphocytes purified from 7 healthy donors (lanes 13-19). In lane 1, recombinant CK2α was loaded as a positive control; in lanes 5 and 8, TBB (2 μM) was added in vitro to exclude the effects on CK2α levels. (B) Results of 2 representative experiments of immunofluorescence microscopy on freshly isolated bone marrow cells from 2 MM patients stained with an antibody recognizing the specific plasma cell marker CD138 (i,iv; green fluorescence), anti-CK2a (ii,v; red fluorescence), and merged images (iii,vi). Original magnification × 60, oil objective.
CK2α expression in MM cells. (A) Top panel: Western blot for CK2α. Bottom panel: Corresponding Western blot for α-tubulin as a loading control (different sets of experiments are shown). Analyses were performed on the same samples as in Figure 1, corresponding to MM cell lines (lanes 2-5, U266, RPMI, or OPM, as indicated), CD138+ purified plasma cells from 5 MM patients (lanes 6-11), normal plasma cells (lane 12, indicated as nPCs), normal CD19+ B lymphocytes purified from 7 healthy donors (lanes 13-19). In lane 1, recombinant CK2α was loaded as a positive control; in lanes 5 and 8, TBB (2 μM) was added in vitro to exclude the effects on CK2α levels. (B) Results of 2 representative experiments of immunofluorescence microscopy on freshly isolated bone marrow cells from 2 MM patients stained with an antibody recognizing the specific plasma cell marker CD138 (i,iv; green fluorescence), anti-CK2a (ii,v; red fluorescence), and merged images (iii,vi). Original magnification × 60, oil objective.
Next, we tested whether the apoptotic process could be partly responsible for this effect. OPM2 and RPMI 8226 MM cells were treated with K27 (at 2.5-μM and 5-μM concentrations) for 24 hours and then were subjected to annexin V/propidium iodide (AV/PI) staining and FACS analysis. The percentage of AV/PI-negative (not apoptotic) cells after treatment progressively decreased in a dose-dependent manner for both cell lines (Figure 3B), thus indicating that apoptosis is triggered by CK2 blockage.
To confirm that CK2 protein sustains MM cell survival, we performed RNA-interference experiments. U266 MM cells were transiently transfected with irrelevant siRNA oligonucleotides or siRNA oligonucleotides directed against the α subunit of CK2; a nonspecific siRNA labeled with cytochrome 3 was cotransfected to track transfected cells and to allow their sorting by FACS. Seventy-two to 96 hours after transfection, cells were FACS sorted, labeled with Annexin V-FITC, and analyzed by FACS. As shown in the representative experiment in Figure 3C, whereas U266 cells transfected with nonspecific siRNA remained viable, a significant proportion of CK2-directed siRNA-transfected MM cells underwent apoptosis (mean fluorescence intensity, 10% and 49% compared with the mock-transfected cells, respectively). In the right panels, Western blot analysis demonstrates the significant down-regulation of the CK2α protein levels achieved in these experiments.
Next, to check whether the inhibition of CK2 caused apoptosis of plasma cells in MM patients, we treated freshly isolated total PB or BM cells from MM patients with K27 (at 5-μM concentrations) for 8 hours. Remarkably, most of the CD138+ malignant plasma cell fractions underwent apoptosis, but the effect was less pronounced in the CD138- population (Figure 3D; top, results of 1 of 4 representative FACS experiments; bottom, results obtained by treating 4 different primary MM samples). Together, these results, showing MM cell apoptosis on pharmacologic and siRNA-induced down-regulation of CK2, support the solid evidence of a role for this kinase in MM cell survival.
Blockage of CK2 in MM cells causes activation of the extrinsic and intrinsic apoptotic pathways and enhances the cytotoxic effect of melphalan
We then sought to investigate the molecular consequences of CK2 blockage. OPM2 and RPMI8226 MM cells were treated with increasing doses of K27, and the activation of the caspase pathways was evaluated by Western blotting. As shown in Figure 4A, pro-caspase-8, pro-caspase-9, and pro-caspase-3 cleavage and the consequent cleavage of the downstream PARP enzyme were elicited in a dose-dependent manner by the addition of K27 to the cultures. Moreover, the release of cytochrome c and SMAC/DIABLO from mitochondria in the cytosol could be demonstrated in both cell lines (Figure 4B). Similar results were obtained using other MM cell lines and other CK2-specific inhibitors, notably TBB and IQA (data not shown). These findings indicate that molecular pathways associated with the induction of apoptosis are elicited in MM when CK2 is inhibited.
Because we observed the induction of MM cell apoptosis by the ablation of CK2 function, we next tested whether CK2 blockage could also render MM cells more sensitive to the cytotoxic effect of a conventional chemotherapeutic agent used in MM therapy— namely, melphalan. To this end, we treated OPM2 cells with increasing doses of melphalan in the presence of vehicle (0.1% DMSO) or K27 at subtoxic concentrations (0.3 and 1.0 μM). At both concentrations of K27, the dose of melphalan that induced 50% growth inhibition was significantly lower than the corresponding dose in the absence of CK2 blockage (Figure 4C; *P < .01, **P < .05). These data show that CK2 enhances MM-cell response to melphalan-induced cytotoxicity.
IL-6 and IGF-I affect CK2 kinase activity and are unable to promote cell growth when CK2 is inhibited
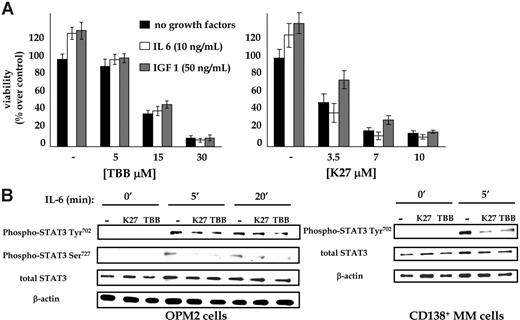
Next, we investigated whether pivotal growth signals for MM cells, namely IL-6 and IGF-I,42,43 could compensate for the loss of CK2 function, thus inducing cell growth even in the presence of CK2 inhibition. As shown in Figure 5A, IL-6 and IGF-I were unable to reverse the dose-dependent cytotoxic effect on MM cells caused by TBB or K27 on OPM2 MM cells. Comparable findings were obtained in similar experiments using IQA and other MM cell lines (data not shown). These results suggest that the pathways activated by these 2 growth factors in MM cells might rely on an intact CK2 function.
Inhibition of CK2 kinase affects IL-6 signaling
To gain more insight into the molecular mechanisms underlying the observed growth arrest and apoptosis on CK2 inhibition even in IL-6-stimulated MM cells, we analyzed the IL-6-triggered molecular events44 in OPM2 MM cell line and in purified CD138+ malignant plasma cells from MM patients. OPM2 cells were serum starved for 4 hours, incubated in serum free-medium with DMSO 0.1% or the CK2 kinase inhibitor K27 (5 μM) or TBB (20 μM) for another 2 hours, and exposed to IL-6 (10 ng/mL) for different time periods. Immunoblot analysis with antibodies against phospho-Tyr702-STAT3, phospho-Ser727-STAT3, and total STAT3 was then performed to check for activation of this IL-6-triggered signaling cascade (Figure 5B). On IL-6 stimulation of OPM2 cells (panels at the left), there was an increase in the levels of phosphorylated STAT3 at residues Tyr702 and Ser727; however, when OPM2 MM cells were preincubated with the CK2 inhibitor K27 or TBB, the IL-6-induced time-dependent STAT3 phosphorylation was decreased, with the strongest effect observed after 5 minutes and affecting Ser727 phosphorylation. In separate experiments (panels at the right), CD138+ MM cells from patients were left untreated or were treated with IL-6 for 5 minutes after incubation in the presence of 0.1% DMSO or with 5 μM or 20 μM TBB. As shown, we observed that even in freshly purified malignant plasma cells, IL-6-triggered STAT3 activation could be hampered by the blockage of CK2 (in these experiments, STAT phosphorylation on Tyr702 was analyzed). A possible inhibition of Jak tyrosine kinases and a general effect on total tyrosine phosphorylation is highly unlikely because the inhibitors used did not display any activity on a panel of tyrosine kinases tested34,35 and the pattern of total phosphotyrosine remained unchanged in treated MM cell lysates (data not shown). Although the exact significance of these findings warrants further investigation, they suggest that proper IL-6-triggered STAT3 activation is modulated by CK2 and involves this kinase in a crucial growth-promoting pathway in MM.
Effects of CK2 blockage on MM cell viability. (A) Top panels: MM cell lines (legend indicated at the far right) were grown for 48 hours in the absence or presence of increasing concentrations (as indicated) of the selective CK2 inhibitors. Viability was measured by the MTT method and is expressed as a percentage of vehicle-treated control. Bottom panels: Level of CK2 enzymatic activity was measured in MM cell (OPM2) lysates treated with the indicated concentration of inhibitors. For each inhibitor, results of 1 of 6 different experiments is shown; data are the mean ± SD. (B) Apoptosis of MM cell lines by CK2 blockage as measured by FACS analysis of annexin V/propidium iodide (AV/PI)-stained MM cells. Graph shows the survival (as AV-negative cells) of OPM2 and RPMI 8226 cells treated for 24 hours with 2 different doses of the CK2 inhibitor K27 (2.5 and 5 μM); data are the mean ± SD. (C) CK2α knock-down by RNA interference is associated with MM cell apoptosis. Left: Histogram shows the amount of AV-positive U266 MM cells after 96 hours from mock transfection or transfection with control (ctr) siRNAs or CK2α-specific siRNAs. Right: Western blot analysis of CK2α levels in mock, control (ctr) siRNA-, and CK2α-directed siRNA-transfected U266 MM cells. (D) Apoptosis of freshly isolated MM cells from patients on CK2 blockage. Top: Results of representative FACS experiment of AV/CD138 staining of freshly isolated BM cells from an MM patient (MM7) treated with K27 (5 μM); FACS analysis of apoptotic cells in the CD138+ and CD138- fractions is shown. Bottom: Graph shows the percentage of apoptotic cells (in the malignant CD138+ and nonmalignant CD138- populations) in freshly isolated PB or BM cells from 4 MM patients (MM1, MM2, MM4, MM7). Cells were treated with vehicle or K27, and apoptosis was assessed 8 hours later.
Effects of CK2 blockage on MM cell viability. (A) Top panels: MM cell lines (legend indicated at the far right) were grown for 48 hours in the absence or presence of increasing concentrations (as indicated) of the selective CK2 inhibitors. Viability was measured by the MTT method and is expressed as a percentage of vehicle-treated control. Bottom panels: Level of CK2 enzymatic activity was measured in MM cell (OPM2) lysates treated with the indicated concentration of inhibitors. For each inhibitor, results of 1 of 6 different experiments is shown; data are the mean ± SD. (B) Apoptosis of MM cell lines by CK2 blockage as measured by FACS analysis of annexin V/propidium iodide (AV/PI)-stained MM cells. Graph shows the survival (as AV-negative cells) of OPM2 and RPMI 8226 cells treated for 24 hours with 2 different doses of the CK2 inhibitor K27 (2.5 and 5 μM); data are the mean ± SD. (C) CK2α knock-down by RNA interference is associated with MM cell apoptosis. Left: Histogram shows the amount of AV-positive U266 MM cells after 96 hours from mock transfection or transfection with control (ctr) siRNAs or CK2α-specific siRNAs. Right: Western blot analysis of CK2α levels in mock, control (ctr) siRNA-, and CK2α-directed siRNA-transfected U266 MM cells. (D) Apoptosis of freshly isolated MM cells from patients on CK2 blockage. Top: Results of representative FACS experiment of AV/CD138 staining of freshly isolated BM cells from an MM patient (MM7) treated with K27 (5 μM); FACS analysis of apoptotic cells in the CD138+ and CD138- fractions is shown. Bottom: Graph shows the percentage of apoptotic cells (in the malignant CD138+ and nonmalignant CD138- populations) in freshly isolated PB or BM cells from 4 MM patients (MM1, MM2, MM4, MM7). Cells were treated with vehicle or K27, and apoptosis was assessed 8 hours later.
Regulation of IκBα degradation by CK2 in MM cells
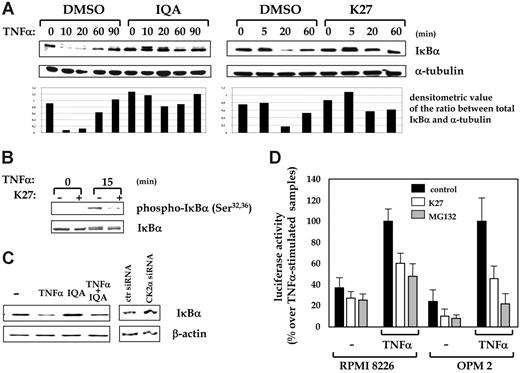
We next asked whether CK2 could regulate the NF-κB signaling pathway, another cascade crucial for MM cell growth. We cultured OPM2 or RPMI 8226 cells in the presence of vehicle (0.1% DMSO) or CK2 inhibitors for 60 minutes, and then we added TNF-α to stimulate NF-κB. IκBα degradation was then assessed by Western blot at different time points. Two representative experiments performed with CK2 inhibitors IQA (25 μM) and K27 (5 μM) are shown in Figure 6A. Notably, we could observe that the TNF-α-stimulated time-dependent IκBα degradation, which was seen in vehicle-pretreated samples, was instead reduced in the IQA- or K27-pretreated cells. Next, we asked whether this alteration could depend on an altered phosphorylation of IκBα at serines 32 and 36, which are typically targeted by the kinase IKK but can also be substrates of CK2.15 As shown in Figure 6B, IκBα phosphorylation at serines 32 and 36 on TNF-α stimulation was decreased when CK2 was blocked by K27. To exclude that the compounds used in these experiments might affect the kinase activity of IKK, we performed in vitro testing on their effect on this kinase, which was not included in the panel of kinases routinely used to evaluate the selectivity of CK2 inhibitors.33 Consistent with their reported selectivity for CK2, we did not find any significant efficacy of CK2 inhibitors on IKK (data not shown).
Molecular effects of CK2 blockage in MM cells. (A) Western blot shows the molecular consequences of CK2 inhibition in MM cells: cleavage of pro-caspases 8, 9, and 3 and of the downstream target PARP on treatment of OPM2 cells with increasing concentrations of K27 for 12 hours. (B) Western blot shows mitochondrial apoptotic mediators cytochrome c and SMAC/DIABLO release in the cytosol of OPM2 and RPMI 8226 cells treated with the indicated increasing concentrations of K27 for 12 hours. (C) CK2 blockage lowers the threshold of sensitivity to the cytotoxic effect of melphalan of MM cells. OPM2 cells were treated with the indicated increasing doses of melphalan in the absence (▪) or in the presence of subtoxic doses of the CK2 inhibitor K27 (0.3 μM, □;1 μM,  ) and were subjected to MTT-based viability assay 48 hours later. Data are the mean ± SD. **P < .05; *P < .02.
) and were subjected to MTT-based viability assay 48 hours later. Data are the mean ± SD. **P < .05; *P < .02.
Molecular effects of CK2 blockage in MM cells. (A) Western blot shows the molecular consequences of CK2 inhibition in MM cells: cleavage of pro-caspases 8, 9, and 3 and of the downstream target PARP on treatment of OPM2 cells with increasing concentrations of K27 for 12 hours. (B) Western blot shows mitochondrial apoptotic mediators cytochrome c and SMAC/DIABLO release in the cytosol of OPM2 and RPMI 8226 cells treated with the indicated increasing concentrations of K27 for 12 hours. (C) CK2 blockage lowers the threshold of sensitivity to the cytotoxic effect of melphalan of MM cells. OPM2 cells were treated with the indicated increasing doses of melphalan in the absence (▪) or in the presence of subtoxic doses of the CK2 inhibitor K27 (0.3 μM, □;1 μM,  ) and were subjected to MTT-based viability assay 48 hours later. Data are the mean ± SD. **P < .05; *P < .02.
) and were subjected to MTT-based viability assay 48 hours later. Data are the mean ± SD. **P < .05; *P < .02.
We then sought to analyze IκBα basal turnover once CK2 function was abrogated. We treated MM cells with vehicle or with TNF-α or IQA or both for 12 hours and assessed total IκBα levels by Western blot. TNF-α stimulation led to a considerable reduction in IκBα compared with untreated samples. On the contrary, the presence of IQA caused IκBα to accumulate. The addition of both TNF-α and IQA resulted overall in accelerated IκBα degradation that was significantly less pronounced than the sample treated with TNF-α alone (Figure 6C, left panels); similar results were obtained using K27 (not shown). Last, we checked the levels of IκBα in U266 MM cells transfected with control siRNA or CK2α-specific siRNA. After 72 to 96 hours of transfection, we could observe that in MM cells in which CK2α was silenced, IκBα levels were more abundant. (Figure 6C, right panels). These results demonstrated that CK2 might be involved in stimulus-induced and basal IκBα phosphorylation and degradation in MM cells.
CK2 inhibition affects NF-κB activity in MM cells
Next, because we observed that CK2-regulated IκBα degradation in MM cells and previous studies showed a dependence on CK2 for NF-κB to achieve full transcriptional competence on TNF-α and IL-1 treatment,16,17 we checked NF-κB transcriptional activity in MM cells in which CK2 was inhibited. As shown in Figure 6D, luciferase assays using OPM2 or RPMI 8226 MM cell lines transiently transfected with a κB-luciferase reporter plasmid revealed that basal and TNF-α-stimulated NF-κB-specific transcriptional activities were hampered by the inhibition of CK2. As a control, cells were treated with the proteasome inhibitor MG132, which also caused a reduction in the NF-κB transcriptional activity.
CK2 activity downstream from growth factors. (A) Effects of CK2 inhibition on MM cell growth (▪) were not reversed by the addition of survival stimuli such as IL-6 (20 ng/mL) (□) or IGF-I (100 ng/mL) ( ). OPM2 cells were cultured with the indicated stimuli in the absence or in the presence of TBB, K27, or both and then were subjected to MTT-based viability assay 48 hours later. Data are the mean ± SD. (B) Western blot analysis of the IL-6/STAT3 cascade in MM cells on CK2 inhibition. MM cells were serum starved for 4 hours (cell lines OPM2) or 2 hours (freshly isolated MM plasma cells from patients), incubated for 2 hours with 0.1% DMSO, (5 μM) K27, or 20 μM TBB and then stimulated for the indicated time points with 10 ng/mL IL-6. For MM cell lines, the levels of phospho-Tyr702 STAT3, phospho-Ser727 STAT3, and total STAT3, and for MM cells from patients the levels of phospho-Tyr702 STAT3 and total STAT3 were assessed, as indicated. β-Actin was used to measure protein loading.
). OPM2 cells were cultured with the indicated stimuli in the absence or in the presence of TBB, K27, or both and then were subjected to MTT-based viability assay 48 hours later. Data are the mean ± SD. (B) Western blot analysis of the IL-6/STAT3 cascade in MM cells on CK2 inhibition. MM cells were serum starved for 4 hours (cell lines OPM2) or 2 hours (freshly isolated MM plasma cells from patients), incubated for 2 hours with 0.1% DMSO, (5 μM) K27, or 20 μM TBB and then stimulated for the indicated time points with 10 ng/mL IL-6. For MM cell lines, the levels of phospho-Tyr702 STAT3, phospho-Ser727 STAT3, and total STAT3, and for MM cells from patients the levels of phospho-Tyr702 STAT3 and total STAT3 were assessed, as indicated. β-Actin was used to measure protein loading.
CK2 activity downstream from growth factors. (A) Effects of CK2 inhibition on MM cell growth (▪) were not reversed by the addition of survival stimuli such as IL-6 (20 ng/mL) (□) or IGF-I (100 ng/mL) ( ). OPM2 cells were cultured with the indicated stimuli in the absence or in the presence of TBB, K27, or both and then were subjected to MTT-based viability assay 48 hours later. Data are the mean ± SD. (B) Western blot analysis of the IL-6/STAT3 cascade in MM cells on CK2 inhibition. MM cells were serum starved for 4 hours (cell lines OPM2) or 2 hours (freshly isolated MM plasma cells from patients), incubated for 2 hours with 0.1% DMSO, (5 μM) K27, or 20 μM TBB and then stimulated for the indicated time points with 10 ng/mL IL-6. For MM cell lines, the levels of phospho-Tyr702 STAT3, phospho-Ser727 STAT3, and total STAT3, and for MM cells from patients the levels of phospho-Tyr702 STAT3 and total STAT3 were assessed, as indicated. β-Actin was used to measure protein loading.
). OPM2 cells were cultured with the indicated stimuli in the absence or in the presence of TBB, K27, or both and then were subjected to MTT-based viability assay 48 hours later. Data are the mean ± SD. (B) Western blot analysis of the IL-6/STAT3 cascade in MM cells on CK2 inhibition. MM cells were serum starved for 4 hours (cell lines OPM2) or 2 hours (freshly isolated MM plasma cells from patients), incubated for 2 hours with 0.1% DMSO, (5 μM) K27, or 20 μM TBB and then stimulated for the indicated time points with 10 ng/mL IL-6. For MM cell lines, the levels of phospho-Tyr702 STAT3, phospho-Ser727 STAT3, and total STAT3, and for MM cells from patients the levels of phospho-Tyr702 STAT3 and total STAT3 were assessed, as indicated. β-Actin was used to measure protein loading.
CK2 regulates the NF-κB pathway in MM cells. (A) TNF-α-induced IκBα degradation is impaired in MM cells by blockage of CK2. RPMI 8226 cells were incubated for 60 minutes with 0.1% DMSO or with the CK2 inhibitors IQA (25 μM) or K27 (5 μM), and then TNF-α (10 ng/mL) was added to the cultures. At the indicated time points, cytosolic proteins were harvested and 30 μg was used in Western blot analysis with an anti-IκBα-specific antibody (even protein loading was checked to assess α-tubulin levels). Below each Western blot, a graph displays the ratio between the intensity of the bands corresponding to IκBα and α-tubulin, as assessed by densitometric analysis. (B) TNF-α-induced IκBα Ser32 and Ser36 phosphorylation is reduced on CK2 inhibition. RPMI 8226 cells were grown for 60 minutes with 0.1% DMSO (-) or in the presence (+) of the CK2 inhibitor K27 (5 μM) and then stimulated for 0 or 15 minutes with TNF-α (10 ng/mL). Levels of phospho-Ser32 and phospho-Ser36 IκBα were then assessed by immunoblotting 30 μg of proteins with a phosphospecific antibody. (C) IκBα accumulation on CK2 inhibition or down-regulation by RNA interference. Left: RPMI 8226 cells were grown for 12 hours in the presence of medium (-), TNF-α (10 ng/mL), the CK2 inhibitor IQA (25 μM), or both, and then IκBα protein levels were assessed by immunoblotting with an anti-IκBα-specific antibody. Right: Similarly, IκBα levels were determined in MM cells in which CK2 mRNA and protein expression were silenced by siRNAs (even protein loading was checked to assess β-actin levels). (D) Blockage of CK2 reduced NF-κB transcriptional activity in MM cells. RPMI 8226 and OPM2 cells were transiently transfected with an NF-κB-driven luciferase reporter construct and the construct pRL-TK (see “Materials and methods” for details). Twenty-four hours later, the CK2 inhibitor K27 (5 μM) (white bars) or the proteasome inhibitor MG132 (15 μM) ( ) was added to the cultures for 90 minutes, and the cells were stimulated with TNF-α (10 ng/mL) or were left untreated (▪). After 6 hours, total luciferase activity was determined. Values in the graph are shown as percentage of the TNF-α-treated samples. Cell viability was higher than 80% in all the experimental conditions (data not shown). Data represent mean ± SD.
) was added to the cultures for 90 minutes, and the cells were stimulated with TNF-α (10 ng/mL) or were left untreated (▪). After 6 hours, total luciferase activity was determined. Values in the graph are shown as percentage of the TNF-α-treated samples. Cell viability was higher than 80% in all the experimental conditions (data not shown). Data represent mean ± SD.
CK2 regulates the NF-κB pathway in MM cells. (A) TNF-α-induced IκBα degradation is impaired in MM cells by blockage of CK2. RPMI 8226 cells were incubated for 60 minutes with 0.1% DMSO or with the CK2 inhibitors IQA (25 μM) or K27 (5 μM), and then TNF-α (10 ng/mL) was added to the cultures. At the indicated time points, cytosolic proteins were harvested and 30 μg was used in Western blot analysis with an anti-IκBα-specific antibody (even protein loading was checked to assess α-tubulin levels). Below each Western blot, a graph displays the ratio between the intensity of the bands corresponding to IκBα and α-tubulin, as assessed by densitometric analysis. (B) TNF-α-induced IκBα Ser32 and Ser36 phosphorylation is reduced on CK2 inhibition. RPMI 8226 cells were grown for 60 minutes with 0.1% DMSO (-) or in the presence (+) of the CK2 inhibitor K27 (5 μM) and then stimulated for 0 or 15 minutes with TNF-α (10 ng/mL). Levels of phospho-Ser32 and phospho-Ser36 IκBα were then assessed by immunoblotting 30 μg of proteins with a phosphospecific antibody. (C) IκBα accumulation on CK2 inhibition or down-regulation by RNA interference. Left: RPMI 8226 cells were grown for 12 hours in the presence of medium (-), TNF-α (10 ng/mL), the CK2 inhibitor IQA (25 μM), or both, and then IκBα protein levels were assessed by immunoblotting with an anti-IκBα-specific antibody. Right: Similarly, IκBα levels were determined in MM cells in which CK2 mRNA and protein expression were silenced by siRNAs (even protein loading was checked to assess β-actin levels). (D) Blockage of CK2 reduced NF-κB transcriptional activity in MM cells. RPMI 8226 and OPM2 cells were transiently transfected with an NF-κB-driven luciferase reporter construct and the construct pRL-TK (see “Materials and methods” for details). Twenty-four hours later, the CK2 inhibitor K27 (5 μM) (white bars) or the proteasome inhibitor MG132 (15 μM) ( ) was added to the cultures for 90 minutes, and the cells were stimulated with TNF-α (10 ng/mL) or were left untreated (▪). After 6 hours, total luciferase activity was determined. Values in the graph are shown as percentage of the TNF-α-treated samples. Cell viability was higher than 80% in all the experimental conditions (data not shown). Data represent mean ± SD.
) was added to the cultures for 90 minutes, and the cells were stimulated with TNF-α (10 ng/mL) or were left untreated (▪). After 6 hours, total luciferase activity was determined. Values in the graph are shown as percentage of the TNF-α-treated samples. Cell viability was higher than 80% in all the experimental conditions (data not shown). Data represent mean ± SD.
CK2 associates with NF-κB p50/p105 in MM cells
Given our findings, we wanted to determine whether CK2 could physically interact with NF-κB members, namely p65 and p50. We performed immunofluorescence and confocal microscopy experiments to analyze the relative localization of the endogenous CK2α and NF-κB p65 or p50/105 in MM cells. CK2α was found to be distributed in the nucleus and in the cytoplasm (Figure 7A, D).45 We observed that NF-κB p65 was predominantly retained in the cytoplasm of OPM2 MM cells, whereas p50/105 was mainly localized in the nucleus (Figure 7B, E). When double staining was performed, we detected CK2α colocalization with p65 in few dots in the cytoplasm (Figure 7C). Remarkably, CK2α was found colocalized abundantly with p50/105 in the nucleus in small, discrete dots (Figure 7F). To confirm the possibility that CK2 is associated with p50/105 in MM cells, we performed coimmunoprecipitation experiments against the endogenous proteins. As shown in Figure 7G, we detected an interaction between the endogenous CK2α and p50/105 in MM cells because we were able to pull down CK2α by immunoprecipitating p50; the reverse was also true (data not shown). These results strongly suggested that CK2α interacts with NF-κB p50/105 in MM cells.
Discussion
In the present study we have shown that protein kinase CK2 is aberrantly active in MM cells and controls their survival. We have also demonstrated that CK2 might be involved in the control of the IL-6 and NF-κB signaling pathways in these cells, and we have shown a previously unrecognized interaction of this kinase with the NF-κB member p50/105.
The increased CK2 protein levels and activity we found in neoplastic MM cells (compared with normal in vitro-generated polyclonal plasma cells and B lymphocytes; Figures 1, 2) was likely not simply related to the proliferation rate because it was comparable in highly proliferating MM cell lines, in slowly proliferating purified CD138+ MM cells freshly isolated from the BM of patients, and in more aggressive extramedullary myelomas (Figure 1; Table 1), suggesting that the perturbation of CK2 function is a direct consequence of the transformed phenotype rather than an epiphenomenon of high cell proliferation, as indicated in other studies.12 Moreover, this concept was corroborated by the lower levels of CK2 expression and activity we detected in normal plasma cells, a significant fraction of which were highly proliferative.29 The biologic consequences of increased activity of CK2 in MM cells are predicted to be relevant because this kinase normally displays basal enzymatic activity directed toward a number of substrates involved in several important cellular processes. Therefore, physiologically, CK2 levels have to be tightly regulated to avoid perturbations of cell homeostasis and behavior. Given that we found that the increase in CK2 protein equally affected α and β subunits in MM cells, the observed dysregulated activity cannot be ascribed to an unbalanced expression of catalytic versus regulatory subunits of CK2.
CK2 associates with NF-κB in MM cells. (A-C) Confocal immunofluorescence microscopy of CK2 (Texas red), NF-κB p65/RelA (green, Alexa 488), and merge showing scant colocalization (white arrows) of the 2 proteins in the cytoplasm in OPM2 cells. (D-F) Confocal immunofluorescence microscopy of CK2 (green, Alexa 488), NF-κB p50/105 (Texas red), and merge showing abundant colocalization (white arrows) of the 2 proteins in the nucleus in OPM2 cells (600× magnification, oil, top panels, 1000× magnification, oil, bottom panels). (G) Association of endogenous CK2α and NF-κB p50/105 in MM cells. Coimmunoprecipitation of endogenous CK2α and NF-κB p50/105 in OPM2 MM cells. Cell lysates were immunoprecipitated with a preimmune rabbit serum (lane 1) or with an antibody recognizing p50/105 (lane 2) and then immunoblotted for CK2α (top) and NF-κB p50/105 (bottom). Input loading is shown in lane 3.
CK2 associates with NF-κB in MM cells. (A-C) Confocal immunofluorescence microscopy of CK2 (Texas red), NF-κB p65/RelA (green, Alexa 488), and merge showing scant colocalization (white arrows) of the 2 proteins in the cytoplasm in OPM2 cells. (D-F) Confocal immunofluorescence microscopy of CK2 (green, Alexa 488), NF-κB p50/105 (Texas red), and merge showing abundant colocalization (white arrows) of the 2 proteins in the nucleus in OPM2 cells (600× magnification, oil, top panels, 1000× magnification, oil, bottom panels). (G) Association of endogenous CK2α and NF-κB p50/105 in MM cells. Coimmunoprecipitation of endogenous CK2α and NF-κB p50/105 in OPM2 MM cells. Cell lysates were immunoprecipitated with a preimmune rabbit serum (lane 1) or with an antibody recognizing p50/105 (lane 2) and then immunoblotted for CK2α (top) and NF-κB p50/105 (bottom). Input loading is shown in lane 3.
We also found that MM cells contain an array of abnormally phosphorylated proteins, some of which are likely to be CK2 targets given their responsiveness to CK2 inhibitors. This finding indicated that plasma cell malignant transformation is accompanied by a deregulated signaling to which CK2 appears to contribute.
Our experiments also showed that the pharmacologic disruption of CK2 signaling in malignant plasma cells with specific inhibitors caused apoptosis (Figures 3, 4A-B). The compounds used in some cases were structurally unrelated, and the concentrations required to induce the cellular effects were roughly proportional to their Ki for CK2 in vitro.31-33 Moreover, our findings that cells in which a CK2 mutant resistant to the inhibitors was transfected are resistant to cell death induced by the same inhibitors indicate that they act specifically on CK2.
Furthermore, we also provided strong molecular evidence of the pivotal role of CK2 kinase in MM cell survival by showing that knocking down CK2α protein expression by RNA interference is accompanied by MM cell apoptosis (Figure 3C). These findings, therefore, strongly support the conclusion that the apoptotic effects observed in this study are actually mediated by CK2.
It was known that CK2 is required for cell survival from in vivo deletion studies46,47 and other investigations, many of which were carried out with the same inhibitors used in this study.7,8,48-51 Our observation that both the intrinsic and the extrinsic apoptotic pathways are triggered on CK2 inhibition in MM cells might reflect a role for this kinase in checkpoints shared by the 2 apoptotic cascades in this cell type. This role might derive from the CK2-dependent control of the rate of caspase-triggered degradation of certain protein substrates as a more general mechanism of which a number of examples are already known.7,10,11,49,52 As previously demonstrated, it is also possible that CK2 prevents the activation of pro-caspase-8 in the absence of death receptor engagement.6 CK2 inhibition enhances TRAIL-induced apoptosis of rhabdomyosarcoma, colon cancer, and prostate cancer cells51,53,54 as well as MM cells (F.A.P., B.M., and G.S., unpublished data, 2006), and recently CK2 was found to control the activation of pro-caspase-8 by caspase-2.55 Furthermore, we have also revealed a role for CK2 in MM cell survival by showing that this kinase protects MM cells from the cytotoxic effect of melphalan, an alkylating DNA agent used as a chemotherapeutic drug in MM therapy (Figure 4C). In this regard, CK2 has recently been shown to play a role in the repair of DNA damage.56
In the present work, we have also shown that IL-6 and IGF-I, 2 MM cell survival factors, very likely involve CK2 activity in their signaling cascades because we found that these 2 important MM growth factors were unable to induce cell growth in the absence of functioning CK2 (Figure 5A). Moreover, with regard to the IL-6 pathway, we demonstrated that CK2 might be an important regulator of the downstream intracellular signaling events triggered by this cytokine, particularly STAT3 phosphorylation on Tyr702 and Ser727 (Figure 5B). Recently, this pathway has been demonstrated to be constitutively activated and to mediate MM cell survival.57 Based on our results, however, the exact mechanism through which CK2 controls the rate of IL-6-dependent STAT3 activation is still unclear. It is unlikely that CK2 inhibitors affected tyrosine phosphorylation because we could not find differences in the patterns of total phosphotyrosine expression in K27- or TBB-treated MM cells. Because Tyr702-STAT3 phosphorylation was found to be impaired, it is conceivable that CK2 could act on early events turned on in this pathway. These results, even though warranting further study, are remarkable because they unravel novel molecular interactions important for MM cell growth. The identification of CK2 targets downstream from IL-6 and other stimuli, such as insulin-like growth factor-I (IGF-I), vascular endothelial growth factor (VEGF),58 and fibroblast growth factor (FGF),59 would add insight into the regulation of these pathways in MM. Such identification is under way in our laboratory.
We also found that CK2 is involved in the NF-κB pathway in MM cells, modulating the degradation of IκBα and the transcriptional activity of NF-κB (Figure 6). Previous studies have shown that CK2-driven IκBα degradation could depend on proteasome-dependent and proteasome-independent mechanisms.60,61 How these processes are regulated is still largely unknown. Interestingly, we have shown that the phosphorylation of IκBα on Ser32 and Ser36 on TNF-α stimulation is partly compromised when CK2 is inhibited. Our results extend the findings of others who established a direct role for CK2 in phosphorylating IκBα Ser32 and Ser36.15 Nonetheless, we did not prove that IκBα is a substrate of CK2 in MM cells; hence, it may be that this effect is indirect. It should be mediated by CK2 because we have shown the unlikelihood that the CK2 inhibitors used in this study directly affected upstream IκBα kinases, such as IKK, as proven by their ineffectiveness in in vitro kinase assay experiments. In addition, a weakened CK2 function was associated with impairment of the TNF-α-induced NF-κB-dependent transcriptional activity. Remarkably, in previous studies, the direct phosphorylation by CK2 on Ser529 has been shown to augment NF-κB p65 transcriptional activity.16 However, little is known about a potential physical association between CK2 and NF-κB. In our experiments (Figure 7), though endogenous CK2α and NF-κB p50/105 were found to colocalize in the nucleus in MM cells and to interact by coimmunoprecipitation, CK2α colocalized scantily with NF-κB p65 in the cytosol and did not coimmunoprecipitate (data not shown). Clearly, the exact significance of these observations, previously unrecognized in MM cells, must be addressed. It is possible that CK2 exerts a positive regulatory effect on the NF-κB-driven gene transcription by enhancing IκBα degradation and, perhaps, by influencing the association of p50/p50 homodimers or p65/p50 heterodimers with DNA or with coactivators/corepressors by means of phosphorylation. It will be instrumental to address whether the interaction of CK2 with the NF-κB pathway in MM (and perhaps in other malignancies) might have a pathogenetic significance or whether it can play a role in the mechanisms underlying novel therapies based on proteasome inhibition and disruption of NF-κB signaling. In conclusion, our data establish a role for the CK2 serine-threonine kinase in the biology of MM plasma cells and suggest the possibility that CK2 might represent a potential target in MM therapy.
Prepublished online as Blood First Edition Paper, May 9, 2006; DOI 10.1182/blood-2005-11-013672.
Supported by grants from the International Myeloma Foundation (F.A.P.), the Associazione Italiana per la Ricerca sul Cancro (AIRC) (G.S., L.A.P.), Istituto Oncologico Veneto (IOV) (G.S.), and the European Commission (Pro-Kinase Research 503467) (L.A.P.).
The publication costs of this article were defrayed in part by page charge payment. Therefore, and solely to indicate this fact, this article is hereby marked “advertisement” in accordance with 18 U.S.C. section 1734.
We thank Dr R. Dalla Favera for critical reading of the manuscript; Drs M. Salvi and S. Sarno (Department of Biological Chemistry, University of Padova, Italy) for providing the CK2 mutant expression vector; Dr S. Indraccolo (Department of Oncological Sciences, University of Padova, Italy) for helpful suggestions; Dr M. E. Donach for reading the manuscript; Dr J. Gordon for his generous gift of CD40L-transfectant L cells; and the protein production team of the Division of Signal Transduction Therapy at the University of Dundee, Scotland (coordinated by Hilary McLauchlan and James Hastie), for providing active IKKβ.




This feature is available to Subscribers Only
Sign In or Create an Account Close Modal