Abstract
Gambogic acid (GA), a xanthone derived from the resin of the Garcinia hanburyi, has been recently demonstrated to bind transferrin receptor and exhibit potential anticancer effects through a signaling mechanism that is not fully understood. Because of the critical role of NF-κB signaling pathway, we investigated the effects of GA on NF-κB–mediated cellular responses and NF-κB–regulated gene products in human leukemia cancer cells. Treatment of cells with GA enhanced apoptosis induced by tumor necrosis factor (TNF) and chemotherapeutic agents, inhibited the expression of gene products involved in antiapoptosis (IAP1 and IAP2, Bcl-2, Bcl-xL, and TRAF1), proliferation (cyclin D1 and c-Myc), invasion (COX-2 and MMP-9), and angiogenesis (VEGF), all of which are known to be regulated by NF-κB. GA suppressed NF-κB activation induced by various inflammatory agents and carcinogens and this, accompanied by the inhibition of TAK1/TAB1-mediated IKK activation, inhibited IκBα phosphorylation and degradation, suppressed p65 phosphorylation and nuclear translocation, and finally abrogated NF-κB–dependent reporter gene expression. The NF-κB activation induced by TNFR1, TRADD, TRAF2, NIK, TAK1/TAB1, and IKKβ was also inhibited. The effect of GA mediated through transferrin receptor as down-regulation of the receptor by RNA interference reversed its effects on NF-κB and apoptosis. Overall our results demonstrate that GA inhibits NF-κB signaling pathway and potentiates apoptosis through its interaction with the transferrin receptor.
Introduction
Although plant-derived products have served humankind as treatments of various ailments for centuries, neither their active components nor their molecular targets are fully understood. Identifying the active chemical entities and their molecular targets can lead to discovering new clinical uses of such products, as in the cases of vincristine, vinblastine, paclitaxel, camptothecin, roscovitin, and homoharringtonine.1 Between 1980 and 2000, as many as 70% of all drugs approved by the US Food and Drug Administration to treat cancer were based on natural sources.1,2
Gambogic acid (GA) is a naturally occurring brownish-to-orange resin called gamboge, which is derived from Garcinia hanburyi. It has a long history of medicinal use in Southeast Asia, and it is used as a folk medicine and coloring agent in China. Recent studies showed that GA can inhibit the growth of a wide variety of tumor cells, including cells of human hepatoma,3 breast cancer,4 gastric carcinoma,5-8 and lung carcinoma.9 Using cell- and caspase-based high-throughput screening assays, Zhang et al identified GA as a potent inducer of apoptosis.4 Studies have also indicated that GA suppresses the growth of human tumors (eg, lung carcinoma,9 and hepatoma).3 How GA mediates these effects is not fully understood, but it has been shown to inhibit telomerase and telomerase reverse transcriptase mRNA expression,3,8,9 inhibit human telomerase reverse transcriptase (hTERT) promoter,8 suppress cyclin-dependent kinase 7 (CDK7)–mediated phosphorylation of CDC2/34,7 down-regulate Bcl-2,5 and interact with c-Myc.3 A recent report suggests that GA mediates its apoptotic effects through its interaction with the transferrin receptor (TfR1).10 Because hTERT, c-Myc, and Bcl-2 gene expression modulated by GA is regulated by NF-κB activation, it is possible that GA mediates its effects by modulating the NF-κB pathway.
NF-κB is a transcription factor that consists of 5 proteins: c-Rel, RelA (p65), RelB, NF-κB1 (p50 and p105), and NF-κB2 (p52).11 They are regulated by inhibitors of the inhibitory subunit of NF-κB (IκB) family of anchorin domain-containing proteins, which includes IκBα, IκBβ, IκBγ, IκBϵ, Bcl-3, p105, and p100.11 When inactive, NF-κB is sequestered in the cytoplasm as a heterotrimer consisting of p50, p65, and IκB subunits. Most carcinogens, inflammatory agents, and tumor promoters, including cigarette smoke, phorbol ester, okadaic acid, H2O2, and tumor necrosis factor (TNF), have been shown to activate NF-κB. In response to an activation signal, the IκBα subunit is phosphorylated at serine residues 32 and 36, ubiquitinated at lysine residues 21 and 22, and degraded through the proteasomal pathway, thus exposing the nuclear localization signals on the p50-p65 heterodimer. The p65 is then phosphorylated, leading to nuclear translocation and binding to a specific sequence in DNA, which in turn results in gene transcription. The phosphorylation of IκBα is catalyzed by IκBα kinase (IKK), which is essential for NF-κB activation. IKK consists of 3 subunits, IKK-α, IKK-β, and IKK-γ (also called NEMO).12 Gene deletion studies have indicated that IKK-α subunit has been shown to be essential for NF-κB activation by most agents.12 NF-κB regulates the expression of several genes whose products are involved in tumorigenesis. These include antiapoptotic genes (eg, Bcl-2, Bcl-xL, cIAP, survivin, and TRAF1); COX-2; MMP-9; genes encoding adhesion molecules, chemokines, and inflammatory cytokines; and cell cycle regulatory genes (eg, cyclin D1 and c-Myc).13 Because apoptosis and tumorigenesis are known to be regulated by NF-κB–regulated gene products, we hypothesized that the NF-κB pathway may play a critical in the mechanism of action of GA.
We investigated whether GA exerts its anticarcinogenic effects by suppressing the NF-κB pathway. We found that GA inhibited the activation of NF-κB; down-regulated NF-κB–regulated gene products; and potentiated apoptosis through its interaction with transferrin receptor.
Materials and methods
Reagents
GA (Figure 1A) was obtained from Gaia Chemical (Gaylordsville, CT). A 25-mM solution of GA was prepared in dimethyl sulfoxide, stored as small aliquots at −20°C, and then diluted as needed in cell culture medium. Bacteria-derived recombinant human TNF purified to homogeneity with a specific activity of 5 × 107 U/mg, was kindly provided by Genentech (South San Francisco, CA). Cigarette smoke condensate (CSC), prepared as previously described,14 was kindly supplied by Dr C. Gary Gairola (University of Kentucky, Lexington, KY). Penicillin, streptomycin, RPMI 1640 medium, and fetal bovine serum (FBS) were obtained from Invitrogen (Grand Island, NY). Phorbol myristate acetate (PMA), lipopolysaccharide (LPS), okadaic acid, H2O2, and anti–β-actin antibody were obtained from Sigma-Aldrich (St Louis, MO). Antibodies against p65, p50, IκBα, cyclin D1, MMP-9, c-Myc, IAP1, IAP2, Bcl-2, Bcl-xL, TRAF1, transferrin receptor1 (CD71), poly (adenosine diphosphatase-ribose) polymerase (PARP), COX-2, and an annexin V staining kit were obtained from Santa Cruz Biotechnology (Santa Cruz, CA). Phospho-specific anti-IκBα (Ser32/36) and phospho-specific anti-p65 (Ser536) were purchased from Cell Signaling (Beverly, MA). Anti–IKK-α and anti–IKK-β antibodies were kindly provided by Imgenex (San Diego, CA). siRNA for human transferrin receptor 1 (TfR1) and scrambled negative control were obtained from Ambion (Austin, TX). The target sequence for TfR1 siRNA was 5′-AAC TTC AAG GTT TCT GCC AGC-3′.10 Oligonucleotide probes used for the analysis of TfR mRNA transcripts were obtained from Invitrogen. Expression vector plasmids for transforming growth factor-β–activated kinase (TAK1) and TAK1-binding protein (TAB1) have been described previously.15
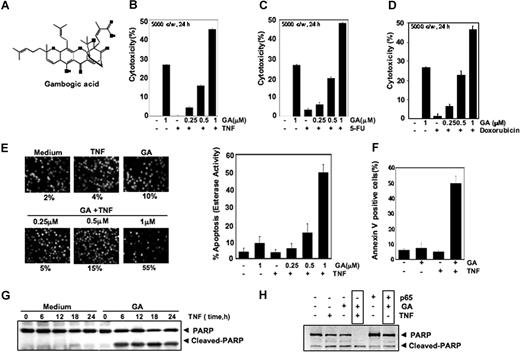
GA potentiates apoptotic effects of TNF and chemotherapeutic agents. (A) The chemical structure of GA. (B-D) GA enhances TNF-, 5-FU–, and doxorubicin-induced cytotoxicity. Five thousand cells were seeded in triplicate in 96-well plates. The cells were pretreated with the indicated concentrations of GA and then incubated with chemotherapeutic agents. Cell viability was then analyzed by the MTT method. (E) GA enhances TNF-induced cytotoxicity. KBM-5 cells were pretreated with indicated concentrations of GA for 4 hours and then incubated with 1 nM TNF for 24 hours. The cells were stained with a live/dead assay reagent for 30 minutes and then analyzed under a fluorescence microscope (left panel). The percentage of apoptosis was plotted as mean plus or minus SD (right panel). (F) GA enhances TNF-induced annexin V–FITC binding. (G) Effect of GA on PARP cleavage. Cells were pretreated with 1.0 μM GA for 4 hours and then incubated with 1 nM TNF for the indicated times. Whole-cell extracts were prepared and analyzed by Western blotting with an anti-PARP antibody. (H) A293 cells were transiently transfected with p65 plasmids. After 24 hours, cells were treated with 1.0 μM GA for 4 hours followed by 1 nM TNF for 24 hours. Whole-cell extracts were prepared and analyzed by Western blotting with an anti-PARP antibody.
GA potentiates apoptotic effects of TNF and chemotherapeutic agents. (A) The chemical structure of GA. (B-D) GA enhances TNF-, 5-FU–, and doxorubicin-induced cytotoxicity. Five thousand cells were seeded in triplicate in 96-well plates. The cells were pretreated with the indicated concentrations of GA and then incubated with chemotherapeutic agents. Cell viability was then analyzed by the MTT method. (E) GA enhances TNF-induced cytotoxicity. KBM-5 cells were pretreated with indicated concentrations of GA for 4 hours and then incubated with 1 nM TNF for 24 hours. The cells were stained with a live/dead assay reagent for 30 minutes and then analyzed under a fluorescence microscope (left panel). The percentage of apoptosis was plotted as mean plus or minus SD (right panel). (F) GA enhances TNF-induced annexin V–FITC binding. (G) Effect of GA on PARP cleavage. Cells were pretreated with 1.0 μM GA for 4 hours and then incubated with 1 nM TNF for the indicated times. Whole-cell extracts were prepared and analyzed by Western blotting with an anti-PARP antibody. (H) A293 cells were transiently transfected with p65 plasmids. After 24 hours, cells were treated with 1.0 μM GA for 4 hours followed by 1 nM TNF for 24 hours. Whole-cell extracts were prepared and analyzed by Western blotting with an anti-PARP antibody.
Cell lines
KBM-5 (human myeloid leukemia) and A293 (human embryonic kidney) were obtained from American Type Culture Collection (Manassas, VA). The H1299 cells were cultured in RPMI 1640 medium, the KBM-5 cells were cultured in Iscove modified Dulbecco medium with 15% FBS, and the A293 cells were cultured in Dulbecco modified Eagle medium supplemented with 10% FBS. For most studies, we used human myeloid KBM-5 cells because they express both types of TNF receptors. All culture media were also supplemented with 100 U/mL penicillin and 100 μg/mL streptomycin.
Electrophoretic mobility shift assay
To determine NF-κB activation, we prepared nuclear extracts and performed an electrophoretic mobility shift assay (EMSA) as described previously.16 For supershift assays, nuclear extracts prepared from TNF-treated cells were incubated with antibodies against either p50 or p65 of NF-κB for 30 minutes at 37°C before the complex was analyzed by EMSA. Preimmune serum was included as the negative control. The dried gels were visualized, and the radioactive bands quantitated with Storm 820 and Imagequant software (Amersham, Piscataway, NJ).
Western blot analysis
To determine the effect of GA on TNF-dependent IκBα phosphorylation, IκBα degradation, p65 translocation, and p65 phosphorylation, cytoplasmic or nuclear extracts were prepared as described previously.17 To detect the cleavage products of PARP and any antiapoptotic and angiogenesis markers, whole-cell extracts were prepared by subjecting GA-treated cells to lyses in lyses buffer (20 mM Tris [pH 7.4], 250 mM NaCl, 2 mM EDTA [pH 8.0], 0.1% Triton X-100, 0.01 μg/mL aprotinin, 0.005 μg/mL leupeptin, 0.4 mM phenylmethylsulfonyl fluoride, and 4 mM NaVO4). Lysates were spun at 20 817g for 10 minutes to remove insoluble material. Supernatant was collected and kept at −80°C. Either cytosolic or nuclear extract or lysates were resolved by sodium dodecyl sulfate–polyacrylamide gel electrophoresis (SDS-PAGE). After electrophoresis, the proteins were electrotransferred to nitrocellulose membranes, blotted with the relevant antibody, and detected by enhanced chemiluminescence reagent (Amersham ECL, Amersham, United Kingdom). The bands obtained were quantitated using NIH Image (National Institutes of Health, Bethesda, MD).
IKK assay
To determine the effect of GA on TNF-induced IKK activation, an IKK assay was performed by a method described previously.18 Briefly, the IKK complex from whole-cell extracts was precipitated with antibody against IKK-α and then treated with protein A/G-agarose beads (Pierce, Rockford, IL). After 2 hours, the beads were washed with whole-cell extract buffer and then resuspended in a kinase assay mixture containing 50 mM HEPES (pH 7.4), 20 mM MgCl2, 2 mM dithiothreitol, 20 μCi (0.74 MBq) [-32P]ATP, 10 μM unlabeled ATP, and 2 μg substrate GST-IκBα (aa 1-54). After incubation at 30°C for 30 minutes, the reaction was terminated by boiling with SDS sample buffer for 5 minutes. Finally, the protein was resolved on 10% SDS-PAGE, the gel was dried, and the radioactive bands were visualized with a Storm 820 imaging system (Amersham). To determine the total amounts of IKK-α and IKK-β in each sample, 30 μg whole-cell proteins were resolved on 10% SDS-PAGE, electrotransferred to a nitrocellulose membrane, and then blotted with either anti–IKK-α or anti–IKK-β antibody.
NF-κB–dependent reporter gene expression assay
To determine the effect of GA on TNF-, TNF receptor (TNFR)–, TNFR-associated death domain (TRADD)–, TRAF2-, NF-κB-inducing kinase (NIK)–, TAK1/TAB1-, and IKK–NF-κB–dependent reporter gene transcription, we performed the secretory alkaline phosphatase (SEAP) assay as previously described19 with the following exceptions. Briefly, A293 cells (5 × 105 cells/well) were plated in 6-well plates and transiently transfected by the calcium phosphate method with pNF-κB-SEAP (0.5 μg). To examine TNF-induced reporter gene expression, we transfected the cells with 0.5 μg SEAP expression plasmid and 1.5 μg control plasmid pCMV-FLAG1 DNA for 24 hours. We then treated the cells with butein for 4 hours and stimulated them with 0.1 nM TNF. The cell culture medium was harvested after 24 hours of TNF treatment. To examine reporter gene expression induced by various genes, A293 cells were transfected with 0.5 μg pNF-κB-SEAP plasmid with 0.5 μg expressing plasmid and 1.5 μg control plasmid pCMV-FLAG1 for 24 hours, treated with butein, and then harvested from cell culture medium after an additional 24 hours of incubation. The culture medium was analyzed for SEAP activity as recommended by the manufacturer (Clontech Laboratories, Mountain View, CA) with a Victor 3 microplate reader (Perkin-Elmer Life and Analytical Sciences, Boston, MA).
Immunocytochemical analysis for NF-κB p65 localization
Immunocytochemical analysis was performed to examine the effect of GA on the nuclear translocation of p65 as previously described.20 Briefly, treated cells were plated on a poly-l-lysine–coated glass slide with a Cytospin 4 centrifuge (ThermoShendon, Pittsburgh, PA), air dried, and fixed with 4% paraformaldehyde. After being washed in phosphate-buffered saline (PBS), the slides were blocked with 5% normal goat serum for 1 hour and then incubated with rabbit polyclonal p65 Ab at a 1:200 dilution. After overnight incubation at 4°C, the slides were washed, incubated with goat anti–rabbit IgG-Alexa Fluor 594 (Invitrogen) at a 1:200 dilution for 1 hour, and counterstained for nuclei with Hoechst 33342 (50 ng/mL) stain for 5 minutes. Stained slides were mounted with mounting medium purchased from Sigma-Aldrich and analyzed under a Labophot-2 fluorescence microscope (Nikon, Melville, NY). Pictures were captured with a Photometrics Coolsnap CF color camera (Nikon) and MetaMorph version 4.6.5 software (Universal Imaging, Sunnyvale, CA).
Live/dead assay
To measure apoptosis, we used the live/dead assay (Molecular Probes, Eugene, OR), which determines intracellular esterase activity and plasma membrane integrity.21 Calcein-AM, a nonfluorescent polyanionic dye, is retained by live cells, in which it produces intense green fluorescence through enzymatic (esterase) conversion. In addition, the ethidium homodimer enters cells with damaged membranes and binds to nucleic acids, thereby producing a bright red fluorescence in dead cells. Briefly, 2 × 105 cells were incubated with 2.5 μM GA for 4 hours and treated with 1 nM TNF up to 24 hours at 37°C. Cells were stained with the live/dead reagent (5 μM ethidium homodimer and 5 μM calcein-AM) and incubated at 37°C for 30 minutes. Cells were analyzed under a Labophot-2 fluorescence microscope (Nikon).
Cytotoxicity assay
The effects of GA on the cytotoxic effects of TNF and other chemotherapeutic agents were determined by the 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyl-tetrazolium bromide (MTT) uptake method as previously described.20 Briefly, 5000 cells were incubated with GA in triplicate in a 96-well plate and then treated with 1 nM TNF, 0.1 μM 5-fluorouracil, and 0.1 μM doxorubicin for 24 hours at 37°C. An MTT solution was added to each well, and incubated for 2 hours at 37°C. An extraction buffer (20% SDS, 50% dimethylformamide) was added, and the cells were incubated overnight at 37°C. Then the optical density was measured at 570 nm using a 96-well MRX Revelation multiscanner (Dynex Technologies, Chantilly, VA).
Annexin V assay
An early indicator of apoptosis is the rapid translocation and accumulation of the membrane phospholipid phosphatidylserine from the cytoplasmic interface of the membrane to the extracellular surface. This loss of membrane asymmetry can be detected using the binding properties of annexin V. To identify apoptosis, we used an annexin V antibody, which was conjugated with fluorescein isothiocyanate fluorescence dye. Briefly, 5 × 105 cells were pretreated with GA (1.0 μM), treated with TNF for 24 hours at 37°C, and subjected to annexin V staining. The cells were washed in PBS, resuspended in 100 μL binding buffer containing a fluorescein isothiocyanate–conjugated anti–annexin V antibody, and then analyzed with a FACSCalibur flow cytometer (BD Biosciences, San Jose, CA).
Down-regulation of transferring receptor by short interfering RNA
Cells (2 × 105) were plated in each well of 6-well plates and allowed to adhere for 24 hours. On the day of transfection, 12 μL Hiperfect transfection reagent (Qiagen, Valencia, CA) was added to 50 nM short interfering RNA (siRNA) in a final volume of 100 μL culture medium. After 48 hours of transfection, cells were recovered and used for appropriate determinations.
Detection of transferrin receptor mRNA by RT-PCR
Total RNA was extracted using Trizol reagent (Invitrogen) according to the manufacturer's instructions. Total RNA (1 μg) was amplified by polymerase chain reactions (PCR) using oligonucleotides specific for exons 5 to 11 of the human TfR1 gene. The sequences of oligonucleotides were 5 s: 5′-CAG TGG CCA ATT TCT TCT CG-3′ and 11as: 5′-CTG CCT CTG GTG TAT CCT CAC A-3′.22 RNA was first converted to cDNA by Superscript reverse transcriptase and then amplified by Platinum Taq polymerase using Superscript One Step reverse-transcription–polymerase chain reaction (RT-PCR) kit (Invitrogen). The relative expression of TfR1 was analyzed using quantitative RT-PCR with GAPDH as an internal control. The RT-PCR reaction mixture contained 12.5 μL of 2 × reaction buffer, 1 μg each of RNA, 0.5 μL each of forward and reverse primers, and 1 μL of RT-Platinum Taq in a final volume of 25 μL. The reaction was performed at 50°C for 30 minutes, 94°C for 2 minutes, 95°C for 30 cycles of 15 seconds each, 54°C for 30 seconds, and 72°C for 45 seconds with extension at 72°C for 5 minutes. PCR products were run on 2% agarose gel and then stained with ethidium bromide. Stained bands were visualized under UV light and photographed.
Results
The purpose of this study was to investigate the effects of GA on the NF-κB activation pathway, NF-κB–regulated gene products, and NF-κB–mediated cellular responses. We focused primarily on TNF-induced cellular effects because the relationship between TNF and the NF-κB activation pathway is well understood.
GA potentiates apoptosis induced by TNF and chemotherapeutic agents
Because NF-κB activation has been shown to inhibit apoptosis induced by TNF and chemotherapeutic agents,23-25 we investigated whether GA affects TNF- and chemotherapeutic agent–induced apoptosis. Results of the MTT uptake method showed that GA increased cytotoxic effects dose dependently induced by TNF (Figure 1B), 5-fluorouracil (Figure 1C), and doxorubicin (Figure 1D). According to results of esterase staining (ie, the live/dead assay), GA dose dependently up-regulated TNF-induced apoptosis from 10% to 55% (Figure 1C). Annexin V staining showed that GA up-regulated TNF-induced apoptosis from 5% to 55% (Figure 1F). Caspase-mediated PARP cleavage likewise showed that GA enhanced the apoptotic effect of TNF (Figure 1G). These results together indicate that GA potentiates the apoptotic effects of both TNF and chemotherapeutic agents.
Overexpression of NF-κB (p65) inhibits the proapoptotic effects of GA
Whether NF-κB (p65) overexpression suppresses the effect of GA was investigated. To determine this, cells were transfected with p65 plasmid and then examined for apoptosis. Results in Figure 1H show that TNF-induced caspase-mediated PARP cleavage was enhanced by GA, but when cells were transfected with p65 plasmid, the effect of GA was abolished. These results indicate that GA potentiates the apoptotic effects of TNF most likely through suppression of NF-κB.
GA suppresses the expression of antiapoptotic gene products
Because NF-κB regulates the expression of the antiapoptotic proteins IAP1/2,26,27 Bcl-2,28 Bcl-xL,29 and TRAF1,30 we investigated whether GA could modulate TNF-induced expression of these antiapoptotic genes. We found that GA blocked TNF-induced expression of these antiapoptotic proteins in a time-dependent manner (Figure 2A).
GA inhibits TNF-induced expression of NF-κB–dependent antiapoptotic, proliferative, and metastatic proteins. (A) GA inhibits the expression of TNF-induced antiapoptotic proteins. KBM-5 cells were incubated with 1.0 μM GA for 4 hours and then treated with 1 nM TNF for the indicated times. Whole-cell extracts were prepared and analyzed by Western blotting with the indicated antibodies. (B,C) GA inhibits TNF-induced expression of c-Myc, cyclin D1, COX-2, ICAM-1, MMP-9, and VEGF. KBM-5 cells were incubated with 1.0 μM GA for 4 hours and then treated with 1 nM TNF for the indicated times. Whole-cell extracts were prepared and analyzed by Western blotting with the relevant antibodies.
GA inhibits TNF-induced expression of NF-κB–dependent antiapoptotic, proliferative, and metastatic proteins. (A) GA inhibits the expression of TNF-induced antiapoptotic proteins. KBM-5 cells were incubated with 1.0 μM GA for 4 hours and then treated with 1 nM TNF for the indicated times. Whole-cell extracts were prepared and analyzed by Western blotting with the indicated antibodies. (B,C) GA inhibits TNF-induced expression of c-Myc, cyclin D1, COX-2, ICAM-1, MMP-9, and VEGF. KBM-5 cells were incubated with 1.0 μM GA for 4 hours and then treated with 1 nM TNF for the indicated times. Whole-cell extracts were prepared and analyzed by Western blotting with the relevant antibodies.
GA suppresses the expression of NF-κB–dependent cell proliferation gene products
We also investigated whether GA can modulate NF-κB–regulated gene products involved in the proliferation of tumor cells. TNF has been shown to induce the expression of c-Myc,31 cyclin D1,32 and COX-2,33 all of which have NF-κB–binding sites in their promoters. Thus, we investigated whether GA inhibits the TNF-induced expression of these proteins. Untreated cells and those pretreated with GA were examined for TNF-induced expression of gene products by Western blot analysis with specific antibodies. GA abolished TNF-induced expression of COX-2, c-Myc, and cyclin D1 (Figure 2B).
GA suppresses the expression of TNF-induced NF-κB–dependent metastatic gene products
Whether GA can modulate NF-κB–regulated gene products involved in the metastasis of tumor cells was investigated. TNF has been shown to induce expression of intracellular adhesion molecule-1 (ICAM-1),34 VEGF,35 and MMP-9,36 all of which have NF-κB–binding sites in their promoters. We found that GA abolished TNF-induced expression of ICAM-1, MMP-9, and VEGF (Figure 2C).
GA inhibits NF-κB activation induced by carcinogens and inflammatory stimuli
Several carcinogens and inflammatory stimuli have been shown to activate NF-κB activation. TNF, CSC, okadaic acid, LPS, PMA, and H2O2 are well-known activators of NF-κB.14,37-41 Using DNA-binding assays, we examined the effect of GA on the activation of NF-κB by all these agents. All 6 agents activated NF-κB in human myeloid leukemia KBM-5 cells, and GA suppressed this activation (Figure 3A). These results suggest that GA acts at a step in the NF-κB activation pathway that is common to all 6 agents.
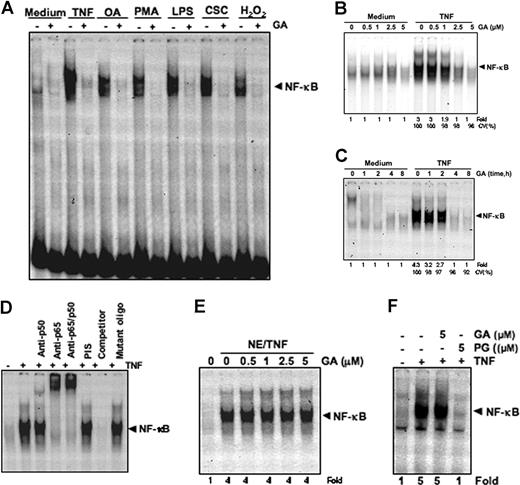
GA inhibits NF-κB activation induced by different stimuli. (A) GA blocks NF-κB activation induced by TNF, okadaic acid (OA), PMA, LPS, CSC, and H2O2. Human myeloid leukemia KBM-5 cells were preincubated with 5 μM GA for 4 hours and then treated with 0.1 nM TNF for 30 minutes, 500 nM OA for 4 hours, 25 ng/mL PMA for 2 hours, 10 μg/mL LPS, and 40 μg/mL CSC and 250 μM H2O2 for 1 hour each. Nuclear extracts were analyzed for NF-κB activation by EMSA. The results shown are representative of 3 independent experiments. (B) Dose-dependent effect of GA on TNF-induced NF-κB activation. KBM-5 cells were incubated with the indicated concentrations of GA for 4 hours and treated with 0.1 nM TNF for 30 minutes. The nuclear extracts were assayed for NF-κB activation by EMSA. CV (%) indicates cell viability of the cells. The results shown are representative of 3 independent experiments. (C) Time-dependent effect of GA on TNF-induced NF-κB activation. KBM-5 cells were preincubated with 5 μM GA for the indicated times and then treated with 0.1 nM TNF for 30 minutes. The nuclear extracts were prepared and assayed for NF-κB activation by EMSA. CV (%) indicates cell viability of the cells. The results shown are representative of 3 independent experiments. (D) NF-κB induced by TNF is composed of p65 and p50 subunits. Nuclear extracts from untreated cells or cells treated with 0.1 nM TNF were incubated with the indicated antibodies, an unlabeled NF-κB oligoprobe, or a mutant oligoprobe. They were then assayed for NF-κB activation by EMSA. (E) The direct effect of GA on the NF-κB–DNA binding. Nuclear extracts were prepared from untreated cells or cells treated with 0.1 nM TNF and incubated for 30 minutes with the indicated concentrations of GA. They were then assayed for NF-κB activation by EMSA. (F) Effect of plumbagin (PG) and GA on the binding of NF-κB to DNA. Nuclear extracts were prepared from untreated cells or cells treated with 0.1 nM TNF and incubated for 30 minutes with the 5-μM concentrations of GA and PG. They were then assayed for NF-κB activation by EMSA.
GA inhibits NF-κB activation induced by different stimuli. (A) GA blocks NF-κB activation induced by TNF, okadaic acid (OA), PMA, LPS, CSC, and H2O2. Human myeloid leukemia KBM-5 cells were preincubated with 5 μM GA for 4 hours and then treated with 0.1 nM TNF for 30 minutes, 500 nM OA for 4 hours, 25 ng/mL PMA for 2 hours, 10 μg/mL LPS, and 40 μg/mL CSC and 250 μM H2O2 for 1 hour each. Nuclear extracts were analyzed for NF-κB activation by EMSA. The results shown are representative of 3 independent experiments. (B) Dose-dependent effect of GA on TNF-induced NF-κB activation. KBM-5 cells were incubated with the indicated concentrations of GA for 4 hours and treated with 0.1 nM TNF for 30 minutes. The nuclear extracts were assayed for NF-κB activation by EMSA. CV (%) indicates cell viability of the cells. The results shown are representative of 3 independent experiments. (C) Time-dependent effect of GA on TNF-induced NF-κB activation. KBM-5 cells were preincubated with 5 μM GA for the indicated times and then treated with 0.1 nM TNF for 30 minutes. The nuclear extracts were prepared and assayed for NF-κB activation by EMSA. CV (%) indicates cell viability of the cells. The results shown are representative of 3 independent experiments. (D) NF-κB induced by TNF is composed of p65 and p50 subunits. Nuclear extracts from untreated cells or cells treated with 0.1 nM TNF were incubated with the indicated antibodies, an unlabeled NF-κB oligoprobe, or a mutant oligoprobe. They were then assayed for NF-κB activation by EMSA. (E) The direct effect of GA on the NF-κB–DNA binding. Nuclear extracts were prepared from untreated cells or cells treated with 0.1 nM TNF and incubated for 30 minutes with the indicated concentrations of GA. They were then assayed for NF-κB activation by EMSA. (F) Effect of plumbagin (PG) and GA on the binding of NF-κB to DNA. Nuclear extracts were prepared from untreated cells or cells treated with 0.1 nM TNF and incubated for 30 minutes with the 5-μM concentrations of GA and PG. They were then assayed for NF-κB activation by EMSA.
GA suppresses TNF-induced NF-κB activation
We first determined the dose and time of exposure to GA required to suppress TNF-induced NF-κB activation. EMSA showed that GA alone had no effect on NF-κB activation, but GA inhibited TNF-mediated NF-κB activation in a dose- and time-dependent manner (Figure 3B,C, respectively).
NF-κB is a complex of proteins in which various combinations of Rel or NF-κB proteins constitute active NF-κB heterodimers that bind specific DNA sequences. To show that the band visualized by EMSA in TNF-treated cells was indeed NF-κB, nuclear extracts from TNF-activated cells were incubated with antibodies to the p50 (NF-κB) and the p65 (RelA) subunit of NF-κB. The resulting bands were shifted to higher molecular masses (Figure 3D), suggesting that the TNF-activated complex consisted of p50 and p65. Preimmune serum (PIS) had no effect on DNA binding. Addition of excess unlabeled NF-κB (cold oligonucleotide, 100-fold) caused a complete disappearance of the band, whereas mutated oligonucleotide had no effect on the DNA binding.
GA does not directly affect binding of NF-κB to the DNA
Some NF-κB inhibitors, such as N-tosyl-l-phenylalanine chloromethyl ketone (a serine protease inhibitor), caffeic acid phenethyl ester, and plumbagin,42-45 directly modify NF-κB protein so that it can longer bind to DNA. Whether GA mediates suppression of NF-κB activation through a similar mechanism was investigated. We incubated nuclear extract from TNF-treated cells with GA and found that GA did not modify the DNA-binding ability of NF-κB proteins (Figure 3E). In comparison, plumbagin (PG) did modify the DNA-binding ability of p65 protein (Figure 3F). These results suggest that GA inhibits NF-κB activation by a mechanism different from that of plumbagin.
GA inhibits TNF-dependent IκBα degradation and phosphorylation
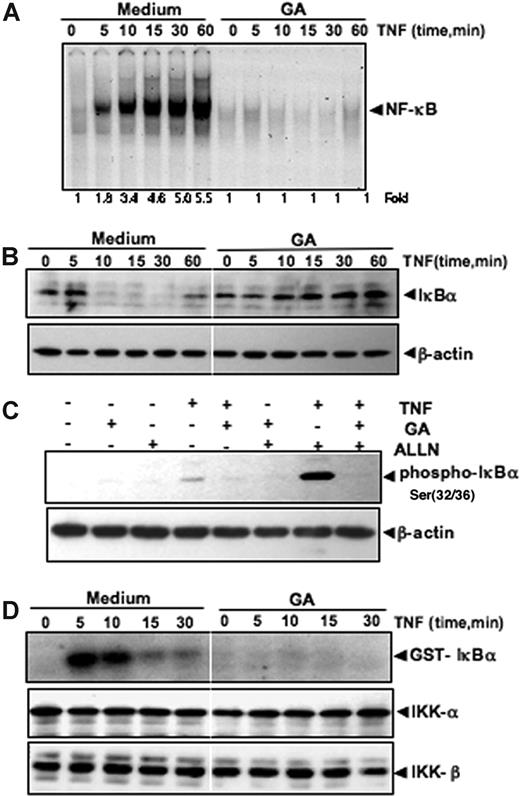
The translocation of NF-κB to the nucleus is preceded by the phosphorylation, ubiquitination, and proteolytic degradation of IκBα. To determine whether inhibition of TNF-induced NF-κB activation was due to inhibition of IκBα degradation, we pretreated cells with GA and then exposed them to TNF for various time periods. We then examined the cells for NF-κB in the nucleus by EMSA and for IκBα degradation in the cytoplasm by Western blot analysis. TNF activated NF-κB in the control cells in a time-dependent manner (Figure 4A). TNF induced NF-κB activation in the control cells in 10 minutes or fewer and peak activation at 30 minutes, but TNF had no effect on GA-pretreated cells. Moreover, TNF induced IκBα degradation in the control cells in 10 minutes, but it had no effect on IκBα degradation in GA-pretreated cells (Figure 4B). These results indicate that GA inhibited both TNF-induced NF-κB activation and IκBα degradation.
GA inhibits TNF-induced IκBα degradation, IκBα phosphorylation, and IKK activation. (A) GA inhibits TNF-induced activation of NF-κB. KBM-5 cells were incubated with 5 μM GA for 4 hours, treated with 0.1 nM TNF for the indicated times, and then analyzed for NF-κB activation by EMSA. (B) Effect of GA on TNF-induced degradation of IκBα. KBM-5 cells were incubated with 5 μM GA for 4 hours and treated with 0.1 nM TNF for the indicated times. Cytoplasmic extracts were prepared and analyzed by Western blotting with antibodies against anti-IκBα. Equal protein loading was evaluated by β-actin. (C) Effect of GA on the phosphorylation by IκBα by TNF. Cells were preincubated with 5 μM GA for 4 hours, incubated with 50 μg/mL ALLN for 30 minutes, and then treated with 0.1 nM TNF for 10 minutes. Cytoplasmic extracts were fractionated and then subjected to Western blotting with phospho-specific IκBα antibody. The same membrane was reblotted with β-actin. (D) Effect of GA on the activation of IKK by TNF. KBM-5 cells were preincubated with 5 μM GA for 4 hours and then treated with 1 nM TNF for the indicated times. Whole-cell extracts were immunoprecipitated with antibody against IKK-α and analyzed by an immune complex kinase assay. To examine the effect of GA on the level of expression of IKK proteins, whole-cell extracts were fractionated on SDS-PAGE and examined by Western blot analysis with anti–IKK-α and anti–IKK-β antibodies.
GA inhibits TNF-induced IκBα degradation, IκBα phosphorylation, and IKK activation. (A) GA inhibits TNF-induced activation of NF-κB. KBM-5 cells were incubated with 5 μM GA for 4 hours, treated with 0.1 nM TNF for the indicated times, and then analyzed for NF-κB activation by EMSA. (B) Effect of GA on TNF-induced degradation of IκBα. KBM-5 cells were incubated with 5 μM GA for 4 hours and treated with 0.1 nM TNF for the indicated times. Cytoplasmic extracts were prepared and analyzed by Western blotting with antibodies against anti-IκBα. Equal protein loading was evaluated by β-actin. (C) Effect of GA on the phosphorylation by IκBα by TNF. Cells were preincubated with 5 μM GA for 4 hours, incubated with 50 μg/mL ALLN for 30 minutes, and then treated with 0.1 nM TNF for 10 minutes. Cytoplasmic extracts were fractionated and then subjected to Western blotting with phospho-specific IκBα antibody. The same membrane was reblotted with β-actin. (D) Effect of GA on the activation of IKK by TNF. KBM-5 cells were preincubated with 5 μM GA for 4 hours and then treated with 1 nM TNF for the indicated times. Whole-cell extracts were immunoprecipitated with antibody against IKK-α and analyzed by an immune complex kinase assay. To examine the effect of GA on the level of expression of IKK proteins, whole-cell extracts were fractionated on SDS-PAGE and examined by Western blot analysis with anti–IKK-α and anti–IKK-β antibodies.
To determine whether the inhibition of TNF-induced IκBα degradation was due to an inhibition of IκBα phosphorylation, we used the proteasome inhibitor N-acetyl-leucyl-leucyl-norleucinal (ALLN) to block degradation of IκBα. Western blotting with an antibody that recognizes the serine-phosphorylated (Ser 32/36) form of IκBα showed that TNF-induced IκBα phosphorylation was strongly suppressed by GA (Figure 4C).
GA inhibits TNF-induced IKK activation
Since GA inhibits the phosphorylation and degradation of IκBα, we tested the effect of GA on TNF-induced IKK activation, which is required for TNF-induced phosphorylation of IκBα. As shown in Figure 4D (upper panel), GA completely suppressed TNF-induced activation of IKK. Neither TNF nor GA had any effect on the expression of IKK-α or IKK-β proteins (Figure 4D bottom panels).
GA inhibits TNF-induced phosphorylation and nuclear translocation of p65
Suppression of IκBα degradation should inhibit the translocation of p65 to the nucleus. We investigated whether GA inhibits TNF-induced p65 nuclear translocation. An immunocytochemistry assay confirmed that GA suppressed translocation of p65 to the nucleus (Figure 5A).
GA inhibits TNF-induced phosphorylation and nuclear translocation of p65. (A) Immunocytochemical analysis of p65 localization. KBM-5 cells were first treated with 5 μM GA for 4 hours at 37°C and then exposed to 0.1 nM TNF for 15 minutes. After the cells were centrifuged, they underwent immunocytochemical analysis. (B,C) GA inhibits TNF-induced nuclear translocation and phosphorylation of p65. KBM-5 cells were either untreated or pretreated with 5 μM GA for 4 hours at 37°C and then treated with 0.1 nM TNF for the indicated times. Nuclear extracts were prepared and analyzed by Western blotting with antibodies against p65 and phospho-specific p65. For loading control of nuclear protein, the membrane was blotted with anti-PARP antibody.
GA inhibits TNF-induced phosphorylation and nuclear translocation of p65. (A) Immunocytochemical analysis of p65 localization. KBM-5 cells were first treated with 5 μM GA for 4 hours at 37°C and then exposed to 0.1 nM TNF for 15 minutes. After the cells were centrifuged, they underwent immunocytochemical analysis. (B,C) GA inhibits TNF-induced nuclear translocation and phosphorylation of p65. KBM-5 cells were either untreated or pretreated with 5 μM GA for 4 hours at 37°C and then treated with 0.1 nM TNF for the indicated times. Nuclear extracts were prepared and analyzed by Western blotting with antibodies against p65 and phospho-specific p65. For loading control of nuclear protein, the membrane was blotted with anti-PARP antibody.
We also determined the effect of GA on TNF-induced nuclear translocation of p65 by Western blot analysis. TNF induced the translocation in a time-dependent manner, and GA suppressed it (Figure 5B). We also investigated the effect of GA on TNF-induced phosphorylation of p65 because phosphorylation is also required for its transcriptional activity.11 In the nuclear fraction from the TNF-treated cells, there was a time-dependent increase in the phosphorylated form of p65 (Ser 536), and GA suppressed it (Figure 5C upper panel).
GA represses TNF-induced NF-κB–dependent reporter gene expression
Although we observed by EMSA that GA blocked NF-κB activation, DNA binding alone does not always correlate with NF-κB–dependent gene transcription, suggesting there are additional regulatory steps.46 To determine the effect of GA on TNF-induced NF-κB–dependent reporter gene expression, we transiently transfected GA-pretreated or untreated cells with the NF-κB–regulated SEAP reporter construct and then stimulated the cells with TNF. An almost 14-fold increase in SEAP activity over the vector control was noted after stimulation with TNF (Figure 6A). Dominant-negative (DN) IκBα abolished most of the TNF-induced SEAP activity. When the cells were pretreated with GA, TNF-induced NF-κB–dependent SEAP expression was inhibited by 43% and 72% at GA concentrations of 1 μM and 5 μM, respectively. These results proved that GA inhibits NF-κB–dependent reporter gene expression induced by TNF.
GA inhibits NF-κB activation induced by different molecules in NF-κB signaling pathway. (A) GA inhibited TNF-induced NF-κB–dependent reporter gene (SEAP) expression. A293 cells treated with the indicated concentrations of GA were transiently transfected with a NF-κB–containing plasmid linked to the SEAP gene. After 24 hours in culture with 0.1 nM TNF, cell supernatants were collected and assayed for SEAP activity. Results are expressed as fold activity over the activity of the vector control. (B,C) GA inhibited NF-κB–dependent reporter gene expression induced by TNF, TNFR1, TRADD, NIK, TRAF2, TAK1/TAB1, IKK, p65, and receptor-interacting protein (RIP). A293 cells were transiently transfected with the indicated plasmids along with a NF-κB–containing plasmid linked to the SEAP gene. Bars indicate standard deviation.
GA inhibits NF-κB activation induced by different molecules in NF-κB signaling pathway. (A) GA inhibited TNF-induced NF-κB–dependent reporter gene (SEAP) expression. A293 cells treated with the indicated concentrations of GA were transiently transfected with a NF-κB–containing plasmid linked to the SEAP gene. After 24 hours in culture with 0.1 nM TNF, cell supernatants were collected and assayed for SEAP activity. Results are expressed as fold activity over the activity of the vector control. (B,C) GA inhibited NF-κB–dependent reporter gene expression induced by TNF, TNFR1, TRADD, NIK, TRAF2, TAK1/TAB1, IKK, p65, and receptor-interacting protein (RIP). A293 cells were transiently transfected with the indicated plasmids along with a NF-κB–containing plasmid linked to the SEAP gene. Bars indicate standard deviation.
TNF-induced NF-κB activation is mediated through sequential interaction of the TNFR with TRADD, TRAF2, NIK, and IKK-β, resulting in phosphorylation of IκBα and subsequent degradation of IκBα and p65 nuclear translocation.47,48 Moreover, recent studies indicate that TAK1 plays a major role in the canonical pathway through its interaction with TAB1 and TAB2.49 To delineate the site of action of GA in the TNF-signaling pathway leading to NF-κB activation, we transfected cells with TNFR1, TRADD, NIK, TRAF2, TAK1/TAB1, IKK, and p65 plasmids. We then monitored NF-κB–dependent SEAP expression in untreated and GA-treated cells. TNFR1, TRADD, TRAF2, NIK, TAK1/TAB1, IKK, and p65 plasmids induced gene expression, and GA suppressed reporter gene expression induced by these plasmids except p65 (Figure 6B). Because IKK activation can cause the phosphorylation of IκBα and p65,50 we suggest that GA inhibits NF-κB activation through inhibition of IKK.
RIP is a serine/threonine kinase that plays an important role in TNF-induced NF-κB activation51,52 ; RIP is ubiquitinated following TNF stimulation, which has been reported to play an important role in NF-κB activation.53 Therefore, we investigated whether GA suppresses TNF-induced NF-κB activation by inhibiting RIP. RIP activated NF-κB reporter gene activity, and GA suppressed its activation in a dose-dependent manner (Figure 6C).
Down-regulation of TfR1 reverses the effect of GA
A recent report suggests that GA mediates its apoptotic effects through its interaction with the TfR1.10 Therefore, we investigated whether depletion of TfR1 affects the activity of GA. The results in Figure 7 show that siRNA but not scrambled control RNA down-regulated the mRNA for TfR1 (Figure 7A left panel), down-regulated the TfR1 protein expression (Figure 7A right panel), reversed the effect of GA on TNF-induced NF-κB activation (Figure 7B), and abolished the effect of GA on TNF-induced apoptosis (Figure 7C). Thus these results clearly demonstrate that TfR1s are needed for the effect of GA.
The effect of GA on NF-κB and on apoptosis are mediated through the transferrin receptor. (A) Down-regulation of TfR1 by RNA interference reverses the effect of GA. A293 cells were transfected with TfR1 si RNA or scrambled (SC) control. After 48 hours, cells were harvested and used for RNA isolation and for protein extraction. RT-PCR was done to determine the TfR1 mRNA expression. RNA levels were normalized to glyceraldehyde-3-phosphate dehydrogenase (GAPDH). Whole-cell extracts were analyzed by Western blotting with an anti-TfR1 antibody. (B) Transfected cells were preincubated with 5 μM GA for 4 hours and then treated with 0.1 nM TNF for 30 minutes. The nuclear extracts were prepared and assayed for NF-κB activation by EMSA. (C) Transfected cells were treated with 1 μM GA for 4 hours followed by 1 nM TNF for 24 hours. The cells were stained with a live/dead assay reagent and analyzed under a fluorescence microscope.
The effect of GA on NF-κB and on apoptosis are mediated through the transferrin receptor. (A) Down-regulation of TfR1 by RNA interference reverses the effect of GA. A293 cells were transfected with TfR1 si RNA or scrambled (SC) control. After 48 hours, cells were harvested and used for RNA isolation and for protein extraction. RT-PCR was done to determine the TfR1 mRNA expression. RNA levels were normalized to glyceraldehyde-3-phosphate dehydrogenase (GAPDH). Whole-cell extracts were analyzed by Western blotting with an anti-TfR1 antibody. (B) Transfected cells were preincubated with 5 μM GA for 4 hours and then treated with 0.1 nM TNF for 30 minutes. The nuclear extracts were prepared and assayed for NF-κB activation by EMSA. (C) Transfected cells were treated with 1 μM GA for 4 hours followed by 1 nM TNF for 24 hours. The cells were stained with a live/dead assay reagent and analyzed under a fluorescence microscope.
Discussion
The goal of this study was to determine whether the anticarcinogenic effects of GA were mediated through modulation of NF-κB pathway. Our results strongly support the hypothesis that GA suppresses NF-κB activation induced by various carcinogens and inflammatory agents. NF-κB inhibition was caused by inhibition of IKK activation and the subsequent suppression of phosphorylation and degradation of IκBα, suppression of p65 phosphorylation, and suppression of NF-κB–dependent reporter gene expression. GA also down-regulated NF-κB–dependent gene products involved in cell proliferation, antiapoptosis, and invasion. This down-regulation led to the potentiation of apoptosis induced by cytokines and chemotherapeutic agents.
This is the first report to suggest that GA can suppress the NF-κB activation pathway. The GA-mediated inhibition of NF-κB activation induced by TNF, okadaic acid, PMA, LPS, CSC, and H2O2 suggests that GA acts at a step common to all these activators. NF-κB activation in response to most of these stimuli requires the activation of IKK. Our results suggest that GA abolishes NF-κB activation by other agents by suppressing IKK activation.
We also investigated how GA inhibits the activation of IKK. Several studies indicate that TAK1 plays a major role in TNF-induced NF-κB activation through its interaction with TAB1 and TAB2. For instance, TAK1 can bind and activate IKK, leading to NF-κB activation.54 TAK1 has also been shown to be recruited by TNFR1 through TRADD, TRAF2, and RIP.15 Our study is the first to show that GA inhibits TAK1-induced NF-κB activation. RIP also mediates the recruitment of TAK1 to the TNF receptor complex and thus plays a very important role in TNF-induced IKK activation and subsequent NF-κB activation.51,53 We found that GA inhibits RIP-induced NF-κB activation.
It has been shown that TNF activates NF-κB through sequential recruitment of TNFR1, TNFR2, NIK, and IKK.47 This is the first study to show that GA suppressed NF-κB activation by all of these plasmids. IKK has been implicated in the phosphorylation of p65,50 which is necessary for p65 transcriptional activity.55 We found that GA suppressed the phosphorylation of p65. Furthermore, GA had no direct effect on the binding of p50-p65 to the DNA.
We also found that GA suppressed TNF-induced expression of cyclin D1, COX-2, and c-Myc, all linked to cell proliferation. Thus, it is possible that suppression of proliferation of human hepatoma cells,3 breast cancer cells,4 gastric carcinoma cells,5-8 and lung carcinoma cells9 by GA is linked to the suppression of the above-listed gene products. Our results are in agreement with a previous report that GA can down-regulate c-Myc expression.8 In addition, we found that GA suppressed the expression of various antiapoptotic gene products, including TRAF1, Bcl-2, Bcl-xL, IAP-1, and IAP-2. The down-regulation of Bcl-2 expression in human gastric carcinoma reported previously6 is in agreement with our results.
Studies of Kasibhatla et al suggest that GA mediates its apoptotic effects through its interaction with the TfR1.10 Our studies showed that GA had no effect on NF-κB activation or on apoptosis, when TfR1s were down-regulated by RNA interference. This down-regulation of TfR1 also suppressed the apoptotic potential of GA. How interaction of GA with the TfR1 modulates TNF signaling is not clear. That TNF could up-regulate TfR1 expression has been shown.56
Since hTERT is also regulated by NF-κB,57 it is possible that hTERT is down-regulated3,8 by the down-regulation of NF-κB. Our results also suggest that GA induced down-regulation of ICAM-1, MMP-9, and VEGF by down-regulating NF-κB, thus suggesting GA has antimetastatic potential. We also clearly provided evidence supporting the ability of GA to potentiate apoptosis induced by TNF, 5-flourouracil (5-FU), and doxorubicin, with effects similar to those reported in tests of a specific inhibitor of NF-κB.21 Overall, our results provided the molecular basis for the antiproliferative and anti-inflammatory effects of GA. Further preclinical and clinical trials are required to determine the full potential of this important constituent of ancient medicine.
The publication costs of this article were defrayed in part by page charge payment. Therefore, and solely to indicate this fact, this article is hereby marked “advertisement” in accordance with 18 USC section 1734.
Acknowledgments
This work was supported by a grant from the Clayton Foundation for Research (B.B.A.), NIH grant CA91844 on lung chemoprevention (B.B.A.), and NIH grant P50CA97007 (B.B.A). Flow cytometric analysis work was supported by NIH CORE grant CA016672–32. B.B.A. is a Ransom Horne Jr Professor of Cancer Research.
We would like to thank Chris Yeager for carefully proofreading the paper and providing valuable comments.
National Institutes of Health
Authorship
Contribution: M.K.P., B.S., K.S.A., and A.B.K. conducted all the experiments; B.B.A. and M.M.C. supervised and wrote the paper.
Conflict-of-interest disclosure: The authors declare no competing financial interests.
Correspondence: Bharat B. Aggarwal, Cytokine Research Laboratory, Department of Experimental Therapeutics, Unit 143, University of Texas M. D. Anderson Cancer Center, 1515 Holcombe Blvd, Houston, TX 77030; e-mail:aggarwal@mdanderson.org.





This feature is available to Subscribers Only
Sign In or Create an Account Close Modal