Clinical outcome of mantle cell lymphoma (MCL) is highly heterogeneous. Tumor cell proliferation as assessed by the Ki-67 index has been shown to yield prognostic information on MCL in many studies using heterogeneously treated patient cohorts. The prognostic value of the Ki-67 index in patients treated with anti-CD20 therapy has not been studied so far. We analyzed the Ki-67 index at primary diagnosis in 249 advanced-stage MCL patients treated within randomized trials. Ki-67 showed high prognostic relevance for overall survival (relative risk 1.27 for 10% higher Ki-67, P < .001), also independently from clinical prognostic factors. The 3 groups with different Ki-67 index of less than 10%, 10% to less than 30%, and 30% or more showed significantly different overall survival in patients treated with CHOP (P = .001) as well as in patients treated with CHOP in combination with anti-CD20 therapy (R-CHOP, P = .013). Thus, the Ki-67 index remains an important prognostic marker in the era of anti-CD20 therapy. The Euro-pean MCL study is registered at www.ClinicalTrials.gov as #NCT00016887.
Introduction
Mantle cell lymphoma (MCL) is an aggressive B-cell non-Hodgkin lymphoma (B-NHL) with a median overall survival (OS) of the patients of 3 to 5 years.1 Clinical outcome of this disease is highly heterogeneous.2 Since MCL patients often present at an advanced age and treatment strategies differ in terms of their potential side effects, several attempts have been made to identify high- and low-risk patients for a risk-adapted therapy.2,,–5 In contrast to many other B-NHLs, increased proliferation of the tumor cells has been shown to be distinctly correlated with shorter survival in MCL. Thus, the proliferation index as assessed by the percentage of Ki-67 positive cells represents an important prognostic marker in several studies.2,6,7 However, the studies published so far aiming at the prognostic value of Ki-67 analyzed patients with heterogeneous therapeutic regimens that were not treated within prospective randomized trials.2,6,7 The addition of anti-CD20 (rituximab) to the chemotherapy protocols improved the outcome and might have changed the risk-factor profile of diffuse large B-cell lymphomas (DLBCL). Thus, BCL2 expression, a marker for unfavorable outcome in DLBCL, might lose its predictive value in DLBCL patients treated with rituximab.8 The introduction of rituximab into the treatment protocols for MCL has improved the outcome substantially.9 However, to date it is uncertain, whether proliferation can predict outcome in MCL treated with rituximab. We studied the tumor-cell proliferation rate in 249 newly diagnosed patients with advanced-stage MCL treated within the randomized trials of the European MCL Network and the German Low Grade Lymphoma Study Group (GLSG; a list of the members of the study group is available as Document S1 on the Blood website; see the Supplemental Materials link at the top of the online article).
Methods
Patients
Local ethics committees of the participating centers approved the study protocol, and written informed consent was obtained from all patients in accordance with the Declaration of Helsinki. Patient specimens were collected from 3 randomized trials of the GLSG9,10 and the European MCL Network.11 One hundred sixteen patients in our cohort received CHOP therapy and 96 patients received CHOP in combination with rituximab (R-CHOP). An additional 37 patients received mitoxantrone, chlorambucil, and prednisone (MCP) without rituximab.
Ki-67 index
Specimens were analyzed in agreement with the local ethical guidelines as well as the approved study protocols. Staining for Ki-67 was done on lymph node biopsies using the antibody Mib-1 or Ki-S5 as described before.2 The proliferation index (percentage of Ki-67 positive lymphoma cells) was assessed by counting 1000 cells in at least 2 representative areas of the lymphoma and the average of both values was used for further analysis.
Statistical methods
The outcome parameter was OS calculated from the day of trial recruitment to death from any cause or the latest follow-up date. The prognostic relevance of Ki-67 as continuous parameter was tested by univariate Cox regression in the entire cohort and then adjusted for the MCC International Prognostic Index (MIPI) prognostic score12 using multiple Cox regression. If Ki-67 was identified as independent prognostic factor, 2 optimal cut-off points were searched maximizing the log-rank statistic for OS and defining 3 reasonably sized risk groups. The candidate cut-off points were 10%, 20%, 30%, and 40%. Using the identified cut-off points we then calculated Kaplan-Meier plots and performed log-rank tests for the subgroups of CHOP– and R-CHOP–treated patients separately. In addition, for each subcohort we adjusted the statistical significance of the categorized Ki-67 index for the MIPI prognostic score12 using the likelihood ratio test in multiple Cox regression.
Results and discussion
As a continuous parameter, Ki-67 showed strong prognostic relevance for OS with a relative risk (RR) of 1.27 for 10% higher Ki-67 (95% CI 1.15 to 1.39, P < .001) in the entire cohort. This was also true independently from the MIPI prognostic score12 (adjusted RR 1.20, 95% CI 1.09 to 1.33, P = .001). All combinations of 2 cut-off points yielded significantly different OS curves. Taking group size into account, the combination of 10% and 30% showed the highest statistical significance (log rank-χ2 32.70, P < .001) with 32%, 52%, and 16% of the patients having a Ki-67 index less than 10%, 10% to less than 30%, and 30% or more.
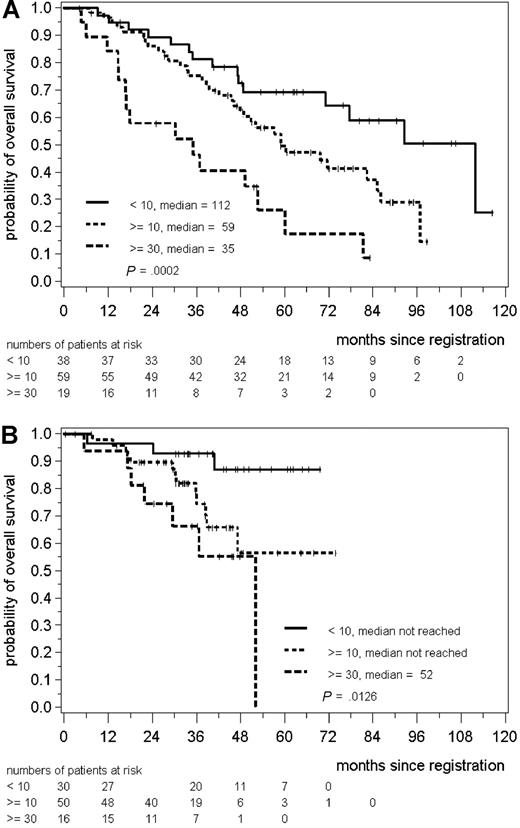
The statistically significant difference in OS between the groups of less than 10%, 10% to less than 30%, and 30% or higher Ki-67 index was also seen in the subgroups of CHOP– (median OS 112, 59, and 30 months, 3 years OS 81%, 75%, 46%, P = .001) and R-CHOP–treated patients (median OS not reached, not reached, 52 months, 3 years OS 93%, 74%, and 66%, P = .013 for R-CHOP, Figure 1). After adjusting for the MIPI score12 the P values were .003 for CHOP- and .14 for R-CHOP–treated patients. The median follow-up was 63 for CHOP– and 39 months for R-CHOP–treated patients, respectively. The follow-up in the R-CHOP–treated cohort was shorter because randomization between CHOP and R-CHOP was started in 2000,9 whereas in the preceding trial CHOP was randomly compared with MCP.10 However, the cohort of CHOP and R-CHOP patients did not differ in respect to clinical parameters like age, stage, lactate dehydrogenase, Eastern Cooperative Oncology Group Performance Status, or bone marrow involvement (data not shown).
Kaplan-Meier plot for overall survival of patients treated with CHOP (A) and R-CHOP (B) stratified in 3 groups according to the Ki-67 index of less than 10% (< 10), 10% to less than 30% (≥10), and 30% or more (≥30) Ki-67 positive cells.
Kaplan-Meier plot for overall survival of patients treated with CHOP (A) and R-CHOP (B) stratified in 3 groups according to the Ki-67 index of less than 10% (< 10), 10% to less than 30% (≥10), and 30% or more (≥30) Ki-67 positive cells.
We have recently shown that Ki-67 index shows strong prognostic relevance in combination with the clinical prognostic index for advanced stage MCL.12 The data presented herein indicate that the Ki-67 index might be a valuable prognostic parameter for MCL patients treated with immunochemotherapy including rituximab. However, compared with CHOP therapy, R-CHOP immunochemotherapy improves response and time to treatment failure of MCL but does not improve OS in our cohort (data not shown) and the randomized comparison.9 Therefore, the prognostic value of the Ki-67 index has to be validated in treatment protocols that improve OS of MCL patients.
In contrast to polymerase chain reaction– or array-based techniques for prediction of prognosis, the Ki-67 index can be cost-effectively evaluated on paraffin embedded tissue. The Ki-67 index thus might be the first biologic parameter that can be included for further risk stratification of MCL. However, standardized methods and guidelines to assess Ki-67 in MCL are needed for future use.
The online version of this article contains a data supplement.
The publication costs of this article were defrayed in part by page charge payment. Therefore, and solely to indicate this fact, this article is hereby marked “advertisement” in accordance with 18 USC section 1734.
Acknowledgment
This work was supported by the European Mantle Cell Lymphoma Network (No. LSHC-CT-2004-503351).
Authorship
Contribution: O.D., G.O., H.W.B., C.L., M.L.H., T.E.F.B., and W.K. generated data; E.H. and M.U. analyzed data; W.H. and M.D. provided data and administrative support; and W.K. and E.H. designed the study and wrote the manuscript.
A list of the participants in the German Low Grade Lymphoma Study Group and the European MCL Network can be found on the Blood website; see the Supplemental Materials link at the top of the online article.
Conflict-of-interest disclosure: The authors declare no competing financial interests.
Correspondence: Wolfram Klapper, Department of Pathology, Hematopathology Section, Niemannsweg 11, 24105 Kiel, Germany; e-mail: wklapper@path.uni-kiel.de.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal