In this issue of Blood, Rodríguez-Caballero and colleagues provide compelling evidence for the role of CMV in the pathogenesis of a specific subtype of LGL proliferations involving CD4+CD8−/+dim T cells. The authors used an elegant array of contemporary methodologies encompassing phenotypic, functional, and genotypic assays to investigate this enigmatic condition and have established a new paradigm for studying these types of disorders.
In a previous study, the authors established evidence for antigen-driven chronic T-cell stimulation and a potential role for cytomegalovirus (CMV) in the ontogeny of CD4+ large granular lymphocyte (LGL) lymphocytosis.1 Central to their new discoveries is the use of microarray gene expression profiling (GEP) to show that CD4+ T cells in patients with CD4+LGL expansions differ significantly from CMV specific memory CD4+ lymphocytes derived from healthy control individuals. The GEP abnormalities described cover a wide variety of genes involved in cell-cycle progression, resistance to apoptosis, and genetic instability. It is reasonable to conclude that abnormal expression of these genes gives a potential mechanism to explain the steady accumulation of the immunosenescent lymphocytes that are characteristic of CD4+LGL proliferations. Furthermore, it appears that a specific peptide, “MQLIPDDYSNTHSTRYVTVK” derived from CMV gB protein, can directly reproduce the anti-CMV response in vitro.
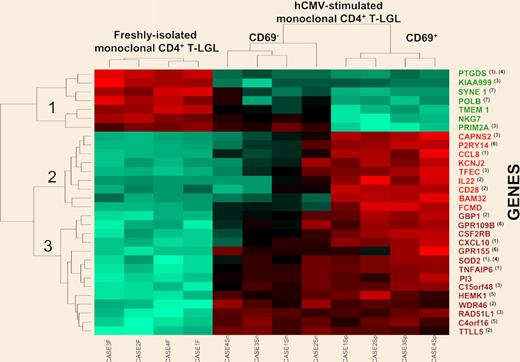
Hierarchical clustering analysis dendrogram of those 30 genes showing the highest differences between freshly isolated and both purified CD69+ and CD69− hCMV-stimulated monoclonal CD4+ T-LGL (n = 12 paired cell samples). The relative level of expression of each gene is represented by a color code where red represents an expression higher than the mean and green represents an expression lower than the mean values. Those genes showing different levels of expression in the distinct cell fractions analyzed are known to be involved in inflammatory (1) and immune (2) responses, cell proliferation and/or cell-cycle progression (3), apoptosis (4), protein synthesis (5), G-protein receptor-mediated cell signaling (6), and the DNA repair and maintenance machinery (7). Of note, both CD69+ and CD69− hCMV-stimulated monoclonal CD4+ T-LGLs showed a high similarity in their levels of expression for most of the genes displayed in the dendrogram, except for NKG7, PRIM2A, CAPNS2, P2RY14, CCL8, KCNJ2, TFEC, IL22, CD28, BAM32, and FCMD. See the complete figure in the article beginning on page 4609.
Hierarchical clustering analysis dendrogram of those 30 genes showing the highest differences between freshly isolated and both purified CD69+ and CD69− hCMV-stimulated monoclonal CD4+ T-LGL (n = 12 paired cell samples). The relative level of expression of each gene is represented by a color code where red represents an expression higher than the mean and green represents an expression lower than the mean values. Those genes showing different levels of expression in the distinct cell fractions analyzed are known to be involved in inflammatory (1) and immune (2) responses, cell proliferation and/or cell-cycle progression (3), apoptosis (4), protein synthesis (5), G-protein receptor-mediated cell signaling (6), and the DNA repair and maintenance machinery (7). Of note, both CD69+ and CD69− hCMV-stimulated monoclonal CD4+ T-LGLs showed a high similarity in their levels of expression for most of the genes displayed in the dendrogram, except for NKG7, PRIM2A, CAPNS2, P2RY14, CCL8, KCNJ2, TFEC, IL22, CD28, BAM32, and FCMD. See the complete figure in the article beginning on page 4609.
T-cell LGL proliferations are a heterogeneous group of disorders with a number of immunophenotypic subtypes characterized by clonal expansions of CD4−CD8+, CD4−CD8− or CD4+CD8−/+dim cells.2 The majority of patients follow a relatively benign clinical course and, although overt leukemic transformation or progression is well documented, it is a rare occurrence.2 The clonal CD8+ T-cell LGLs are typically associated with rheumatoid arthritis, neutropenia, and a CD16+CD56−CD57+ phenotype.3 The CD4+ LGL expansions are also typically benign and often associated with the presence of other neoplasia.4 Classification of these disorders is contentious and, though many cases are incidental findings, they are often classified as LGL leukemias in the absence of more appropriate terminology. Indeed, almost all of these cases lack any recognizable and consistent cytogenetic or molecular genetic abnormalities that are the hallmarks of classic hematologic neoplasms.
This current study has provided clear evidence that chronic antigenic stimulation of T cells by CMV can lead to persistent monoclonal expansions of vβ13.1/CD4+NKa+CD8dim+ lymphocytes in patients with an HLADRB1 0701 genotype. The deregulation of key genes involved in cell-cycle progression, resistance to apoptosis, and genetic instability allows these cells to accumulate in excess of what is required to control CMV infection. Furthermore, the clear association with HLA subtype shows the complex nature of interaction between virus and host immune system. Not only have molecular profiling studies of this kind begun to reveal similar insights into CD8+ LGL expansions, but they've also begun to challenge the current way we classify these conditions.5,6 Improving the understanding of what occurs at the molecular genetic level opens up options of developing therapy to control these abnormal proliferations of cells that are usually so effectively controlled as part of a normal immune response.
Conflict-of-interest disclosure: The author declares no competing financial interests. ■

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal