Abstract
Abstract 3899
Poster Board III-835
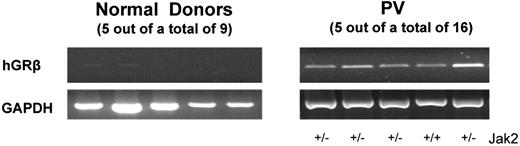
Glucocorticoids delay the maturation of erythroid cells retaining them in the proliferative phase. This effect is mediated by synergistic transcriptional and membrane-associated pathways. The glucocorticorticoid receptor (GR) upon activation forms a transcriptional complex with phosphorylated STAT-5 (P-STAT) which binds and alters the expression of a subset of the erythropoietin (EPO)- and stem cell factor (SCF)-target genes. In addition, the physical interaction between the GR and the EPO receptor (EPO-R) in the cytoplasm suppresses the ability of both receptors to phosphorylate STAT-5 when activated together. In addition to the canonical αGR signalling isoform, cells may express the dominant negative βGR isoform which antagonizes αGR activity. Erythroid cells (EBs) generated from peripheral blood mononuclear cells (PBMC) from patients with Polycythemia Vera (PV) and those obtained from normal donors stimulated with dexamethasone (D), an agonist of GR, and estradiol (E) in culture do not mature but continue proliferating. To investigate the molecular basis for this observation, cell number, differentiation status (by FACS and gene expression profiling), ability to phosphorylate STAT-5 and GR isoform expression of EBs generated after 11-13 days of cultures from PBMC obtained from normal donors or patients with PV stimulated with SCF, interleukin-3 and EPO, alone or in combination with DE, were monitored. Normal donors (15-20) and PV (16, 13 V617FJAK2 heterozygous and 3 V617FJAK2 homozygous, 7 at diagnosis and 11 1-180 months later) were obtained. In the absence of DE, normal PBMC generated cells belonging to multiple lineages, only ∼57% of which were represented by EBs at all stages of maturation. In the presence of DE, normal PBMC generated 2-5-times greater cell numbers the majority (>85%) of which were proEBs which expressed 10-fold lower levels of GATA1 and β-globin than that expressed by EBs generated in the absence of DE. PBMC from PV patients generated 102-103 more cells, mostly proEB, than that generated by normal PBMC, irrespective of the presence of DE. PV EBs generated in the presence or absence of DE had a V617FJAK2 allele burden of ∼38% (irrespectively of the allele burden of primary granulocytes), and expressed similar levels of GATA1, GATA2, NFE2, WT1 and β-globin. The levels of these mRNA were not statistically different from those expressed by normal EBs generated in the presence of DE. When stimulated with D and EPO, normal EBs failed to phosphorylate and translocate to the nucleus STAT-5. Therefore, normal EBs failed to mature due to D inactivating EPO signalling. By contrast, STAT-5 phosphorylation and nuclear translocation were constitutive in EBs generated by PV PBMC and EPO and D were either ineffective or actually decreased STAT-5-phosphorylation. To clarify this apparent contradiction, the GR isoforms expressed by normal and PV EBs were compared. Normal EBs expressed only αGR while PV EBs expressed both αGR and βGR (see Figure). Therefore, in PV EBs, although STAT-5 is constitutively phosphorylated it may be incapable of inducing erythroid maturation because it is associated with a transcriptionally inactive complex by βGR. These results suggest that activation of the expression of the dominant negative βGR isoform may contribute to the pathogenesis of PV by leading to an expansion of the EB compartment which eventually contributes to development of erythrocytosis.
No relevant conflicts of interest to declare.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal