Abstract
Abstract 1768
Although advances in therapy have improved outcomes for patients (pts) with FL, no optimal patient management strategy for pts with FL grade 3 has been identified largely due to debate regarding the benefits of anthracyclines for this subtype.
The NLCS is a multi-center, longitudinal, observational study that collects data on treatment and outcomes for pts with FL diagnosed at 265 community (80%) and academic practices in the United States from 2004–2007. Pts enrolled in NLCS with a diagnosis of FL grade 3 as recorded by the treating physicians were examined to compare patterns of presentation, treatment strategies, and outcomes. No central pathology review was required. Data from pts who received watchful waiting (WW), single agent rituximab (R), R with cyclophosphamide, adriamycin, vincristine, and prednisone (R-CHOP), or R with cyclophosphamide, vincristine, and prednisone (R-CVP) as initial therapy were summarized using median and range for continuous variables and frequencies for categorical variables. Univariate associations between demographic, baseline disease characteristics, treatment setting, and initial treatment strategy, were tested using a standard χ2 test where sample size allowed. Relationship and hazard ratios for progression free survival (PFS) were estimated using Cox regression models adjusted for FL International Prognostic Index (FLIPI).
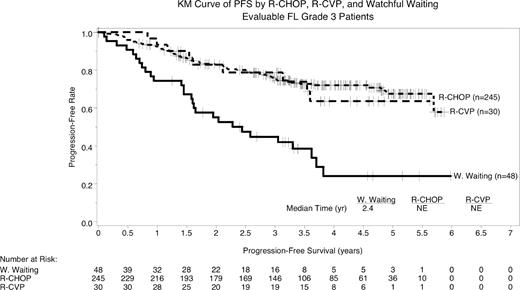
Of 2736 pts enrolled in NLCS, 500 had grade 3 FL. FL grade 3 pts had a median age of 64 years (range 26–97 years), 53% were female, and 67% had stage 3/4 disease. Pts were evenly distributed across FLIPI categories (30% good, 33% intermediate and 37% poor risk). Initial therapies for pts with FL grade 3 were WW (n=48), R alone (n=41), R-CVP (n=30), R-CHOP (n=245), R with another anthracycline (n=26), R with other chemotherapy (n=28), radiation alone (n=14), combined modality therapy (n=26), and regimens without R (n=42). Pts with FL grade 3 who received R-CHOP were younger than those who received R-CVP but similar in other FLIPI factors (Table). Events, defined as death or disease progression, occurred for 58% of patients who watched and waited and 33% of those who received treatment. Treated patients had improved PFS (median not reached [NR] 95% CI 66 months-NR) versus WW (median 29 months; 95% CI 19–43 months). Pts who received R-CHOP had improved PFS compared with WW (hazard ratio [HR] 3.09 95% CI 1.97– 4.86) but not R-CVP (HR 1.06 95% CI 0.52–2.14; Figure)
In this large observational dataset of FL grade 3 patients, R-CHOP was the most commonly used first-line regimen. These results suggest that R with chemotherapy provides meaningful PFS benefits over WW for this population. Prospective randomized trials in pathologically identified pts are needed to evaluate the benefit of R-CHOP versus non-anthracycline regimens.
| . | R-CHOP . | R-CVP . | R alone . | WW . |
|---|---|---|---|---|
| . | n=245 . | n=30 . | n=41 . | n=48 . |
| Age ≥60 yrs | 130/245 (53%) | 20/30 (67%) | 30/41 (73%) | 34/48 (71%) |
| Elevated LDH | 64/200 (32%) | 8/24 (33%) | 9/31 (29%) | 4/30 (13%) |
| >4 Nodal Sites | 89/240 (37%) | 11/30 (37%) | 12/40 (30%) | 9/44 (20%) |
| Hgb <12 | 56/234 (24%) | 8/29 (28%) | 11/39 (28%) | 9/39 (23%) |
| Stage 3/4 | 179/245 (73%) | 25/30 (83%) | 28/40 (70%) | 24/47 (51%) |
| . | R-CHOP . | R-CVP . | R alone . | WW . |
|---|---|---|---|---|
| . | n=245 . | n=30 . | n=41 . | n=48 . |
| Age ≥60 yrs | 130/245 (53%) | 20/30 (67%) | 30/41 (73%) | 34/48 (71%) |
| Elevated LDH | 64/200 (32%) | 8/24 (33%) | 9/31 (29%) | 4/30 (13%) |
| >4 Nodal Sites | 89/240 (37%) | 11/30 (37%) | 12/40 (30%) | 9/44 (20%) |
| Hgb <12 | 56/234 (24%) | 8/29 (28%) | 11/39 (28%) | 9/39 (23%) |
| Stage 3/4 | 179/245 (73%) | 25/30 (83%) | 28/40 (70%) | 24/47 (51%) |
Flowers:Millenium: Research Funding; Prescription Solutions: Consultancy; Genentech: Consultancy; Biogen/Idec: Consultancy; Celgene: Consultancy. Taylor:Genentech: Consultancy; Roche: Stock Ownership. Byrtek:Genetech: Consultancy; Roche: Stock Ownership. Hirata:Genentech: Employment; Roche: Stock Ownership. Cerhan:Genentech: Consultancy, Honoraria, Research Funding. Hainsworth:Genentech: Consultancy, Research Funding. Link:Genentech: Consultancy, Research Funding; Biogen Idec: Consultancy, Research Funding; Millennium Pharm: Consultancy, Research Funding; GlaxoSmithKline: Research Funding. Friedberg:Genentech: Consultancy.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal