Abstract
Abstract 2406
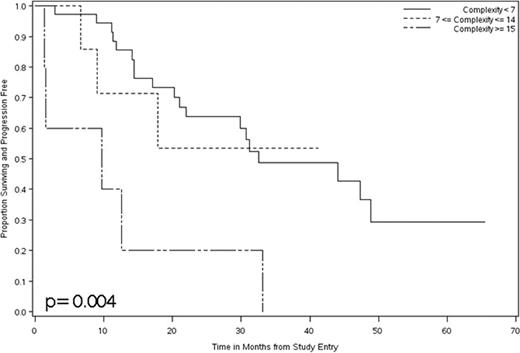
The outcome for a given CLL patient is difficult to predict. While there are promising models, they require collation of multiple clinical and laboratory parameters, and it remains to be seen whether they will apply to typical CLL patients in the community. To further dissect out explanations for this dramatic clinical heterogeneity, we sought to understand genomic complexity of clonal B-cells as a possible explanation of clinical variability with specific application to genomic complexity as a predictor of therapeutic response and clinical outcome in CLL. Thus we wished to identified global gains and losses of genetic material in order to define copy-number abnormalities (CNA) in 48 clinically progressive CLL patients who were about to be treated on a chemoimmunotherapy protocol. This protocol was previously reported by us (Blood. 109:2007) and had an induction phase with pentostatin (2 mg/m2), cyclophosphamide (600 mg/m2) and rituximab (375 mg/m2) given every 3 weeks for 6 cycles and then responding patients were followed ever three months until relapse. In order to estimate CNA, we employed array-based comparative genomic hybridization (aCGH) using a one-million oligonucleotide probe array format on the leukemic B-cells from the 48 patients entering this trial. In those same patients, the aCGH data were compared to a) FISH detecxtable data using a panel for the common recurring genetic defects seen in CLL and b) to their clinical outcome on this trial. With aCGH we found that 288 CNA were identified (median of 4 per patient; range 0–32) of which 215 were deletions and 73 were gains. The aCGH method identified most of the FISH detected abnormalities with a complete concordance for 17p13.1- deletion (17p-) between aCGH and FISH. We also identified chromosomal gain or loss in ≥6% of the patients on chromosomes 3, 8, 9, 10, 11, 12, 13, 14 and 17. We found that CLL patients with ≥15 CNA had a significantly worse progression free survival (PFS) than patients with <15 CNA (p=0.004)(figure). Patients with ≥15 CNA also had a shorter duration of response than those with <15 CNA (p=0.0726). Of interest, more complex genomic features were found both in patients with a 17p13.1 deletion and in more favorable genetic subtypes such as 13q14.1. Thus, for 5 patients with >15 CNAs the following FISH patterns were seen: +12/13q14.1-x1/13q14.1 -x2, 13q14.1 ×1 (n=2), and 17p13.1 (n=2). In addition, a 17p- by FISH was positively associated with the number of CNA and total deletion size. The odds of having an overall response decreased by 28% (95% CI: 5–55%; p=0.015) with each additional CNA for the 17p13.1- patients. In addition to defining genomic complexity as the total number of CNA for each patient, we also defined complexity as the sum of the lengths of all interstitial chromosomal gains and losses. When defined as the total size of chromosomal gains or losses, genomic complexity was significantly associated with 17p13.1 and worse overall clinical response. In summary, this analysis utilized the global assessment of copy number abnormalities using a high-resolution aCGH platform for clinically progressive CLL patients prior to initiation of their treatment. One outcome was that we found higher genomic complexity was associated with shorter progression-free survival, reduced duration of response and predicted a poor response to treatment. In addition since we did find genomic complexity in more traditionally favorable FISH categories, such as 13q14.1 type defects, this may explain why some of the latter patients do not fare as well as might be expected even with aggressive chemoimmunotherapy approaches. This study adds information on the association between inferior trial response and increasing genetic complexity as CLL progresses.
Off Label Use: Pentostatin. Kipps: GlaxoSmithKline: Consultancy, Membership on an entity's Board of Directors or advisory committees, Research Funding; Genentech: Research Funding; Celgene: Consultancy, Membership on an entity's Board of Directors or advisory committees; Celgene: Research Funding; Genzyme: Research Funding; Memgen: Research Funding; Igenica: Consultancy, Membership on an entity's Board of Directors or advisory committees; Sanofi Aventis: Research Funding; Abbott Laboratories: Research Funding.

This icon denotes an abstract that is clinically relevant.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal