Abstract
Hemolytic uremic syndrome (HUS) is commonly associated with Shiga toxin (Stx)–producing Escherichia coli O157:H7 infection. This study examined patient samples for complement activation on leukocyte-platelet complexes and microparticles, as well as donor samples for Stx and lipopolysaccharide (O157LPS)–induced complement activation on platelet-leukocyte complexes and microparticles. Results, analyzed by flow cytometry, showed that whole blood from a child with HUS had surface-bound C3 on 30% of platelet-monocyte complexes compared with 14% after recovery and 12% in pediatric controls. Plasma samples from 12 HUS patients were analyzed for the presence of microparticles derived from platelets, monocytes, and neutrophils. Acute-phase samples exhibited high levels of platelet microparticles and, to a lesser extent, monocyte microparticles, both bearing C3 and C9. Levels decreased significantly at recovery. Stx or O157LPS incubated with donor whole blood increased the population of platelet-monocyte and platelet-neutrophil complexes with surface-bound C3 and C9, an effect enhanced by costimulation with Stx and O157LPS. Both Stx and O157LPS induced the release of C3- and C9-bearing microparticles from platelets and monocytes. Released microparticles were phagocytosed by neutrophils. The presence of complement on platelet-leukocyte complexes and microparticles derived from these cells suggests a role in the inflammatory and thrombogenic events that occur during HUS.
Introduction
Typical hemolytic uremic syndrome (HUS) is associated with gastrointestinal infections caused by Shiga toxin (Stx)–producing bacteria such as enterohemorrhagic Escherichia coli (EHEC), of which E coli O157:H7 is the most common clinical isolate.1 HUS is characterized by microangiopathic hemolytic anemia, thrombocytopenia, and acute renal failure. The renal glomerular lesion, termed thrombotic microangiopathy, features microthrombi in the capillaries, leading to their occlusion and profound endothelial injury.2 Thrombocytopenia is presumably secondary to platelet consumption in microthrombi along the damaged endothelial wall.3,4
Gastrointestinal infection with EHEC may result in diarrhea or hemorrhagic colitis, followed in severe cases by HUS.1 Although the bacteria are noninvasive,5 virulence factors gain access to the circulation after passing the intestinal barrier and are therefore presumed to reach the kidney during HUS.6 Both Stx and E coli O157:H7 lipopolysaccharide (O157LPS) have been detected in the circulation bound to platelets, monocytes, and neutrophils.7,8
Stx is a bipartite molecule composed of a pentamer of B subunits that bind to the globotriaosylceramide (Gb3) receptor and an enzymatically active A subunit. The A subunit inhibits eukaryotic cell protein synthesis.9 Both Stx and O157LPS have been shown to activate platelets, monocytes, and neutrophils.7 O157LPS binds to and activates platelets directly via TLR-4 in complex with CD62 (P-selectin),10 and Stx binds to preactivated platelets.3,11 Binding of the B subunit of Stx1 (Stx1B) alone induces platelet activation.3
Stx and O157LPS induce the formation of platelet-leukocyte complexes in whole blood.7 These complexes form mainly between platelets and monocytes and express tissue factor, particularly under conditions of high shear stress. Tissue factor was also detected on microparticles released from these blood cells.7 These in vitro findings were confirmed in whole blood from HUS patients,7 and suggested that platelet-leukocyte complexes, and microparticles derived thereof, could contribute to the prothrombotic state occurring during HUS.
In the current study, we investigated whether complement was activated on blood cell complexes and on the surface of blood cell–derived microparticles in Stx-associated HUS. Activation of platelets, monocytes, and neutrophils is accompanied by complement deposition.12-14 Complement activation on the surface of platelets and leukocytes is followed by the formation of the membrane-attack complex (MAC) C5b-9.15,16 In addition to its lytic capacity, MAC may induce cell activation and proliferation, as described previously.17 Deposition of C5b-9 on platelets leads to the release of microparticles.18 Microparticles released from platelets and monocytes during complement deposition may contribute to a prothrombotic state when they are coated with tissue factor, as was previously shown in atypical HUS mediated by factor H mutations.15
The aim of the present study was to investigate whether complement is deposited on platelet-leukocyte complexes and microparticles derived from these cells during Stx-associated HUS, and if Stx and O157LPS can induce complement deposition, thus contributing to the inflammatory and prothrombotic mechanisms of this disease.
Methods
Subjects
Blood samples were available from 12 patients (Table 1) diagnosed with EHEC-associated HUS and treated at the Department of Pediatrics, Lund University Hospital. There were 8 boys and 4 girls ages 1-11 years (median age 3 years). Patient samples were taken within 3 days (median 4 hours) of admission while the children still had clinical signs of HUS, defined as hemolytic anemia (hemoglobin levels < 100 g/L), thrombocytopenia (platelet counts < 140 × 109/L), and acute renal failure. Blood was also obtained from the children 2-9 months after recovery. EHEC infection was detected as described previously.19 Blood samples were also available from 6 pediatric controls, 3 girls and 3 boys ages 1-15 years (median 9.5 years). These children were seen at the outpatient clinic of the Department of Pediatrics, Lund University Hospital, for unrelated conditions such as reflux nephropathy, immune deficiency, or metabolic disorders, and did not have a history of HUS or diarrhea at the time of sampling. Whole blood was available from 5 of these pediatric controls, and plasma was available from all 6. Blood samples were also obtained from 3 patients with acute renal failure without any history of HUS or diarrhea at the time of blood sampling. These patients were seen at the Department of Pediatrics, Lund University Hospital, for acute myeloid leukemia (one 13-year-old boy) or at the Department of Nephrology, Lund University Hospital, for acute renal failure associated with hypertension and sepsis (1 adult woman and 1 adult man). Blood was also obtained from 12 healthy adult volunteers (7 women and 5 men) not using any medications. Samples from patients, controls, and healthy donors were taken with the informed written consent of the subjects or their parents in accordance with the Declaration of Helsinki. The study was performed with the approval of the ethics committee of the Medical Faculty, Lund University.
Characteristics of HUS patients included in this study
| Patient . | Sex . | Age at diagnosis, y . | Prodromal diarrhea . | Laboratory findings . | Extrarenal symptoms . | E coli serotype§ . | ||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Hemolytic anemia,* hemoglobin (lowest), g/L . | Platelet count × 109 (lowest), ref 140-400 . | Renal failure . | Complement levels . | |||||||||
| Creatinine (highest), μmol/L† . | Urea (highest), mmol/L‡ . | Duration of peritoneal dialysis . | C3, g/L (ref (0.77-1.38) . | C4, g/L (ref (0.12-0.33) . | ||||||||
| 1 | M | 3 | +¶ | 62 | 21 | 103 | 15.6 | NA | NA | O157 | ||
| 2 | M | 3 | +¶ | 72 | 44 | 48 | 5.1 | NA | NA | O157 | ||
| 3 | M | 7 | + | 56 | 18 | 453 | 27.8 | 17 d | 1.11 | 0.16 | O157 | |
| 4 | M | 1 | + | 58 | 27 | 88 | 16.3 | 0.96 | 0.22 | O26 | ||
| 5 | F | 3 | +¶ | 63 | 35 | 286 | 21.1 | 21 d# | 0.79 | 0.05** | Rectal prolapse | O157 |
| 6 | F | 2 | + | 43 | 10 | 96 | 16.7 | 1.21 | 0.16 | Rectal prolapse | O157 | |
| 7 | M | 2 | +¶ | 63 | 31 | 174 | 17.3 | 1.01 | 0.18 | O145 | ||
| 8 | M | 10 | + | 49 | 35 | 468 | 37.5 | 10 d | 1.02 | 0.21 | O157 | |
| 9 | F | 2 | +¶ | 57 | < 10 | 64 | 10.9 | NA | NA | O157 | ||
| 10 | F | 11 | +¶ | 55 | 27 | 280 | 28.2 | 1.26 | 0.14 | O157 | ||
| 11 | M | 10 | +¶ | 41 | 30 | 379 | 28.3 | 15 d | 1.08 | 0.15 | Coma, hemiplegia, seizures | O157 |
| 12 | M | 5 | +¶ | 66 | < 10 | 188 | 20.6 | 4 d | 0.95 | 0.06** | O157 | |
| Patient . | Sex . | Age at diagnosis, y . | Prodromal diarrhea . | Laboratory findings . | Extrarenal symptoms . | E coli serotype§ . | ||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Hemolytic anemia,* hemoglobin (lowest), g/L . | Platelet count × 109 (lowest), ref 140-400 . | Renal failure . | Complement levels . | |||||||||
| Creatinine (highest), μmol/L† . | Urea (highest), mmol/L‡ . | Duration of peritoneal dialysis . | C3, g/L (ref (0.77-1.38) . | C4, g/L (ref (0.12-0.33) . | ||||||||
| 1 | M | 3 | +¶ | 62 | 21 | 103 | 15.6 | NA | NA | O157 | ||
| 2 | M | 3 | +¶ | 72 | 44 | 48 | 5.1 | NA | NA | O157 | ||
| 3 | M | 7 | + | 56 | 18 | 453 | 27.8 | 17 d | 1.11 | 0.16 | O157 | |
| 4 | M | 1 | + | 58 | 27 | 88 | 16.3 | 0.96 | 0.22 | O26 | ||
| 5 | F | 3 | +¶ | 63 | 35 | 286 | 21.1 | 21 d# | 0.79 | 0.05** | Rectal prolapse | O157 |
| 6 | F | 2 | + | 43 | 10 | 96 | 16.7 | 1.21 | 0.16 | Rectal prolapse | O157 | |
| 7 | M | 2 | +¶ | 63 | 31 | 174 | 17.3 | 1.01 | 0.18 | O145 | ||
| 8 | M | 10 | + | 49 | 35 | 468 | 37.5 | 10 d | 1.02 | 0.21 | O157 | |
| 9 | F | 2 | +¶ | 57 | < 10 | 64 | 10.9 | NA | NA | O157 | ||
| 10 | F | 11 | +¶ | 55 | 27 | 280 | 28.2 | 1.26 | 0.14 | O157 | ||
| 11 | M | 10 | +¶ | 41 | 30 | 379 | 28.3 | 15 d | 1.08 | 0.15 | Coma, hemiplegia, seizures | O157 |
| 12 | M | 5 | +¶ | 66 | < 10 | 188 | 20.6 | 4 d | 0.95 | 0.06** | O157 | |
NA indicates not available.
Hemolytic anemia was defined as hemoglobin levels < 100 g/L, elevated reticulocyte counts, elevated lactic dehydrogenase, and unconjugated bilirubin. All patients had a negative direct antiglobulin test.
Reference value < 5 years: 14-37μM; 5-15 years: 25-68μM.
Reference value < 5 years: 1.7-5.0mM; 5-16 years: 1.7-7.3mM.
All strains produced Stx2.
Bloody diarrhea.
Due to peritonitis also treated with hemodialysis.
C4 level normalized after 10 days.
Blood collection
Whole blood from patients and controls was drawn by venipuncture into Vacutainer tubes (Becton Dickinson) containing 0.5 mL of 0.129M sodium citrate and platelet-poor plasma obtained by centrifugation at 2600g for 15 minutes. To prevent the spontaneous formation of platelet-leukocyte complexes, whole blood used for in vitro stimulation experiments was drawn by venipuncture via a butterfly needle (Plasti Medical) into plastic tubes (Becton Dickinson) containing 0.27 mL of 0.109M sodium citrate and immediately diluted (1:2) in sterile, LPS-free RPMI 1640 medium (Invitrogen) containing 10μM Gly-Pro-Arg-Asp (Sigma-Aldrich). Alternatively, whole blood was centrifuged at 200g for 20 minutes to obtain platelet-rich plasma and diluted (1:2) in sterile, LPS-free RPMI 1640 medium. Whole-blood samples were used for the determination of platelet-leukocyte complexes by flow cytometry. Platelet-poor plasma samples were further processed to quantify and assay microparticles (by flow cytometry and immunoblotting) or used for analysis of C3a(desArg) or terminal complement complex (TCC) levels by ELISA. Platelet-rich plasma was used to determine C3 or C9 on platelets by flow cytometry. Whole blood or platelets were used within 2 hours of sampling and plasma samples were stored at −80°C until assayed.
Stimulation of whole blood by Stx2, Stx1, Stx1B, or LPS
Whole blood was stimulated with Stx2, Stx1, Stx1B, and/or LPS according to a method described previously7 except for modification of the stimulation time (which was reduced to 40 minutes) and the inclusion of the Stx1 and Stx1B experiments. Stx2 (a gift from T. G. Obrig, Department of Microbiology and Immunology, University of Maryland School of Medicine, Baltimore, MD) and Stx1 and Stx1B (a gift from J. Brunton, Department of Microbiology, Toronto General Hospital, Toronto, ON) were all used at a final concentration of 2.8 × 10−9M diluted in LPS-free PBS (PAA Laboratories). The LPS content of the Stx2 and Stx1 preparations was assayed by the limulus amebocyte assay (Coatex) and found to be less than 50 pg/mL (the detection limit). Likewise, whole blood was incubated with O157LPS (a gift from P. I. Tarr, Department of Pediatrics, Division of Gastroenterology and Nutrition, Washington University, St Louis, MO). O157LPS was purified by phenol extraction).20 Whole blood was also incubated with non-EHEC-LPS O111:B4 (Sigma-Aldrich). Both O157LPS and O111LPS were used at a final concentration of 0.5 μg/mL as determined by the limulus amebocyte assay.
Detection of platelet-leukocyte complexes
Platelet-leukocyte complexes were detected as described previously.7 Briefly, whole blood was incubated with mouse anti–human CD42b:allophycocyanin (APC; at a concentration of 1:8 to detect platelets) and mouse anti–human CD38:PE (RPE; at a concentration of 1:10 to detect monocytes) simultaneously to detect platelet-monocyte complexes. Similarly, whole blood was incubated with mouse anti–human CD42b:APC and mouse anti–human CD66:RPE (at a concentration of 1:10 to detect neutrophils) simultaneously to detect platelet-neutrophil complexes. Mouse IgG1:APC and mouse IgG1:RPE were used as isotype controls. All antibodies were from BD Biosciences.
The experimental conditions are described in the supplemental Methods and in supplemental Figure 1 (available on the Blood Web site; see the Supplemental Materials link at the top of the online article). These conditions were the basis for further experiments used for the detection of complement on complexes.
Detection of Stx2 or O157LPS binding to platelet-leukocyte complexes
Binding of Stx2 or O157LPS to platelet-leukocyte complexes was detected as described previously.7,10 Briefly, whole blood was incubated with mouse monoclonal anti-Stx2 IgG1 antibody (11E10, 200ng/mL, a gift from T. G. Obrig) diluted in 0.1% saponin (Sigma-Aldrich)/PBS or mouse anti-O157LPS (10μg/mL, a gift from R. Johnson, Public Health Agency of Canada, Guelph, ON) diluted in PBS. Mouse IgG1 (Dako) was used as a negative control for both antibodies, and rabbit anti–mouse:RPE F(ab′)2 (1:1000; Dako) was the secondary antibody.
Detection of surface-bound C3 or C9 on platelet-leukocyte complexes
C3 or C9 deposition on platelet-leukocyte complexes was detected by incubation of whole blood with an antibody mixture of chicken anti–human C3:FITC (1:600; Diapensia) or mouse anti–human C9:FITC (1:50; Hycult Biotech), CD42b:APC, and mouse anti–human CD38:RPE, or mouse anti–human CD66:RPE. Chicken anti–human insulin:FITC (1:600; Diapensia) or mouse IgG1:FITC (1:50; Hycult Biotech) were used as irrelevant antibodies.
Inhibition of complement deposition on platelet-leukocyte complexes
Whole blood was diluted 1:1 in a combination of RPMI 1640 and 25mM EDTA–Veronal-buffered saline (EDTA-VBS) solution (145mM NaCl, 1.8mM Na-diethyl-barbiturate, 3mM 5.5′-diethyl barbiturate acid, pH 7.3) or RPMI and 10mM EGTA-VBS solution. Ca2+ and Mg2+ concentrations were adjusted to 0.1 mg/mL to resemble the concentrations in RPMI alone before incubation with Stx2 and/or O157LPS for 40 minutes at 37°C. Alternatively, whole blood was incubated with rat anti-Gb3 (20μg/mL; Beckman Coulter) to block the Stx receptor, rabbit anti–human TLR4 (20 μg/mL; eBiosciences) to block the LPS receptor, or a combination of both antibodies for 1 hour at room temperature before incubation with Stx2 and/or O157LPS for 40 minutes at 37°C. Rat IgM (Beckman Coulter) and rabbit IgG (Dako) were used as irrelevant antibodies. C3 or C9 deposition was detected as described in “Detection of surface-bound C3 or C9 on platelet-leukocyte complexes” using chicken anti–human C3:FITC and mouse anti–human C9:FITC.
Isolation of microparticles
Microparticles were isolated from patient whole blood or from whole blood stimulated with or without agonists (Stx2, Stx1, Stx1B, or LPS). Platelet-free plasma was obtained from whole blood by an initial centrifugation step at 2600g for 15 minutes, followed by a 5-minute centrifugation at 9900g at room temperature to remove larger apoptotic bodies. Microparticles were washed twice in HBSS without Ca2+/Mg2+ (PAA Laboratories), centrifuged at 20 800g for 5 minutes, and resuspended in annexin V–binding buffer (BD Biosciences). All buffers and cell media were filtered to exclude larger particles using a 0.2-μm filter (Schleicher-Schuell) before use. The number of microparticles per milliliter of plasma was calculated as described previously.15 Briefly, a defined quantity of 6.0-μm nonfluorescent calibration beads (BD Biosciences) was added to each sample tube and run concurrently with the microparticles, thus allowing for quantitative determination of microparticles. The analysis was terminated when 10 000 beads were counted and the absolute number of microparticles determined by the formula N = (microparticles/beads counted) × (beads added/μL plasma) × the dilution factor.
Labeling of microparticles with cell-specific antibodies
Microparticle subpopulations were determined by incubation with antibodies directed against membrane-specific antigens on platelets, monocytes, and neutrophils. This was achieved using a combination of mouse anti–human CD42b:RPE (1:20; BD Biosciences), mouse anti–human CD38:PerCP-Cy5.5 (1:20; BD Biosciences), or mouse anti–human CD66:RPE, respectively. Surface-bound C3 or C9 on microparticles were detected by incubation with chicken anti–human C3:FITC, mouse anti–human C9:FITC, or mouse anti–human C5b-9 (1:1000; Quidel). Nonspecific binding was determined by subtraction of bound irrelevant antibodies. Irrelevant control antibodies with equivalent protein:fluorescein ratios were used for this purpose: mouse IgG1:RPE, IgG1:PerCP-Cy 5.5 (BD Biosciences), or IgG1:FITC, as well as chicken anti–human insulin or mouse IgG1 (Dako), followed by rabbit anti–mouse:FITC F(ab′)2 (1:500; Dako).
Characterization of C3 or C9 on platelets and platelet-derived microparticles
Platelet-rich plasma was obtained from whole blood, diluted 1:2 in RPMI 1640 medium, and incubated with Stx2 (2.8 × 10−9M) or O157LPS (0.5 μg/mL) or left unstimulated for 40 minutes at 37°C, followed by centrifugation at 2000g for 10 minutes to isolate platelets, which were then washed once in PBS. Likewise, platelet-derived microparticles were prepared from whole blood stimulated with Stx2 or O157LPS as described in “Isolation of microparticles.” Platelets or platelet-derived microparticles were incubated with a combination of mouse anti–human CD42b:APC (1:10) and mouse anti–human activated C3b (1:100; Hycult Biotech), mouse anti–human iC3b (1:100; Thermo Scientific), or mouse anti–human C3c (1:1000; Quidel), respectively, for 20 minutes. Mouse IgG2a, IgG2b, and IgG1 were used as negative control antibodies, and rabbit anti–mouse:FITC F(ab′)2 (Dako) was used as the secondary antibody.
Platelets or microparticles were incubated with a combination of mouse anti–human CD42b:APC and mouse anti–human C9 neoantigen (1:100; Hycult Biotech) or mouse anti–human C5b-9. Mouse IgG1 was the negative control antibody and rabbit anti–mouse:FITC F(ab′)2 was the secondary antibody.
Detection of membrane-bound complement regulators on microparticles
Membrane-bound complement regulators on microparticles was determined by incubation of microparticles with mouse anti–human CD35:FITC (complement receptor 1 [CR1]; 1:20), mouse anti–human CD55:APC (decay accelerating factor [DAF]; 1:40), or mouse anti–human CD46:RPE (membrane cofactor protein [MCP]; 1:50). Subpopulations of microparticles were identified by incubation with mouse anti–human CD42b:RPE or CD42b:APC, mouse anti–human CD38PerCP-Cy5.5, or mouse anti–human CD66:FITC (1:20) or CD66:RPE, respectively. Mouse IgG1:FITC, IgG2a:APC, and IgG1:RPE were used as irrelevant antibody controls (for anti-CR1, DAF, and MCP antibodies, respectively). Mouse IgG1:RPE, IgG1:APC, IgG1:PerCP-Cy 5.5, and IgG1:FITC were used as irrelevant control antibodies for identification of microparticle populations. All antibodies were from BD Biosciences.
Isolation of neutrophils
Neutrophils were isolated under LPS-free conditions from citrated peripheral blood collected from healthy donors using 1-step density gradient centrifugation with Polymorphprep (Nycomed), followed by hypotonic lysis of erythrocytes. Lysis was stopped by the addition of 10× PBS (6:1). Neutrophils were washed twice in PBS and resuspended in RPMI 1640 medium with GlutaMAX (Invitrogen), resulting in a suspension consisting of ∼ 96% neutrophils as determined by flow cytometry using mouse anti–human CD66:RPE antibody.
Detection of microparticle phagocytosis by neutrophils
Microparticles were isolated from Stx2- or O157LPS-stimulated or unstimulated whole blood, washed twice in PBS, and stained with 0.1μM 2′,7′-bis-(2-carboxyethyl)-5-(and-6)-carboxyfluorescein-acetoxy-methylester (BCECF-AM; Invitrogen) for 30 minutes at 37°C, washed in PBS, and resuspended in a buffer of RPMI 1640 with GlutaMAX. BCECF-AM is nonfluorescent when extracellular but becomes fluorescent when converted to BCECF by intracellular esterases.
Neutrophils (1 × 106/mL) were incubated with microparticles (2.5 × 106/mL) stained with BCECF-AM and 20% (vol/vol) plasma from ABO-matched individuals for 1 hour at 37°C with end-over-end rotation. Samples were washed twice and kept on ice until assayed. Phagocytosis of microparticles was detected as described in “Flow cytometry acquisition and interpretation of data.”
Flow cytometry acquisition and interpretation of data
Whole blood samples were lysed with FACSLyse (Dako). Flow cytometry was performed using a FACSCanto II instrument with FACSDiva software Version 6.0 (Becton Dickinson Immunocytometry Systems). Platelet-leukocyte complexes and microparticles were defined and detected as described previously.7 Determination of C3 or C9 deposition on platelet-leukocyte complexes and microparticles was performed using 3- or 4-color analysis for simultaneous detection of platelet-leukocyte complexes or microparticles and C3 or C9. In phagocytosis experiments, neutrophils were selectively identified by appropriate settings of forward- and side-scatter and measured for green fluorescence from associated BCECF-labeled microparticles.
Immunoblotting to detect C3 and C9 on microparticles
Microparticles were isolated from 1 mL of Stx2- or O157LPS-stimulated or unstimulated whole blood. Samples were diluted (1:10) and subjected to 10% sodium dodecyl sulfate–polyacrylamide gel electrophoresis under reducing conditions. Proteins were transferred onto equilibrated polyvinylidene difluoride membranes and blocked with casein solution (Vector Laboratories). Immunoblotting was performed with rabbit anti–human C3c (Dako) or mouse anti–human iC3b, followed by goat anti–rabbit HRP or goat anti–mouse HRP (Dako), respectively. Signal was detected by chemiluminescence using ECL Plus (Amersham).
Detection of C3a(desArg) or terminal complement complex in plasma
C3a(desArg) and human TCC in plasma from patients or controls was quantified using a C3a assay kit or TCC kit (both from Hycult Biotech) following the manufacturer's instructions.
Alternatively, citrated whole blood (diluted 1:2 in RPMI 1640) was stimulated with Stx2 or O157LPS or left unstimulated for 40 minutes at 37°C, and platelet-free plasma was obtained by centrifugation at 2000g for 15 minutes. Samples were stored at −80°C until being analyzed with the C3a(desArg) and TCC kits.
Statistical analysis
Comparison of blood cells incubated with the various agonists or unstimulated blood cells, generation of microparticles in stimulated or unstimulated whole blood, microparticles in patient plasma or control plasma, and C3 or C9 on complexes or microparticles were analyzed by the Mann-Whitney U test. Correlations between clinical manifestations and microparticles were analyzed using the Spearman ρ test. P values ≤ .05 were considered significant. Statistical analyses were performed using PASW 18 software (SPSS).
Results
C3 bound to platelet-leukocyte complexes in a patient with HUS
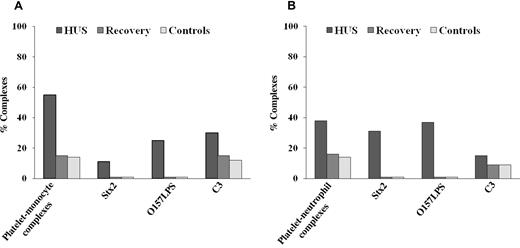
Stx2, O157LPS, and C3 deposition on circulating platelet-monocyte or platelet-neutrophil complexes (Figure 1A-B) were measured in whole blood from 1 patient with E coli O157:H7–associated HUS taken during the acute phase of disease (patient 1 in Table 1) and compared with pediatric controls (n = 5). C3 was detected by flow cytometry on 30% of platelet-monocyte complexes and on 15% of platelet-neutrophil complexes during the acute phase of disease, but decreased to 14% of platelet-monocyte complexes and 9% of platelet-neutrophil complexes after recovery, levels that were comparable to the pediatric controls (Figure 1A-B). C9 was not determined on the patient's platelet-leukocyte complexes. Stx2 was detected on 15% of platelet-monocyte complexes and on 30% of platelet-neutrophil complexes. Similarly, O157LPS was detected on 30% of platelet-monocyte complexes and on 37% of platelet-neutrophil complexes. No binding of Stx2 or O157LPS was detected after recovery or in the pediatric controls.
Circulating platelet-leukocyte complexes and surface-bound C3 in a patient with EHEC-associated HUS. A pediatric HUS patient had increased levels of circulating platelet-monocyte (A) and platelet-neutrophil (B) complexes during the acute phase of disease compared with levels obtained after recovery and with pediatric controls (n = 5). Levels of platelet-monocyte (A) and platelet-neutrophil (B) complexes with surface-bound C3 were elevated during the acute phase of disease and decreased at recovery to levels similar to those seen in the pediatric controls.
Circulating platelet-leukocyte complexes and surface-bound C3 in a patient with EHEC-associated HUS. A pediatric HUS patient had increased levels of circulating platelet-monocyte (A) and platelet-neutrophil (B) complexes during the acute phase of disease compared with levels obtained after recovery and with pediatric controls (n = 5). Levels of platelet-monocyte (A) and platelet-neutrophil (B) complexes with surface-bound C3 were elevated during the acute phase of disease and decreased at recovery to levels similar to those seen in the pediatric controls.
Patients with HUS had increased levels of circulating microparticles with bound C3 and C9
Plasma levels of circulating microparticles with surface-bound C3 or C9 were measured by flow cytometry during the acute phase of the disease and compared with pediatric controls (n = 6) and patients with acute renal failure (n = 3). Plasma taken during the acute phase showed significantly higher levels of circulating microparticles with surface-bound C3 and C9 (Table 2) compared with the controls. Mean fluorescence intensity (MFI) levels of C3, C9, and C5b-9 on platelet- and leukocyte-derived microparticles are presented in supplemental Figure 2. Microparticles originated primarily from platelets followed by monocytes. Levels at recovery were similar to those found in the controls.
Numbers and cellular origin of microparticles with surface-bound C3 or C9 in plasma from HUS patients
| . | CD42b-positive microparticles, 103/mL . | CD38-positive microparticles, 103/mL . | CD66-positive microparticles, 103/mL . | |||
|---|---|---|---|---|---|---|
| C3-positive . | C9-positive . | C3-positive . | C9-positive . | C3-positive . | C9-positive . | |
| Acute phase (n = 12) | 818* (552-1001) | 780* (654-1031) | 83† (48-128) | 84† (50-124) | 1.8* (1.3-2.5) | 1.5* (1.1-2.4) |
| Recovery (n = 12) | 64 (15-342) | 28 (11-258) | 35 (8-285) | 36 (10-304) | 0.2 (0.1-0.4) | 0.2 (0.1-0.6) |
| Pediatric controls (n = 6) | 73 (54-271) | 69 (55-284) | 48 (17-216) | 38 (15-202) | 0.3 (0.1-0.6) | 0.4 (0.2-0.7) |
| Renal failure controls (n = 3) | 65 (35-132) | 52 (10-109) | 41 (18-198) | 38 (7-124) | 5 (2-133) | 2.7 (2-93) |
| . | CD42b-positive microparticles, 103/mL . | CD38-positive microparticles, 103/mL . | CD66-positive microparticles, 103/mL . | |||
|---|---|---|---|---|---|---|
| C3-positive . | C9-positive . | C3-positive . | C9-positive . | C3-positive . | C9-positive . | |
| Acute phase (n = 12) | 818* (552-1001) | 780* (654-1031) | 83† (48-128) | 84† (50-124) | 1.8* (1.3-2.5) | 1.5* (1.1-2.4) |
| Recovery (n = 12) | 64 (15-342) | 28 (11-258) | 35 (8-285) | 36 (10-304) | 0.2 (0.1-0.4) | 0.2 (0.1-0.6) |
| Pediatric controls (n = 6) | 73 (54-271) | 69 (55-284) | 48 (17-216) | 38 (15-202) | 0.3 (0.1-0.6) | 0.4 (0.2-0.7) |
| Renal failure controls (n = 3) | 65 (35-132) | 52 (10-109) | 41 (18-198) | 38 (7-124) | 5 (2-133) | 2.7 (2-93) |
Data are expressed as the median and range (in parentheses) of circulating microparticles positive for each membrane-specific marker per milliliter.
P < .001 and †P < .01 comparing microparticles in plasma from patients with pediatric controls.
Laboratory findings—hemoglobin, platelet counts, creatinine, urea, dialysis (+ or −), C3 and C4 levels, and extrarenal complications (+ or −)—during the acute phase of HUS (Table 1) were compared with the levels of C3 and C9 on platelet-derived microparticles (103/mL) and did not show a significant correlation (data not shown).
Patients with HUS had increased levels of C3a(desArg) and TCC in plasma
Plasma levels of C3a(desArg) and TCC were measured during the acute phase (n = 10, patients 1-10) and after recovery and compared with pediatric controls (n = 6). Levels were elevated during the acute phase of disease, as presented in Table 3.
C3a(desArg) and TCC levels in plasma from HUS patients
| . | n . | C3a(desArg) median, ng/mL (range) . | TCC median, AU/mL (range) . |
|---|---|---|---|
| Acute phase | 10* | 465† (280-690) | 5.8‡ (4.0-7.3) |
| Recovery | 10* | 109 (72-180) | 1.4 (0.7-2.4) |
| Pediatric controls | 6 | 90 (52-124) | 0.98 (0.68-2.5) |
| . | n . | C3a(desArg) median, ng/mL (range) . | TCC median, AU/mL (range) . |
|---|---|---|---|
| Acute phase | 10* | 465† (280-690) | 5.8‡ (4.0-7.3) |
| Recovery | 10* | 109 (72-180) | 1.4 (0.7-2.4) |
| Pediatric controls | 6 | 90 (52-124) | 0.98 (0.68-2.5) |
Plasma samples analyzed from patients 1-10 (Table 1).
P < .01 and ‡P < .05 comparing plasma taken during the acute phase with pediatric controls. There was no significant difference between plasma taken after recovery and pediatric controls.
Stx and O157LPS induced C3 and C9 deposition on platelet-leukocyte complexes
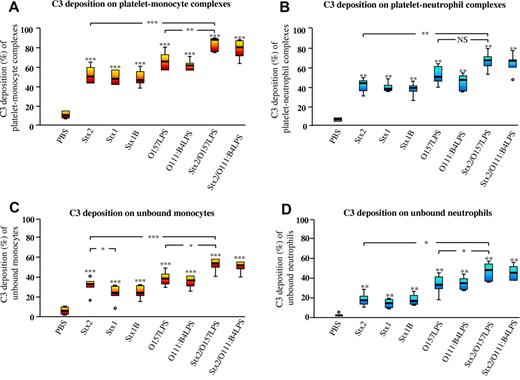
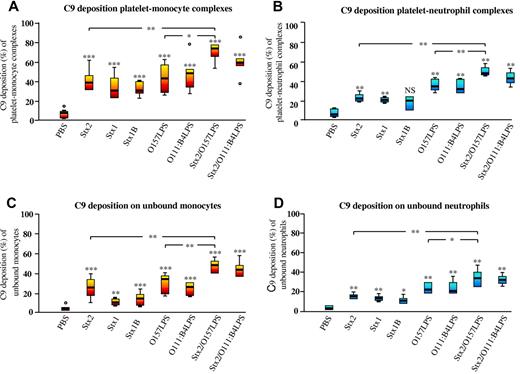
Incubation of whole blood with Stx2, Stx1, or Stx1B induced a significant increase in platelet-monocyte and platelet-neutrophil complexes with surface-bound C3 (Figure 2A-B) compared with PBS-treated whole blood. Similarly, Stx2 or Stx1 induced a significant increase in platelet-monocyte and platelet-neutrophil complexes with surface-bound C9 (Figure 3A-B), whereas Stx1B only induced a significant increase in surface-bound C9 in the platelet-monocyte population. Stimulation with O157LPS also induced C3 and C9 deposition. Costimulation with Stx2 and O157LPS increased the population of platelet-monocyte complexes with surface-bound C3 (Figure 2A-B) and the population of platelet-monocyte and platelet-neutrophil complexes with surface-bound C9 (Figure 3A-B) compared with PBS-treated samples or Stx2 or O157LPS alone. Both C3 (Figure 2C-D) and C9 (Figure 3C-D) bound to a greater extent to monocytes and neutrophils in complex with platelets than to platelet-free monocytes or neutrophils.
Surface-bound C3 on platelet-leukocyte complexes induced by Stx2, Stx1, Stx1B, O157LPS, and/or O111LPS. Incubation of whole blood with Stx2, Stx1, Stx1B, or O157LPS induced C3 deposition on platelet-monocyte (A) and platelet-neutrophil (B) complexes and a lesser increase of C3 on unbound monocytes (C) and unbound neutrophils (D). Data are expressed as box-plots depicting the median and range regarding the percentage of the platelet-monocyte or platelet-neutrophil population or the percentage of unbound monocytes or neutrophils that were positive for C3 (n = 12 experiments). The horizontal line within each box represents the median. The upper and lower limits of the box represent the interquartile range. The lower and upper limits represent the range. C3 deposition on complexes or on unbound monocytes or neutrophils in whole blood incubated with an agonist was compared with PBS-treated whole blood. *P < .05; **P < .01; ***P < .001. NS indicates not significant.
Surface-bound C3 on platelet-leukocyte complexes induced by Stx2, Stx1, Stx1B, O157LPS, and/or O111LPS. Incubation of whole blood with Stx2, Stx1, Stx1B, or O157LPS induced C3 deposition on platelet-monocyte (A) and platelet-neutrophil (B) complexes and a lesser increase of C3 on unbound monocytes (C) and unbound neutrophils (D). Data are expressed as box-plots depicting the median and range regarding the percentage of the platelet-monocyte or platelet-neutrophil population or the percentage of unbound monocytes or neutrophils that were positive for C3 (n = 12 experiments). The horizontal line within each box represents the median. The upper and lower limits of the box represent the interquartile range. The lower and upper limits represent the range. C3 deposition on complexes or on unbound monocytes or neutrophils in whole blood incubated with an agonist was compared with PBS-treated whole blood. *P < .05; **P < .01; ***P < .001. NS indicates not significant.
Surface-bound C9 on platelet-leukocyte complexes induced by Stx2, Stx1, Stx1B, Ol57LPS, and/or O111LPS. Incubation of whole blood with Stx2, Stx1, Stx1B, or O157LPS induced C9 deposition on platelet-monocyte (A) and platelet-neutrophil (B) complexes and to a lesser extent on unbound monocytes (C) and unbound neutrophils (D). Data are expressed as box-plots depicting the median and range regarding the percentage of the platelet-monocyte or platelet-neutrophil population or the percentage of unbound monocytes or neutrophils that were positive for C9 (n = 12 experiments). C9 deposition on complexes or unbound monocytes or neutrophils in whole blood incubated with an agonist was compared with that in PBS-treated whole blood. *P < .05; **P < .01; ***P < .001. NS indicates not significant.
Surface-bound C9 on platelet-leukocyte complexes induced by Stx2, Stx1, Stx1B, Ol57LPS, and/or O111LPS. Incubation of whole blood with Stx2, Stx1, Stx1B, or O157LPS induced C9 deposition on platelet-monocyte (A) and platelet-neutrophil (B) complexes and to a lesser extent on unbound monocytes (C) and unbound neutrophils (D). Data are expressed as box-plots depicting the median and range regarding the percentage of the platelet-monocyte or platelet-neutrophil population or the percentage of unbound monocytes or neutrophils that were positive for C9 (n = 12 experiments). C9 deposition on complexes or unbound monocytes or neutrophils in whole blood incubated with an agonist was compared with that in PBS-treated whole blood. *P < .05; **P < .01; ***P < .001. NS indicates not significant.
Stx2- and/or O157LPS-stimulated whole blood induced complement activation via the alternative pathway
Whole blood diluted in RPMI/EDTA-VBS and stimulated with Stx2 and/or O157LPS exhibited marked reduction of surface-bound C3 or C9 on platelet-monocyte complexes (Table 4). Whole blood diluted in RPMI1640/EDTA-VBS and stimulated with Stx2 and/or O157LPS exhibited slightly reduced surface-bound C3 and C9 on platelet-monocyte complexes compared with unstimulated whole blood diluted in RPMI alone. Inhibition experiments were not performed for platelet-neutrophil complexes. The results indicate that Stx2 and/or O157LPS activated complement mostly via the alternative pathway.
C3 and C9 deposition on platelet-monocyte complexes occurred via the alternative pathway
| Agonist . | RPMI . | EDTA-VBS/RPMI . | EGTA-VBS/RPMI . | |||
|---|---|---|---|---|---|---|
| C3-positive . | C9-positive . | C3-positive . | C9-positive . | C3-positive . | C9-positive . | |
| Unstimulated | 7 (5-8) | 5 (2-9) | 9 (8-11) | 5 (1-13) | 10 (7-12) | 4 (2-13) |
| Stx2 | 44 (10-82) | 15 (15-21) | 15 (7-27) | 7 (3-15) | 34 (10-40) | 17 (2-28) |
| O157LPS | 58 (35-81) | 31 (20-46) | 20 (16-44) | 8 (7-16) | 47 (10-53) | 21 (14-25) |
| Stx2/O157LPS | 60 (46-87) | 28 (15-40) | 22 (12-43) | 10 (8-21) | 55 (14-67) | 25 (5-31) |
| Agonist . | RPMI . | EDTA-VBS/RPMI . | EGTA-VBS/RPMI . | |||
|---|---|---|---|---|---|---|
| C3-positive . | C9-positive . | C3-positive . | C9-positive . | C3-positive . | C9-positive . | |
| Unstimulated | 7 (5-8) | 5 (2-9) | 9 (8-11) | 5 (1-13) | 10 (7-12) | 4 (2-13) |
| Stx2 | 44 (10-82) | 15 (15-21) | 15 (7-27) | 7 (3-15) | 34 (10-40) | 17 (2-28) |
| O157LPS | 58 (35-81) | 31 (20-46) | 20 (16-44) | 8 (7-16) | 47 (10-53) | 21 (14-25) |
| Stx2/O157LPS | 60 (46-87) | 28 (15-40) | 22 (12-43) | 10 (8-21) | 55 (14-67) | 25 (5-31) |
Data are expressed as the percentage of platelet-monocyte complexes positive for surface-bound C3 or C9 and are shown as the median and range (in parentheses) from 3 different experiments.
Blocking of Stx2 and/or O157LPS binding reduced platelet-monocyte complex formation and surface-bound C3 and C9
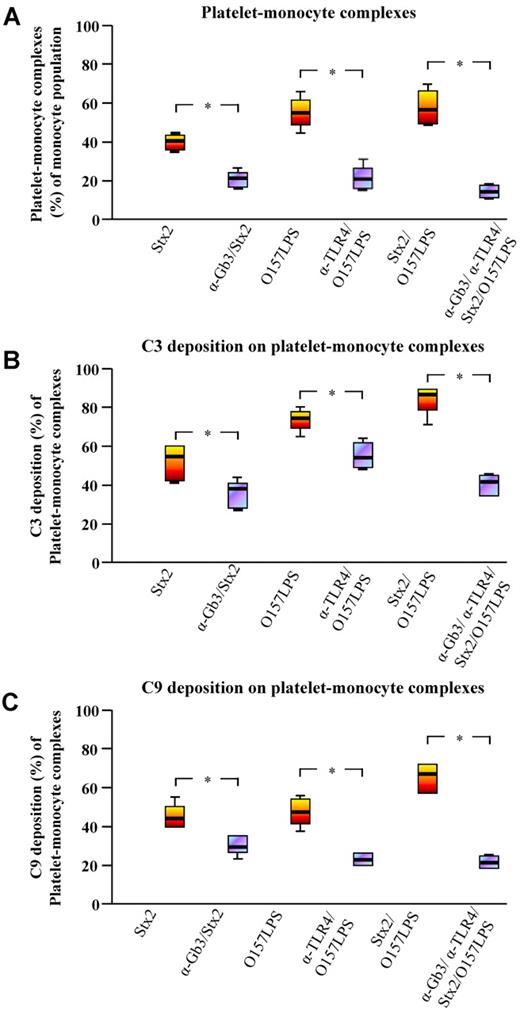
Incubation of whole blood with anti-Gb3 before incubation with Stx2 reduced platelet-monocyte complex formation significantly by 67%. Similarly, incubation of whole blood with anti-TLR4 before incubation with O157LPS reduced platelet-monocyte complex formation by 76% (Figure 4A). An even more prominent reduction of 80% was noted when whole blood was preincubated with both anti-Gb3 and anti-TLR4 before the addition of Stx2 and O157LPS simultaneously (Figure 4A). Likewise, preincubation with anti-Gb3 and/or anti-TLR4 reduced surface-bound C3 (Figure 4B) and C9 (Figure 4C) in Stx2 and/or O157LPS-stimulated samples. The most pronounced reduction of complex formation and surface-bound C3 or C9 was noted in whole blood preincubated with a combination of anti-Gb3 and anti-TLR4 before coincubation with Stx2 and O157LPS. The irrelevant antibodies used, rabbit IgG as well as rat IgM, did not inhibit complex formation or C3 and C9 deposition (data not shown). Inhibition experiments were not performed for platelet-neutrophil complexes.
Inhibition of platelet-monocyte complex formation and complement deposition. (A) Blocking of Stx2 and/or O157LPS binding by preincubation of whole blood with anti-Gb3 and/or anti-TLR4 before incubation with Stx2 and/or O157LPS reduced platelet-monocyte complex formation significantly. C3 (B) and C9 (C) deposition on the blood-cell complexes was similarly reduced by anti-Gb3 and anti-TLR4 in these experiments. Data are expressed as box-plots depicting median and range (n = 4 experiments). Complex formation and C3 or C9 deposition on complexes in whole blood incubated with an agonist were compared with that in whole blood preincubated with anti-Gb3 and/or anti-TLR4 before the addition of the agonist. *P < .05.
Inhibition of platelet-monocyte complex formation and complement deposition. (A) Blocking of Stx2 and/or O157LPS binding by preincubation of whole blood with anti-Gb3 and/or anti-TLR4 before incubation with Stx2 and/or O157LPS reduced platelet-monocyte complex formation significantly. C3 (B) and C9 (C) deposition on the blood-cell complexes was similarly reduced by anti-Gb3 and anti-TLR4 in these experiments. Data are expressed as box-plots depicting median and range (n = 4 experiments). Complex formation and C3 or C9 deposition on complexes in whole blood incubated with an agonist were compared with that in whole blood preincubated with anti-Gb3 and/or anti-TLR4 before the addition of the agonist. *P < .05.
Stx and O157LPS induced the release of microparticles with surface-bound C3 and C9
Levels of microparticles with surface-bound C3 or C9 were measured in whole blood stimulated with Stx2, Stx1, Stx1B, O157LPS, or a combination of Stx2 and O157LPS. The agonists induced a significant increase in microparticles with surface-bound C3 or C9 compared with PBS-treated samples (Table 5). The highest levels of microparticles were noted in whole blood costimulated with Stx2 and O157LPS compared with Stx2 or O157LPS alone. The combination of Stx2 and O157LPS induced a significant increase in C3- and C9-positive microparticles derived from platelets compared with each agonist alone.
Numbers and cellular origin of microparticles and microparticles with surface-bound C3 and/or C9
| Agonist . | Total amount of microparticles, 103/ml . | CD42b-positive microparticles, 103/mL . | CD38-positive microparticles, 103/mL . | CD66-positive microparticles, 103/mL . | |||
|---|---|---|---|---|---|---|---|
| C3-positive . | C9-positive . | C3-positive . | C9-positive . | C3-positive . | C9-positive . | ||
| Stx1 | 1992* (1068-2395) | 702* (259-871) | 717* (268-870) | 336* (245-528) | 312* (236-598) | 0.3NS (0.2-0.4) | 0.2NS (0.1-0.4) |
| Stx1B | 1301* (967-2364) | 766* (505-864) | 721* (478-819) | 264* (136-479) | 351* (116-519) | 0.1NS (0.1-0.2) | 0.2NS (0.1-0.3) |
| Stx2 | 1341* (1145-2003) | 845* (745-991) | 839* (776-990) | 421* (155-638) | 455* (170-685) | 0.3NS (0.1-0.5) | 0.2NS (0.1-0.3) |
| O157LPS | 1749* (1487-2128) | 859* (528-1047) | 822* (512-1055) | 487* (300-739) | 499* (281-732) | 1.4* (1.0-1.7) | 1.5* (0.9-1.8) |
| O111:B4LPS | 1351* (525-2970) | 805* (499-1003) | 831* (514-1019) | 415* (178-880) | 453* (185-848) | 1.4* (0.5-1.8) | 1.5* (0.5-1.7) |
| Stx2/O157LPS | 2015* (1632-2785) | 1466* (811-1895)† | 1414* (817-1861)† | 312* (269-735) | 328* (275-761) | 1.9* (1.5-2.2) | 1.8* (1.4-2.3) |
| Stx2/O111:B4LPS | 1430* (931-2623) | 994* (725-1468) | 1218* (802-1865) | 286* (251-626) | 295* (268-640) | 1.7* (1.5-1.9) | 1.6* (1.2-1.9) |
| PBS | 240 (153-565) | 99 (54-202) | 85 (48-212) | 67 (34-154) | 63 (28-151) | 0.3 (0.1-0.3) | 0.2 (0.1-0.3) |
| Agonist . | Total amount of microparticles, 103/ml . | CD42b-positive microparticles, 103/mL . | CD38-positive microparticles, 103/mL . | CD66-positive microparticles, 103/mL . | |||
|---|---|---|---|---|---|---|---|
| C3-positive . | C9-positive . | C3-positive . | C9-positive . | C3-positive . | C9-positive . | ||
| Stx1 | 1992* (1068-2395) | 702* (259-871) | 717* (268-870) | 336* (245-528) | 312* (236-598) | 0.3NS (0.2-0.4) | 0.2NS (0.1-0.4) |
| Stx1B | 1301* (967-2364) | 766* (505-864) | 721* (478-819) | 264* (136-479) | 351* (116-519) | 0.1NS (0.1-0.2) | 0.2NS (0.1-0.3) |
| Stx2 | 1341* (1145-2003) | 845* (745-991) | 839* (776-990) | 421* (155-638) | 455* (170-685) | 0.3NS (0.1-0.5) | 0.2NS (0.1-0.3) |
| O157LPS | 1749* (1487-2128) | 859* (528-1047) | 822* (512-1055) | 487* (300-739) | 499* (281-732) | 1.4* (1.0-1.7) | 1.5* (0.9-1.8) |
| O111:B4LPS | 1351* (525-2970) | 805* (499-1003) | 831* (514-1019) | 415* (178-880) | 453* (185-848) | 1.4* (0.5-1.8) | 1.5* (0.5-1.7) |
| Stx2/O157LPS | 2015* (1632-2785) | 1466* (811-1895)† | 1414* (817-1861)† | 312* (269-735) | 328* (275-761) | 1.9* (1.5-2.2) | 1.8* (1.4-2.3) |
| Stx2/O111:B4LPS | 1430* (931-2623) | 994* (725-1468) | 1218* (802-1865) | 286* (251-626) | 295* (268-640) | 1.7* (1.5-1.9) | 1.6* (1.2-1.9) |
| PBS | 240 (153-565) | 99 (54-202) | 85 (48-212) | 67 (34-154) | 63 (28-151) | 0.3 (0.1-0.3) | 0.2 (0.1-0.3) |
Data are expressed as median and (range) of circulating microparticles positive for each membrane-specific marker per milliliter. Values were derived from six different experiments.
NS indicates not significant.
P < .001 comparing microparticles released in whole blood stimulated with Stx2, Stx 1, Stx1B, LPS or Stx/LPS with PBS-treated whole blood.
Costimulation of whole blood with Stx2 and O157LPS induced an increase in platelet microparticles with surface-bound C3 or C9 compared with Stx2 (P < .001) or O157LPS (P < .001) alone, while no significant increase was noted on monocyte microparticles in whole blood costimulated with Stx2 and O157LPS compared with each agonist alone.
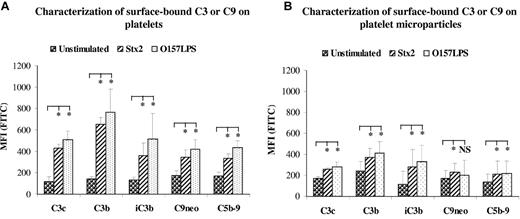
Characterization of surface-bound C3 or C9 on platelets and platelet-derived microparticles
To further characterize surface-bound C3 on platelets and platelet-derived microparticles, 2 monoclonal anti-C3 antibodies were used, one that binds to activated C3b and one that binds to iC3b. These antibodies specifically react with neoepitopes exposed in activated and conformationally altered C3 but not in native C3. Both antibodies bound to platelets and platelet-derived microparticles from Stx2- or O157LPS-stimulated whole blood (Figure 5A-B), indicating that surface-bound C3 was activated C3b, which was to some extent further processed to iC3b. The presence of C3 and iC3b on microparticles stimulated with Stx and O157LPS was further confirmed by immunoblotting. The results are summarized in supplemental Results and in supplemental Figure 3.
Characterization of surface-bound C3 or C9 on platelets and on platelet-derived microparticles. Incubation of platelet-rich plasma (A) or whole blood (B) with Stx2 or O157LPS induced deposition of C3c, C3b, iC3b, C9neo, or C5b-9 on the surface of platelets (A) and to a lesser degree on platelet-derived microparticles (B). Data are expressed as median ± SD (n = 5 experiments). C3 or C9 deposition on platelets (in platelet-rich plasma) or platelet microparticles (in whole blood) incubated with agonists was compared with that in unstimulated samples. *P < .05; **P < .001. NS indicates not significant.
Characterization of surface-bound C3 or C9 on platelets and on platelet-derived microparticles. Incubation of platelet-rich plasma (A) or whole blood (B) with Stx2 or O157LPS induced deposition of C3c, C3b, iC3b, C9neo, or C5b-9 on the surface of platelets (A) and to a lesser degree on platelet-derived microparticles (B). Data are expressed as median ± SD (n = 5 experiments). C3 or C9 deposition on platelets (in platelet-rich plasma) or platelet microparticles (in whole blood) incubated with agonists was compared with that in unstimulated samples. *P < .05; **P < .001. NS indicates not significant.
Surface-bound C9 on platelets were characterized by the use of 2 antibodies specifically reacting with the neoepitope in C9 (C9neo) or neoepitopes in the C5b-9 complex. Both antibodies bound to the surface of platelets and platelet-derived microparticles (Figure 5A-B), indicating the formation of MAC on the surface of platelets and microparticles. O157LPS appeared to induce more deposition of C3b on platelets and platelet-derived microparticles than Stx2, although the difference was nonsignificant. Both stimulants induced significant C3 and C9 deposition compared with unstimulated platelets.
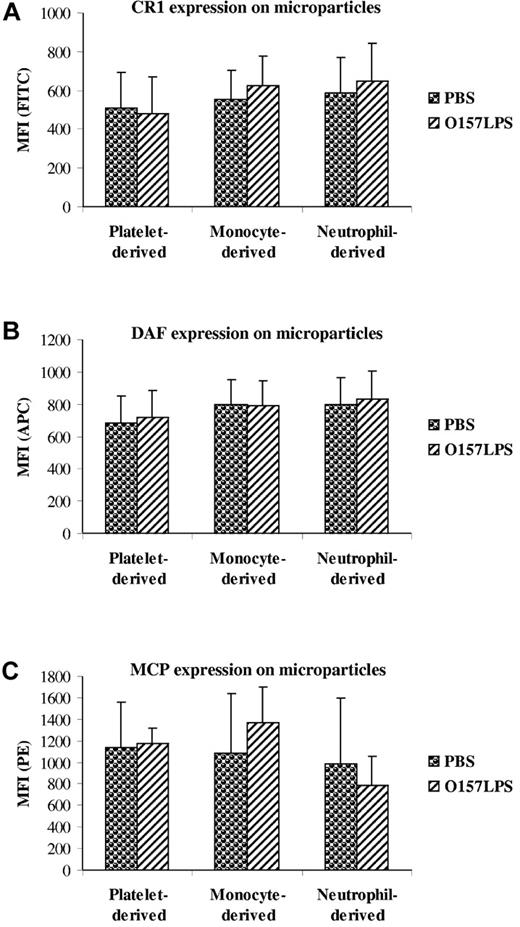
Microparticles expressed complement regulators on their surface
The expression of surface-bound complement regulators was examined on microparticles from O157LPS-stimulated whole blood. CR1 (Figure 6A), DAF (Figure 6B), and MCP (Figure 6C) were detected on microparticles from platelets, monocytes, and neutrophils, and there was no significant difference between stimulated and unstimulated microparticles.
Expression of complement regulators on microparticles. Microparticles from O157LPS-stimulated whole blood were incubated with mouse anti–human CD35:FITC (CR1; A), CD55:APC (DAF; B), or CD46:RPE (MCP; C), showing binding of the antibodies to platelet-, monocyte-, and neutrophil-derived microparticles. Data are presented as the mean fluorescence intensity (MFI) ± SD from 5 independent experiments.
Expression of complement regulators on microparticles. Microparticles from O157LPS-stimulated whole blood were incubated with mouse anti–human CD35:FITC (CR1; A), CD55:APC (DAF; B), or CD46:RPE (MCP; C), showing binding of the antibodies to platelet-, monocyte-, and neutrophil-derived microparticles. Data are presented as the mean fluorescence intensity (MFI) ± SD from 5 independent experiments.
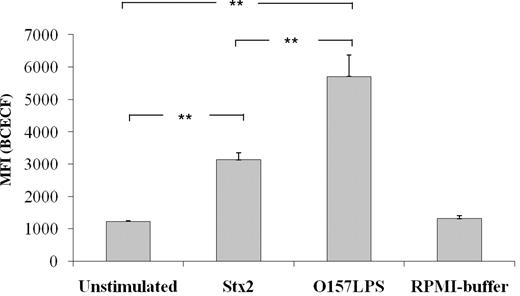
Microparticles released in Stx2- or O157LPS-stimulated whole blood were phagocytosed by neutrophils
Microparticles were isolated from Stx2- or O157LPS-stimulated whole blood, labeled with a fluorescent dye (BCECF-AM), and incubated with neutrophils. Microparticles from Stx2- or O157LPS-stimulated samples were found to be phagocytosed by neutrophils, because the increased fluorescence indicated intracellular localization of the microparticles (Figure 7). Microparticles from unstimulated samples were taken up by neutrophils to a lesser extent (Figure 7). Because equivalent concentrations of microparticles (2.5 × 106/mL) were incubated with neutrophils (1 × 106/mL), these results indicate that microparticles derived from Stx2- or O157:H7-stimulated microparticles were preferentially phagocytosed.
Phagocytic uptake of microparticles by neutrophils. Incubation of neutrophils with microparticles isolated from Stx2- or O157LPS-stimulated whole blood induced phagocytic uptake of the microparticles compared with microparticles isolated from unstimulated whole blood. Data are presented as the MFI ± SD from 5 independent experiments. Uptake of microparticles from Stx2- or O157LPS-stimulated whole blood was compared with that from unstimulated whole blood. **P < .01.
Phagocytic uptake of microparticles by neutrophils. Incubation of neutrophils with microparticles isolated from Stx2- or O157LPS-stimulated whole blood induced phagocytic uptake of the microparticles compared with microparticles isolated from unstimulated whole blood. Data are presented as the MFI ± SD from 5 independent experiments. Uptake of microparticles from Stx2- or O157LPS-stimulated whole blood was compared with that from unstimulated whole blood. **P < .01.
Stx and O157LPS induced C3a(desArg) and TCC release in whole blood
Levels of C3a(desArg) and TCC were measured in Stx2- or O157LPS-stimulated whole blood. Both Stx2 and O157LPS induced a significant increase in C3a(desArg) and TCC concentrations (Table 6) compared with unstimulated samples.
C3a(desArg) and TCC levels in plasma
| Agonist . | C3a(desArg) ng/mL median (range) . | TCC AU/mL median (range) . |
|---|---|---|
| Stx2 | 235 (175-350)* | 6.0 (4.0-10.0)* |
| O157LPS | 385 (200-430)* | 5.8 (3.8-7.0)* |
| Unstimulated | 90 (58-129) | 1.4 (1.2-2.3) |
| Agonist . | C3a(desArg) ng/mL median (range) . | TCC AU/mL median (range) . |
|---|---|---|
| Stx2 | 235 (175-350)* | 6.0 (4.0-10.0)* |
| O157LPS | 385 (200-430)* | 5.8 (3.8-7.0)* |
| Unstimulated | 90 (58-129) | 1.4 (1.2-2.3) |
Data were generated from 5 different experiments.
P < .05 comparing Stx2- or O157LPS-stimulated samples with unstimulated samples.
Discussion
The results of the current study reveal the presence of C3 on platelet-leukocyte complexes in a patient with EHEC-associated HUS. Activated leukocytes and platelets release microparticles during HUS,7 and these blood cell–derived microparticles, particularly those originating from platelets, were shown here to have C3, C9, and C5b-9 deposition on their surfaces. Patients also exhibited elevated levels of C3a and TCC in their plasma. Stimulation of blood cells with Stx2, Stx1, Stx1B, or O157LPS in vitro induced the formation of platelet-leukocyte complexes bearing C3 and C9. Stx and/or O157LPS induced the release of platelet- and leukocyte-derived microparticles bearing C3 and C9 on their surfaces. In addition, Stx and O157LPS induced the release of C3a and TCC in whole blood. The results of this study provide evidence for complement activation on circulating platelets and leukocytes, as well as microparticles derived from these cells, during EHEC-associated HUS, and suggest that Stx and O157LPS contribute to this process. Complement activation on these blood cells could further enhance the inflammatory and thrombotic process.
C3b binds to platelets via P selectin and to neutrophils and monocytes via CR1.12,21 The opsonin iC3b binds to these cells via CR3 (the integrin receptor CD11b/CD18).22,23 We have shown the presence of C3c, iC3b, C3b, C9, and C5b-9 on the platelet surface, particularly after stimulation with Stx2 and O157LPS. Microparticles released from cells during apoptosis and activation24 may bind complement components such as C1q, C3, and C4.25 When stimulated with Stx and O157LPS, platelet-derived microparticles were also demonstrated to have C3c, iC3b, C3b, C9, and C5b-9 on their surfaces. Complement activation on platelets, platelet-leukocyte complexes, and platelet-derived microparticles may be a mechanism of physiologic clearance of unwanted or apoptotic cells and cell blebs,26 but it may also activate the cells.15,27,28
Platelet- and leukocyte-derived microparticles, which were coated with C3 and C9, underwent neutrophil phagocytosis. Complement labeling is a well-studied phenomenon whereby opsonized bacteria undergo phagocytosis by professional phagocytes such as neutrophils, monocytes, macrophages, and, to a lesser extent, dendritic cells,29 and iC3b is an important opsonin in this process.30 Complement-coated microspheres undergo neutrophil phagocytosis.31 The same process appears to occur when complement-coated microparticles are incubated with neutrophils, as shown in the present study, and this may represent a protective mechanism by which neutrophils ingest procoagulant microparticles to prevent excessive thrombosis.
The formation of the C5b-9 complex induces cytolysis when present in high amounts, but in sublytic amounts, cell activation is induced, resulting in the secretion of cytokines, prostanoids, and free radicals.17,32-34 C5b-9 assembly on platelets promotes the formation of the prothrombinase complex, allowing the generation of thrombin.35 Thrombin and C5b-9 induce the exposure of negatively charged phospholipids serving as a procoagulant surface on the platelet membrane,36 thus enabling clot formation. Interestingly, thrombin generation precedes renal injury in HUS.37 In the absence of C3, thrombin can function as a C5 convertase cleaving C538 and has therefore been suggested to induce C3-independent complement activation.39 There appears to be cross-talk between the complement and thrombotic systems, and C5b-9 deposition on blood cells may promote thrombus formation. This is further supported by the finding that complement C3 and C9 deposition on platelets induced both their activation and the release of tissue factor–bearing microparticles.15 In vitro stimulation of blood cells with Stx and O157LPS led to the formation of blood-cell complexes bearing complement C3 and C9, suggesting that complement activation could occur on the cell surface even before membrane blebbing. We propose that the observed complement activation on blood cells may partake in the inflammatory process by enhanced opsonization and chemotaxis,40 and may also induce a prothrombotic state by enhancing platelet aggregation.13
Stx is cytotoxic for most cells, but not for blood cells.3,41,42 Conversely, the life span of neutrophils is extended by Stx.43 This toxin has a stimulatory effect on platelets, monocytes, and neutrophils.3,41,44 Stx and O157LPS induced C3 and C9 deposition on platelets, monocytes, and, to a lesser extent, neutrophils, and even Stx1B alone induced complement deposition, indicating that toxin binding to its receptor was sufficient to induce C3 and C9 deposition. However, Stx and O157LPS may not be the only underlying causes of complement deposition on blood cells during HUS, because complement activation has been documented in other conditions associated with acute renal failure, probably due to complement activation on dialysis membranes.45 Most of the patients studied here did not undergo hemodialysis. However, we cannot exclude the possibility that Stx and O157LPS were not the only factors promoting complement activation, and that inflammatory mediators and uremic toxins may have also contributed to the process.
Previous studies have shown that complement may be activated via the alternative pathway during EHEC-associated HUS, and these patients have low levels of C346 and elevated levels of complement factor Bb and sC5b-9.47 Stx was reported to activate complement via the alternative pathway in the fluid phase48 and to induce C3 deposition on human microvascular endothelial cells, thus promoting thrombus formation.4 Combined with the results presented here, the results of these studies indicate that complement may be activated via the alternative pathway during EHEC-associated HUS, which may promote disease activity by further chemotaxis, tissue damage, and platelet activation. These findings may have implications for future treatment strategies, because complement inhibitors such as the humanized anti-C5 antibody eculizumab have been shown to be effective in complement-mediated diseases such as atypical HUS.49 However, further studies are required because the use of complement inhibitors during active infectious disease may pose a risk by postponing intestinal bacterial clearance.
In summary, this study provides evidence for complement activation on blood cells and blood cell–derived microparticles during EHEC-associated HUS, a process that could be related to contact between Stx and/or O157LPS and blood cells, as shown in vitro. This suggests that complement activation occurs in the circulation during HUS. Although complement activation is not the primary event occurring during EHEC infection, we speculate that it may contribute to blood cell activation, which, combined with the release of microparticles, free radicals, and cytokines, may even contribute to end-organ damage of the kidneys in HUS.
The online version of this article contains a data supplement.
The publication costs of this article were defrayed in part by page charge payment. Therefore, and solely to indicate this fact, this article is hereby marked “advertisement” in accordance with 18 USC section 1734.
Acknowledgments
We thank Professor Mårten Segelmark (Department of Nephrology, Skåne University Hospital Lund) for plasma samples from adult patients and Dr Dominik Turkiewicz (Department of Pediatrics, Skåne University Hospital Lund) for a plasma sample from a child with acute renal failure. We also acknowledge the excellent technical assistance of Ann-Charlotte Kristoffersson (Department of Pediatrics, Lund University) with immunoblotting.
This study was supported by grants from the Swedish Research Council (K2010-65X-14008), by the Torsten and Ragnar Söderberg Foundation, by the Fund for Renal Research, by Crown Princess Lovisa's Society for Child Care, by the Konung Gustaf V's 80-årsfond, by Fanny Ekdahl's Foundation (all to D.K.), and by the Queen Silvia Jubilee Fond (to L.S.). D.K. is also the recipient of a clinical experimental research fellowship from the Royal Swedish Academy of Sciences.
Authorship
Contribution: A.-l.S. designed and performed the research, analyzed data, and wrote the paper; L.S. performed preliminary experiments, analyzed data, and assisted in writing the paper; and D.K. designed the research, analyzed data, and wrote the paper.
Conflict-of-interest disclosure: D.K. was the national coordinator in Sweden of the multicenter trial of culizumab (Alexion Pharmaceuticals) in patients with atypical hemolytic uremic syndrome. The remaining authors declare no competing financial interests.
Correspondence: Diana Karpman, Department of Pediatrics, Lund University, 22185 Lund, Sweden; e-mail: diana.karpman@med.lu.se.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal