Abstract
Heparin derivative-based therapy has evolved from unfractionated heparin (UFH) to low-molecular-weight heparins (LMWHs) and now fondaparinux, a synthetic pentasaccharide. Contrary to UFH or LMWHs, fondaparinux is not neutralized by protamine sulfate, and no antidote is available to counteract bleeding disorders associated with overdosing. To make the use of fondaparinux safer, we developed an antithrombin (AT) variant as a potent antidote to heparin derivatives. This variant (AT-N135Q-Pro394) combines 2 mutations: substitution of Asn135 by a Gln to remove a glycosylation site and increase affinity for heparins, and the insertion of a Pro between Arg393 and Ser394 to abolish its anticoagulant activity. As expected, AT-N135Q-Pro394 anticoagulant activity was almost abolished, and it exhibited a 3-fold increase in fondaparinux affinity. AT-N135Q-Pro394 was shown to reverse fondaparinux overdosing in vitro in a dose-dependent manner through a competitive process with plasma AT for fondaparinux binding. This antidote effect was also observed in vivo: administration of AT-N135Q-Pro394 in 2.5-fold molar excess versus plasma AT neutralized 86% of the anti-Xa activity within 5 minutes in mice treated with fondaparinux. These results clearly demonstrate that AT-N135Q-Pro394 can reverse the anticoagulant activity of fondaparinux and thus could be used as an antidote for this drug.
Introduction
Unfractionated heparin (UFH) and low-molecular-weight heparins (LMWHs) are well-established anticoagulants commonly used to prevent or treat thromboembolic disorders and to prevent clotting of extracorporeal blood during hemodialysis and cardiac surgery. Besides the continuous monitoring required for UFH, both UFH and LMWHs have serious side effects, such as hemorrhage, heparin-induced thrombocytopenia,1 or adverse reactions related to a contaminating product in heparins derived from animal tissues.2 Fondaparinux is a synthetic pentasaccharide whose structure and mechanism of action present several homologies with UFH and LMWH. It will thus be designated as a heparin derivative in the present work. Fondaparinux is the first factor Xa (FXa) selective inhibitor that has received approval for clinical use. It has been evaluated in different clinical trials and appears at least as effective as enoxaparin (a LMWH) for several indications. It also appears safer compared with UFH or LMWHs because of its synthetic origin and its favorable pharmacokinetic profile, which allows a daily injection without continuous monitoring. Furthermore, only few rare cases of thrombocytopenia under fondaparinux treatment were reported, with controversy on a direct causal relationship between the drug and the symptoms in one case.3-5
As with every anticoagulant, heparin derivatives are associated with a bleeding risk, and with fondaparinux, this risk is potentiated by its long half-life of approximately 17 hours.6 UFH and LMWHs can be totally or partially reversed by the administration of protamine sulfate, a nonspecific heparin inhibitor, but protamine sulfate is totally inactive toward fondaparinux. Moreover, the clinical use of protamine sulfate is associated with adverse side effects, including anaphylactic reactions,7 and some researchers8,9 have suggested that protamine sulfate may possess intrinsic anticoagulant properties. Indeed, the administration of excess protamine to neutralize UFH in cardiac surgery has been associated with increased bleeding.10 Thus, a specific and effective antidote to fondaparinux and other heparin derivatives would be helpful in overdose situations and to neutralize excess of heparins after extracorporeal blood circulation.
The anticoagulant activity of heparins relies on their ability to bind antithrombin (AT), a physiologic inhibitor of major procoagulant proteases such as FXa and thrombin. Binding of heparin derivatives to AT increases its anticoagulant activity by several orders of magnitude.11 Once AT has formed a complex with proteases, heparin derivatives are released and can further catalyze the inhibition of proteases by other AT molecules. To develop a specific antidote for all heparin derivatives, we designed an AT variant devoid of anticoagulant activity that exhibits a greater affinity for heparin derivatives than plasma AT (pAT). Such a variant is expected to compete with circulating pAT for heparin binding, neutralizing an excess of anticoagulant in an inert complex.
Inhibition of proteases by AT involves interaction of the inhibitor reactive center loop (RCL) within the protease active site, cleavage of the RCL, and irreversible complex formation. The conformation and amino acid sequence surrounding the scissile bond on the RCL (especially Arg393 and Ser394 at positions P1 and P1′, respectively, according to Schechter and Berger's nomenclature12 ) are critical for protease inhibition. Thus, to abolish AT anticoagulant activity, a Pro residue was inserted between Arg393 and Ser394, inducing a twist on the RCL able to prevent interaction with the target protease. Trypsin-like proteases are also unable to cleave before a proline in P1′ position.13 Moreover, interaction with heparin derivatives depends on the glycosylation state of AT.14 To gain affinity for heparin derivatives and thus increase the antidote potential of AT variant, Asn135 was substituted with Gln to block N-glycosylation.15 Consequently, the AT variant (designated as AT-N135Q-Pro394), designed as an antidote for heparin derivatives, bears 2 point mutations: insertion of a Pro in position 394 and Asn135Gln substitution. We describe herein the production of this AT variant and demonstrate its ability to reverse the anticoagulant activity of heparin derivatives.
Methods
Preparation of recombinant ATs
The shuttle vector pENTR (Invitrogen) carrying full-length AT cDNA (pENTR-AT) was prepared from pCMV6-AT (Origene clone TC110831). Mutagenesis was performed with the use of the QuickChange II Site-Directed Mutagenesis Kit according to the manufacturer's recommendations (Agilent Technologies). First, substitution of codon Asn135 by codon Gln was performed with pENTR-AT as a template to generate pENTR-AT-N135Q, and then a Pro codon was inserted between codons Arg393 and Ser394 to prepare vector pENTR-AT-N135Q-Pro394. Both cDNAs were transferred into eukaryote expression vector pCDNA 3.2 by recombination by the use of Gateway LR Clonase II Enzyme Mix (Invitrogen). The final expression constructs were verified by sequencing.
HEK-293 cells (LGC Standards), grown in “DMEM/F-12” containing 2mM L-glutamine, 100 U/mL penicillin, 100 μg/mL Streptomycin, and 5% fetal bovine serum (Invitrogen), were transfected with 20 μg of plasmid DNA by calcium-phosphate coprecipitation. Stable expression cell lines were selected by G418 and screened for AT secretion by enzyme-linked immunosorbent assay and Western blotting (Human Antithrombin BioAssay ELISA Kit; Euromedex). For both mutated ATs, a single stable expression clone was expanded into “cell factories nunclon” (Nunc) as previously described.16 Conditioned media, harvested daily, were treated with 5mM benzamidine and 5mM ethylene diamine tetraacetic acid (EDTA) and stored at −20°C after contrifugation. AT variants were purified from conditioned media by heparin affinity on a HiPrep Heparin FF 16/10 column (GE Healthcare) equilibrated with 20mM phosphate buffer, 0.4M NaCl, and 0.1mM EDTA, pH 7.4. Nonspecifically bound proteins were eluted in the same buffer containing 0.8M NaCl, and high-affinity AT was step-eluted in the same buffer containing 2M NaCl. This latter fraction was loaded on a Hiload 16/60 superdex 200 column (GE Healthcare) equilibrated in 20mM phosphate buffer, 0.02M NaCl, and 0.1mM EDTA, pH 7.4, to decrease salt concentration. Fractions between 75 mL and 95 mL after injection were pooled and then applied on a 1-mL resource Q column (GE Healthcare) equilibrated in the same buffer. AT was eluted with a linear gradient from 0.02M to 0.5M NaCl and concentrated by ultrafiltration with a 30 000-Da cutoff membrane. The AT concentration was estimated by absorbance at 280nM with an absorption coefficient ϵ = 0.65 g−1.L.cm−1, and the integrity of purified AT was tested by electrophoresis on 10% acrylamide/bisacrylamide gel in native and denaturing conditions followed by silver staining. Finally, AT preparations were aliquoted and stored at −80°C before use for functional assay.
Kinetics of proteases inhibition by ATs
The second-order rate constants (kon) for inhibition of FXa and thrombin (Enzyme Research Laboratories) were measured in the presence of a saturating concentration of fondaparinux (Arixtra; GSK) or UFH (heparin sodium; Panpharma) under pseudo first-order conditions in a continuous or discontinuous assay.17 All experiments were performed at 25°C in kinetic buffer (20mM phosphate buffer, 0.15M NaCl, 0.1mM EDTA, and 0.1% polyethylene glycol-8000, pH 7.4). With pAT or AT-N135Q, inhibition kinetics were initiated by the addition of enzyme (2nM) to a prewarmed mix containing AT (20-200nM), fondaparinux (200nM), or UFH (1 U/mL) and chromogenic substrate (200μM, S2238 for thrombin or S2765 for FXa; Instrumentation laboratory). Substrate hydrolysis was measured at 405 nm, and the kon values were obtained by fitting the data to the equation for slow-binding inhibition and corrected for the competition introduced by the substrate.18 For AT-N135Q-Pro394, kon values were estimated in a discontinuous assay in which FXa or thrombin (100nM) were incubated with AT (5μM) in the presence of fondaparinux (10μM) or UFH (10 U/mL) for a period of time ranging from 10 minutes to 24 hours. Residual FXa or thrombin activity was then measured by 20-fold dilution of the previous mixture in S2765 or S2238 (200μM). Absorbance at 405 nm was recorded, and the kon values were estimated by fitting the substrate hydrolysis initial rate curve as a function of incubation time to one phase exponential decay equation.
Affinity of ATs for fondaparinux
Dissociation equilibrium constants (KD) were estimated by monitoring AT intrinsic fluorescence enhancement upon fondaparinux binding with the use of a LS50B spectrofluorimeter (PerkinElmer Life Sciences) with excitation and emission wavelengths of 280 (slit 3) and 340 (slit 5) nm, respectively.19 The reactions were performed in kinetic buffer in the presence of 100nM AT and fondaparinux (0-400nM), and data were fitted to the equilibrium binding equation by nonlinear regression assuming a stoichiometry of 1. Before the estimation of KD, heparin binding sites were titrated in AT samples with the use of AT 1μM and fondaparinux 0-10μM. Data were recorded with excitation and emission slits of 3 nm and fitted to the same equation. Plasma-derived AT (Aclotine, LFB) was used as a positive control.
Evaluation of heparin derivative reversal properties of AT-N135Q-Pro394 in plasma
Antidote potential of AT-N135Q-Pro394 to heparin derivatives was first evaluated by its ability to decrease heparin-induced anti-FXa activity with the Sta-Rotachrom Heparin assay (Diagnostica-Stago) in a pool of citrated normal human plasma. Plasma was supplemented with UFH (0.05-1.8 U/mL), LMWH (enoxaparin [Lovenox], 0.1-3 U/mL; Sanofi-Aventis), or fondaparinux (0.1-3 mg/L) and incubated with AT-N135Q-Pro394 (1:1 volume ratio) in kinetic buffer. After 5 minutes, anti-FXa activity was measured following the manufacturer's recommendations. With the use of a modified Sta-stachrom ATIII assay (Diagnostica-Stago), antidote potential of AT-N135Q-Pro394 was also evaluated by its ability to inhibit anti–thrombin activity. In brief, plasma supplemented with UFH was incubated with AT-N135Q-Pro394 (1:1 volume ratio) and then diluted in chromogenic substrate. The reaction was then initiated by the addition of bovine thrombin diluted in kinetic buffer. Absorbance increase at 405 nm was measured, and anti–thrombin activity was compared to a standard curve established with varying UFH concentrations.
AT-N135Q-Pro394 antidote activity in mice
The antidote activity of AT-N135Q-Pro394 was evaluated in Swiss female albino mice (Janvier). Animal experiments were approved by the University of Paris ethical committee and performed in accordance with the European guidelines for animal experimentation. Twelve mice, 8 for placebo (phosphate-buffered saline [PBS]; Invitrogen) and 4 for AT-N135Q-Pro394, were included in this pilot study. The mean body weight of animals was 20 ± 2 g, and mice were randomly divided in 2 groups. Each mouse was first anesthetized with sodium pentobarbital administered intraperitoneally and then received fondaparinux, 0.5 mg/kg (subcutaneous administration). After 10 minutes, placebo (PBS) or AT-N135Q-Pro394 diluted in PBS was injected in the retro-orbital plexus. Five minutes later, blood was withdrawn by vena cava puncture with the use of syringes containing trisodium citrate. Plasma was obtained by immediate centrifugation for 15 minutes at 2300g and stored at −40°C until analysis. Anticoagulant activity of fondaparinux in mouse plasma was evaluated by measuring anti-FXa activity with the Sta-Rotachrom heparin assay as described previously in the section.
Results
Anticoagulant activity of AT variants compared with pAT
To evaluate impact of the mutations on anticoagulant activity, second-order rate constants (kon) for inhibition of FXa or thrombin by AT variants or pAT were estimated in the presence of saturating heparin derivative concentration (UFH and fondaparinux were used to catalyze thrombin and FXa inhibition, respectively). Protease inhibition rate constants were found comparable for AT-N135Q and pAT (Table 1), suggesting that the procedure for production of recombinant AT did not significantly impair AT anticoagulant activity. However, inhibition rate constants of FXa and thrombin by AT-N135Q-Pro394 were barely measurable, even at high AT concentration, and found at least 3 orders of magnitude lower than kon values measured with AT-N135Q. This finding showed that protease inhibition reactions were too slow, even in the presence of fondaparinux or UFH. Therefore, as expected, AT-N135Q-Pro394 was mostly devoid of anticoagulant activity.
Second-order rate constants (kon) of FXa or thrombin inhibition by pAT and AT variants in the presence of indicated heparin derivative.
| Antithrombin . | kon (M−1.s−1) ± SEM . | |
|---|---|---|
| FXa + fondaparinux . | Thrombin + UFH . | |
| pAT | (1.67 ± 0.11) × 105 | (1.02 ± 0.09) × 107 |
| AT-N135Q | (2.20 ± 0.07) × 105 | (1.38 ± 0.16) × 107 |
| AT-N135Q-Pro394 | 32.6 ± 4.4 | 15.2 ± 2.5 |
| Antithrombin . | kon (M−1.s−1) ± SEM . | |
|---|---|---|
| FXa + fondaparinux . | Thrombin + UFH . | |
| pAT | (1.67 ± 0.11) × 105 | (1.02 ± 0.09) × 107 |
| AT-N135Q | (2.20 ± 0.07) × 105 | (1.38 ± 0.16) × 107 |
| AT-N135Q-Pro394 | 32.6 ± 4.4 | 15.2 ± 2.5 |
Fast inhibition rate constants (kon > 104 M−1.s−1) were estimated in a continuous assay, whereas slow inhibition rate constants (kon < 104 M−1.s−1) were estimated in a discontinuous assay as described in lines 1 and 2. Errors represented the SEM from 2 or more experiments.
AT indicates antithrombin; FXa, factor Xa; pAT, plasma AT; and UFH, unfractionated heparin.
Affinity of plasma and recombinant ATs for fondaparinux
Olson et al15 have shown that AT N-glycosylation interferes with heparin derivative binding and that removal of the Asn135 glycosylation site increases AT heparin affinity. Fondaparinux affinity of both AT variants was compared with pAT. Concentration of each AT samples (pAT, AT-N135Q, and AT Pro394-N135Q) estimated by heparin binding site titration was found similar to AT concentration estimated by absorbance at 280 nm (data not shown). KD assays were performed with 100nM AT (as estimated by heparin binding site titration) and fondaparinux concentrations ranging from 0 to 400 nM (Table 2). Fondaparinux bound to pAT with a KD = 22.0 ± 3.4nM, which is comparable with published data,20 whereas its binding to AT-N135Q or AT-N135Q-Pro394 was approximately 3-fold greater (KD = 8.6 ± 1.4 nM, and 8.3 ± 1.3 nM for AT-N135Q and AT-N135Q-Pro394, respectively). As expected, Asn135Gln substitution leads to an increased affinity for fondaparinux, but the Pro394 insertion has no effect on fondaparinux binding.
(KD) of pAT and AT variants with fondaparinux binding
| Antithrombin . | KD ± SEM, nM . |
|---|---|
| pAT | 22.0 ± 3.3 |
| AT-N135Q | 8.6 ± 1.4 |
| AT-N135Q-Pro394 | 8.3 ± 1.3 |
| Antithrombin . | KD ± SEM, nM . |
|---|---|
| pAT | 22.0 ± 3.3 |
| AT-N135Q | 8.6 ± 1.4 |
| AT-N135Q-Pro394 | 8.3 ± 1.3 |
KD were estimated by intrinsic fluorescence enhancement of AT (100nM) upon fondaparinux (0-400nM) binding, assuming a stoechiometry of 1:1.
AT indicates antithrombin; KD, dissociation constants; and pAT, plasma AT.
Evaluation of antidote potential of AT-N135Q-Pro394 in human plasma
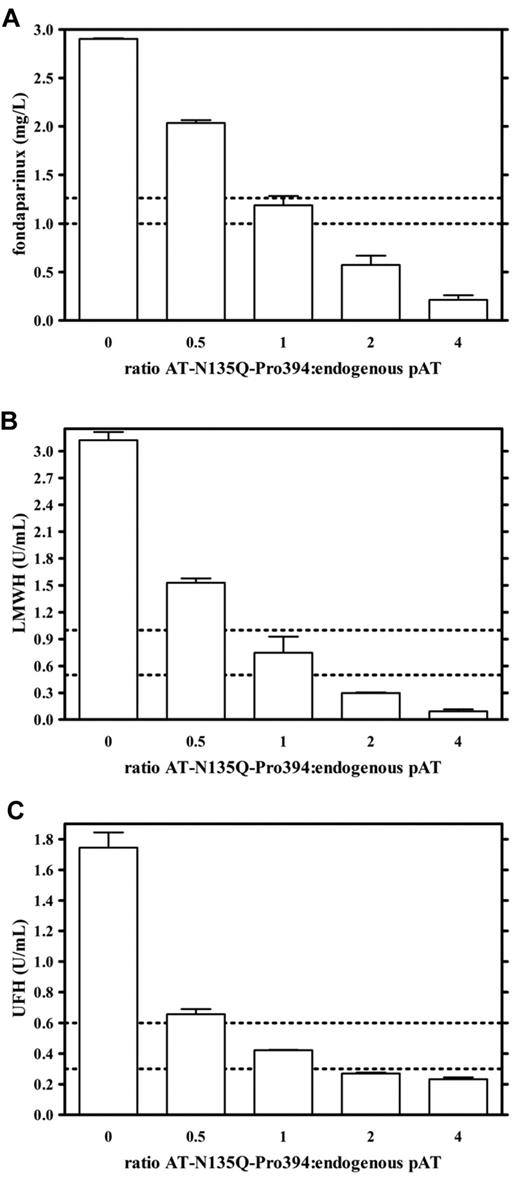
AT-N135Q-Pro394 was tested for its ability to restore a normal coagulability level in human plasma overdosed with UFH, LMWHs, or fondaparinux. During curative treatment of venous thromboembolic disease, optimal fondaparinux concentration ranges between 1.0 mg/L and 1.26 mg/L in plasma.21 To mimic an overdose, fondaparinux was added to a human plasma pool at a concentration of 3 mg/L. Anticoagulant activity was then evaluated by the measurement of anti-FXa activity of fondaparinux-overdosed plasma incubated with mutated AT-N135Q-Pro394 at ratios of 0:1, 0.5:1, 1:1, 2:1, and 4:1 compared with endogenous pAT (with pAT measured at 0.15 g/L). In this ex vivo assay, anti-FXa activity was proportional to the amount of fondaparinux bound to endogenous pAT present in tested plasma (Figure 1A). AT-N135Q-Pro394, at a concentration equal to the endogenous pAT concentration, was able to significantly decrease anti-FXa activity to the level of a curative treatment (from 3 mg/L to 1.23 ± 0.10 mg/L). When AT-N135Q-Pro394 was tested at a 2- or 4-fold molar excess compared with endogenous pAT, fondaparinux concentration decreased to 0.59 ± 0.10 mg/L or 0.22 ± 0.05 mg/L, respectively.
Evaluation of antidote effect of AT-N135Q-Pro394 in heparin derivative–overdosed plasma on the basis of anti-FXa activity measurement. Antidote potential of AT-N135Q-Pro393 was estimated according to its ability to decrease heparin-induced anti-FXa activity in plasma containing fondaparinux 3 mg/L (A), LMWH 3 U/mL (B), or UFH 1.8 U/mL (C). These heparin derivative concentrations correspond to approximately 3-fold the maximal curative dose. AT-N135Q-Pro394 was added to tested plasma at 0, 75, 150, 300, and 600 mg/L, which corresponded to ratios variant to pAT of 0, 0.5, 1, 2, and 4, respectively. Dashed lines indicate maximal and minimal curative concentration for each heparin derivative.
Evaluation of antidote effect of AT-N135Q-Pro394 in heparin derivative–overdosed plasma on the basis of anti-FXa activity measurement. Antidote potential of AT-N135Q-Pro393 was estimated according to its ability to decrease heparin-induced anti-FXa activity in plasma containing fondaparinux 3 mg/L (A), LMWH 3 U/mL (B), or UFH 1.8 U/mL (C). These heparin derivative concentrations correspond to approximately 3-fold the maximal curative dose. AT-N135Q-Pro394 was added to tested plasma at 0, 75, 150, 300, and 600 mg/L, which corresponded to ratios variant to pAT of 0, 0.5, 1, 2, and 4, respectively. Dashed lines indicate maximal and minimal curative concentration for each heparin derivative.
Anticoagulant activity determined on the basis of anti-FXa activity was also evaluated in plasma containing UFH and LMWs at high doses. To mimic an overdose, UFH was added to human plasma at a concentration of 1.8 U/mL and LMWHs at 3 U/mL. These concentrations correspond to 3 times the maximal curative concentration. A dose-dependent antidote activity was observed: At a concentration equal to the endogenous pAT concentration, AT-N135Q-Pro394 decreased heparin anticoagulant activity of 76% in both LMWH- or UFH-overdosed plasma (Figure 1B,C). This result means that AT-N135Q-Pro394 acts as a potent antidote to all heparin-like anticoagulants, decreasing a 3-fold overdose within the therapeutic concentration range when added at a ratio 1:1 compared with endogenous pAT.
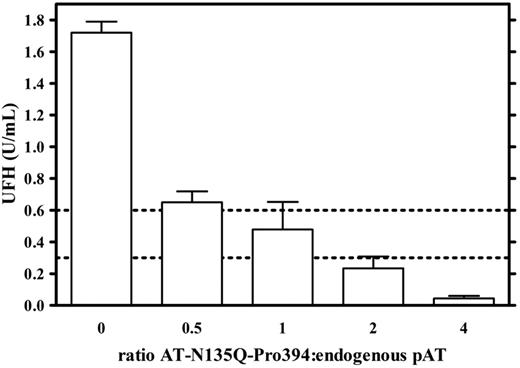
To confirm the antidote activity of AT-N135Q-Pro394, its ability to restore normal anti–thrombin activity was also evaluated in plasma containing UFH at 1.8 U/mL, that is, 3 times the maximal therapeutic dose. As observed previously, the addition of AT-N135Q-Pro394 reduced UFH anticoagulant activity in plasma from 1.8 to 0.48 ± 0.17 U/mL, 0.23 ± 0.07 U/mL, and 0.04 ± 0.02 U/mL when added at 1-, 2-, and 4-fold endogenous pAT concentration, respectively (Figure 2). Taken together, these data strongly suggest that AT-N135Q-Pro394 could be used as a reversal agent of all heparin derivatives.
Evaluation of antidote effect of AT-N135Q-Pro394 in UFH-overdosed plasma on the basis of anti–thrombin activity measurement. Antidote efficacy of AT-N135Q-Pro394 was estimated in UFH (1.8 U/mL) containing plasma for its ability to decrease heparin-induced anti–thrombin activity. AT variant was added to tested plasma at 0, 75, 150, 300, and 600 mg/L, which corresponded to ratios variant to pAT of 0, 0.5, 1, 2, and 4, respectively. Dashed lines indicate maximal and minimal curative dose of UFH.
Evaluation of antidote effect of AT-N135Q-Pro394 in UFH-overdosed plasma on the basis of anti–thrombin activity measurement. Antidote efficacy of AT-N135Q-Pro394 was estimated in UFH (1.8 U/mL) containing plasma for its ability to decrease heparin-induced anti–thrombin activity. AT variant was added to tested plasma at 0, 75, 150, 300, and 600 mg/L, which corresponded to ratios variant to pAT of 0, 0.5, 1, 2, and 4, respectively. Dashed lines indicate maximal and minimal curative dose of UFH.
The antidote potential of this AT variant also depends on its in vivo biodisponibility. Indeed, with a stronger affinity for heparin derivatives, AT-N135Q-Pro394 could also bind more tightly than endogenous pAT to heparan sulfate exposed on the endothelial cell surface. To evaluate the role of blood-vessel endothelium on AT variant trapping, ability of AT-N135Q-Pro394 to decrease anti-FXa activity in plasma overdosed with fondaparinux was determined after preincubation on a human umbilical vein endothelial cell (HUVEC) surface. This preincubation on the cell surface minimally influenced the effect of AT variant because at a ratio of 1:1, 2:1, and 4:1 to pAT, AT-N135Q-Pro394 decreased fondaparinux from 3 mg/L to 1.69 ± 0.08 mg/L, 0.94 ± 0.1 mg/L, and 0.27 ± 0.04 mg/L, respectively (compared with 1.23 ± 0.10 mg/L, 0.59 ± 0.10 mg/L, and 0.22 ± 0.05 mg/L in the absence of HUVEC). This finding suggests that AT-N135Q-Pro394 should remain available to compete with endogenous pAT in vivo. However, in the present experimental conditions, the ratio between reaction mixture volume (50 μL) and cell surface in a 96-well plate coated with HUVECs (0.3 cm2) mimics a rather large blood vessel. In microcirculation, the volume/surface ratio could be much lower and antidote trapping by heparan-sulfate could influence its bioavailability.
Evaluation of antidote potential of AT-N135Q-Pro394 in vivo
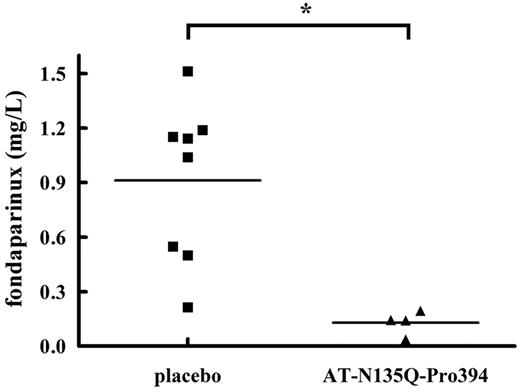
The antidote activity of AT-N135Q-Pro394 was finally evaluated in vivo in mice treated with fondaparinux. Because of a limited supply of the AT variant, only a small number of assays could be performed. A bolus of AT-N135Q-Pro394 (n = 4) or PBS (n = 8) was injected into the retro-orbital plexus of mice 10 minutes after a subcutaneous administration of 0.5 mg/kg fondaparinux corresponding to 5-fold the curative dose in humans. AT-N135Q-Pro394 was administered at a ratio of 2.5:1 to pAT, considering that endogenous pAT concentration in mice is 0.15 g/L (not shown). At this concentration, AT-N135Q-Pro394 treatment resulted in a significant decrease in anti-FXa activity in treated mice. Indeed, 5 minutes after the intravenous administration of the antidote, fondaparinux concentration was estimated at 0.13 ± 0.03 mg/L in antidote-treated animals versus 0.91 ± 0.16 mg/L in untreated animals. This finding corresponds to a decrease in anti-FXa activity of 86% ± 4% (Figure 3). These results clearly demonstrate that, in vivo, the AT variant could reverse the anticoagulant activity of fondaparinux and thus could also be used as an antidote in humans.
Evaluation of antidote effect of AT-N135Q-Pro394 in mice receiving a high dose of fondaparinux. In vivo antidote activity of AT-N135Q-Pro394 (∼ 20 μg/g) versus placebo (PBS) was estimated in mice pretreated with fondaparinux (0.5 mg/kg). Five minutes after intravenous administration of the antidote, blood was withdrawn by puncture in the vena cava, and plasma was obtained by a centrifugation. Anticoagulant activity of fondaparinux was evaluated in plasma of AT-N135Q-Pro394 or placebo-treated mice by measurement of anti-FXa activity as described for heparin derivative reversal properties of AT-N135Q-Pro 394 in plasma. Means ( ± SD) in each group were compared by unpaired t test and found significantly different with P < .05.
Evaluation of antidote effect of AT-N135Q-Pro394 in mice receiving a high dose of fondaparinux. In vivo antidote activity of AT-N135Q-Pro394 (∼ 20 μg/g) versus placebo (PBS) was estimated in mice pretreated with fondaparinux (0.5 mg/kg). Five minutes after intravenous administration of the antidote, blood was withdrawn by puncture in the vena cava, and plasma was obtained by a centrifugation. Anticoagulant activity of fondaparinux was evaluated in plasma of AT-N135Q-Pro394 or placebo-treated mice by measurement of anti-FXa activity as described for heparin derivative reversal properties of AT-N135Q-Pro 394 in plasma. Means ( ± SD) in each group were compared by unpaired t test and found significantly different with P < .05.
Discussion
In the present work we designed and validated the use of an AT variant as an antidote to all heparin derivatives, including fondaparinux. This variant was devoid of anticoagulant activity and bound heparin derivatives with a better affinity than pAT to capture an excess of anticoagulant in an inert complex by a competition mechanism.
Many recombinant or natural variants exhibiting a decreased activity have been described. Most of them exhibit a mutation on the AT RCL that impairs interaction of the AT RCL within the protease catalytic center, resulting in a decreased AT anticoagulant activity.22 The design of an AT variant able to act as an antidote to heparin derivatives requires a dramatic loss of anticoagulant activity to prevent any amplification of the bleeding disorder when administered at high concentrations. The choice of a Pro residue insertion in P1′ position was determined as the result of protease catalytic groove specificity studies. Trypsin-like enzymes exhibit an extreme selectivity of S1 and S1′ subsites with a clear preference for P1-Arg, whereas P1′-Pro is known to prohibit proteolytic cleavage.13 AT-N135Q-Pro394 exhibits an antithrombin or anti-FXa inhibitory activity < 0.001% of pAT activity. This indicates that P1′-Pro insertion is more deleterious than the P1-Arg to His substitution as in natural variant AT Glasgow23 and comparable with P1-Arg deletion in AT London24 in terms of abolition of AT anticoagulant activity. Even if saturated with heparin derivatives, AT-N135Q-Pro394 is unable to neutralize half of thrombin or FXa generation in less than 12 hours/μM. This AT variant can thus be safely used as a heparin antidote with no risk to increase bleeding.
Another aim was to increase the affinity of AT variant for heparin derivatives to favor their trapping in an inert complex. AT-N135Q-Pro394 binds to fondaparinux approximately 3 times more tightly than pAT. This gain in affinity is sufficient to observe the expected antidote activity of AT-N135Q-Pro394 but is slightly lower compared with published data. Indeed β-like AT binds heparin derivatives 3.5- to approximately 36-fold stronger than α-AT, depending on whether one considers plasma or recombinant AT, and also depending on the expression system and purification procedure.25,26 Moreover, the gain in affinity for fondaparinux of the AT variant described in this study is entirely attributable to Asn135Gln substitution, whereas Pro394 insertion does not impact fondaparinux binding. It was anticipated that insertion of a Pro residue in position P1′ would disrupt P1-Arg interaction with the AT core and therefore favor “activated” AT conformation, with the loop expelled from β-sheet A. This “activated” conformer is known to bind heparin with a stronger affinity.23,27 This was not observed, suggesting that Pro394 insertion might induce distortions in AT-N135Q-Pro394 that counteract with the gain in affinity for heparins.
Because of a limited capacity of the recombinant AT production process described in the present work, the in vivo experiments were performed at a single dose of fondaparinux and antidote. However, the results clearly show that AT-N135Q-Pro394 can neutralize fondaparinux in vivo. This strongly supports the concept of an inactive AT variant as antidote to heparin derivatives but the quantity of AT produced is currently incompatible with the management of preclinical and clinical evaluation of the antidote. Interestingly, a large-scale production process of AT in transgenic goat milk was recently developed,28 and the resulting wild-type transgenic AT (Atryn) was approved by the Food and Drug Administration for a therapeutic use29 in patients with inherited AT deficiency. Notably, Atryn already has a 4-fold greater affinity for heparin than pAT,30 a difference that could be increased even more with N135Q mutation. Pharmacokinetic characteristics of transgenic wild-type AT are already available in humans.31 Transgenic wild-type AT has an anticoagulant activity comparable with pAT but shows a shorter half-life compared with pAT. The process for large-scale AT production can be developed for AT-N135Q-Pro394, allowing the future determination of its pharmacologic and pharmacokinetic properties.
Elevated fondaparinux concentrations in plasma may be related to impaired renal function or the treated patient having a low body weight.32 Under those circumstances, the accumulation of fondaparinux could likely be neutralized by the administration of an AT variant at a tolerable concentration. In very rare cases, overdose may originate from a misadministration of the drug, and fondaparinux may reach a much greater plasma concentration than pAT so that neutralization of the anticoagulant activity would require very high doses of antidote. Toxicity and maximal dose of antidote that can be administered remain to be determined. It will be particularly important to determine whether the administration of such an AT variant is susceptible to inducing an immunogenic reaction. Although few changes were performed in the amino acid sequence of AT, we cannot exclude an immunization against AT-N135Q-Pro394 of patients exposed to the antidote. The lack of glycosylation induced by the N135Q mutation mimics the natural β isoform of AT and should not induce an immunogenic response. However, consequences of Pro insertion in position 394 of the RCL are hardly predictable, and the immunogenicity of the antidote remains to be determined.
In the actual context of oral anticoagulant development, the availability of an antidote to fondaparinux may be an asset. Indeed, fondaparinux would become the first direct anti-FXa anticoagulant drug having an antidote to secure its use, giving this drug an advantage compared with oral direct FXa inhibitors. The difficulty in neutralizing fondaparinux activity in the event of side effects or overdose was anticipated for the development of a long-acting pentasaccharide, idraparinux. Idraparinux was synthesized coupled to a biotin moiety, allowing neutralization by avidin.33 However, neither idraparinux nor biotinylated idraparinux has been approved for clinical use, leaving fondaparinux as the only anticoagulant pentasaccharide available. In a previous report, authors demonstrated that recombinant factor VIIa is able to reverse the anticoagulant effect of the fondaparinux in healthy volunteers.34 However, such an unspecific approach needs consideration before administration to patients with an increased thrombotic risk. The principal advantage of the antidote presented in this study lies in its specific activity for direct neutralization of heparin derivatives. Hemorrhagic state in case of heparin derivative overdose and antidote administration may be easily controlled by anti-FXa activity monitoring.
Finally, the antidote presented in the present work is specific and effective for all heparin derivatives. In this respect, this antidote is of particular interest to substitute the use of protamine sulfate, which is, so far, considered as the treatment of choice for the management of patients who develop bleeding complications while on UFH.35 Clinical use of protamine is associated with significant adverse side effects, and in practice this drug is unwieldy. The present antidote could be an alternative to protamine sulfate for UFH neutralization in involuntary overdosing, but also in cardiopulmonary bypass during cardiac surgery. Indeed, despite many disadvantages, protamine is routinely administered postoperatively to reverse the high concentrations of UFH required for patients undergoing cardiopulmonary bypass36 because no other antidote is available. Furthermore, the development of an antidote to reverse fondaparinux activity could open a new drug application for fondaparinux in anticoagulation of cardiopulmonary bypass during cardiac surgery. Indeed, the cardiac surgical procedure is associated with the a high incidence of the development of antiplatelet factor 4/heparin antibodies,37 and fondaparinux already has been evaluated to ensure anticoagulation of extracorporeal circulation devices during hemodialysis sessions.38,39
The results of the present study indicate that the AT variant AT-N135Q-Pro394 is effective as an antidote toward heparins. Preclinical and clinical evaluation will determine how broadly useful this antidote may be in the management of patients with fondaparinux or other heparin derivative treatments, depending on overdose intensity or hemorrhage severity.
The publication costs of this article were defrayed in part by page charge payment. Therefore, and solely to indicate this fact, this article is hereby marked “advertisement” in accordance with 18 USC section 1734.
Acknowledgments
We thank Sylvain Clauser for proofreading the manuscript.
This work was supported by research grants from FRM (Fondation pour la Recherche Médicale), OSEO, Université Paris Sud, and the Leducq fondation (LINAT). J.F. received a grant from GEHT (Groupe de travail sur l'hémostase et la thrombose).
Authorship
Contribution: E.P.B., V.P., and D.B. designed and supervised research; E.P.B. and J.F. conducted most of the experiments; and E.P.B. and D.B. wrote the manuscript.
Conflict-of-interest disclosure: The authors declare no competing financial interests.
Correspondence: Elsa P. Bianchini, EA4531, Faculté de Pharmacie, 5 rue Jean-Baptiste Clément, 92296 Châtenay-Malabry Cedex, France; e-mail: elsa.bianchini@u-psud.fr.