Abstract
Abstract 1210
Von Willebrand disease (VWD), the most frequently diagnosed bleeding disorder, is characterized by variable expressivity and incomplete penetrance. Bleeding severity in type 1 VWD does not always correlate with plasma VWF levels, except in cases of severe deficiency. It is possible that the phenotypic variability observed in type 1 VWD is related to the final ratio of mutant vs. wild-type (WT) subunits in the mature VWF multimeric structure. The aim of this study was to determine the role of mutant:WT transfection ratio on von Willebrand factor (VWF) expression, secretion and degradation in VWD type 1 mutations.
Type 1 VWD mutations with reported normal multimer distribution were chosen from the D'-D3 region of VWF. Mutations of cysteine residues were eliminated to avoid interference with inter- and intra-chain disulfide linkages. Mutations were generated by performing site-directed mutagenesis on full-length human VWF cDNA within the pcDNA3.1(-)A vector, which appends VWF with a Myc-His tag (denoted mH). The following mutations were generated: M771I, R782Q, R924W, I1094T and T1156M.
Mutant VWF was co-transfected with WT VWF contained within the pCIneo vector (mutant mH:WT pCIneo ratios investigated were 1:3, 2:2, 3:1, 4:0). Recombinant (r)VWF expression was measured using ELISA and concentrations were determined by comparison to a standard curve generated with pooled normal plasma. Multimer composition was analyzed using SDS-agarose gel electrophoresis followed by Western blot. Statistical significance was determined using one-way ANOVA with post-hoc Tukey test.
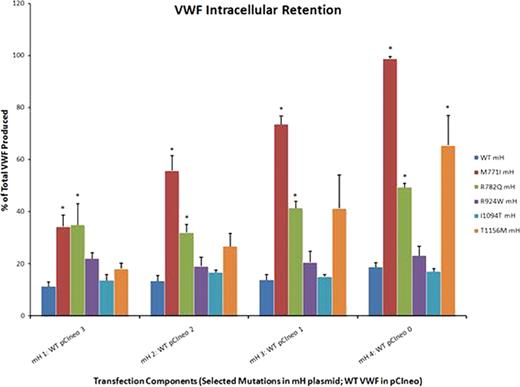
Homozygous expression of R924W or I1094T had no effect on rVWF expression or secretion compared to WT, while M771I, R782Q and T1156M significantly increased intracellular protein retention. Co-expression of M771I or R782Q at varying ratios with WT was able to partially correct rVWF secretion, although intracellular retention remained significantly higher than WT at all ratios (n=3, * p<0.05, Figure 1). Co-expression with WT cDNA was also able to correct T1156M retention in a dose-dependent manner (n=3, Figure 1), as described previously [Lethagen, Thromb and Haemost, 2002]. Multimer analysis of co-transfection supernatants exhibited normal and full distribution of multimers, as expected for type 1 VWD mutations.
Effect of mutant:WT co-transfection ratios on rVWF intracellular retention. *p<0.05 when compared to corresponding WT mH:WT pCIneo value (dark blue bar in graph).
Effect of mutant:WT co-transfection ratios on rVWF intracellular retention. *p<0.05 when compared to corresponding WT mH:WT pCIneo value (dark blue bar in graph).
Others have shown previously that heterodimers of WT and C1149R VWF, a type 1 VWD mutation, are degraded by the proteasome [Bodo et al, Blood, 2001], presumably via recognition of a folding defect within the mutant subunit. In order to determine the role of proteasomal degradation in the decreased secretion levels of our mutants, we performed experiments in the presence of the proteasome inhibitor MG-132. Treatment of co-transfected cells (mutant:WT 2:2) with 1 mM MG-132 for 16 hours prior to harvesting did not significantly affect secretion or overall expression of rVWF, suggesting that this pathway is not involved in the regulation of the expression of our mutants.
Our data demonstrate that M771I, T1156M and R782Q induce a significant increase in intracellular retention compared to WT protein, which could contribute to a quantitative deficiency in type 1 VWD, while R924W and I1094T do not appear to interfere with VWF production or secretion. Variable levels of intracellular retention have been observed in a previous study of VWF mutations identified in type 1 VWD patients [Eikenboom, et al, J Thromb Haemost, 2009]. While one interpretation of these results is that R924W or I1094T may not be causative mutations in type 1 VWD, other mechanisms including protein clearance and function remain to be explored. Although type 1 VWD mutations variably affect expression and secretion levels in vitro, studying platelet rolling on these mutants at a range of physiological shear stresses will provide valuable information regarding whether the degree of incorporation of mutant subunits into VWF multimers can affect supramolecular structure, and ultimately, hemostatic function.
No relevant conflicts of interest to declare.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal