Abstract
Abstract 2036
There are limited numbers of studies focusing on the role of hematopoietic stem cell transplantation (HSCT) as a salvage treatment for acute promyelocytic leukemia (APL) after relapse in the all-trans-retinoic acid (ATRA) era. We retrospectively analyzed the outcomes of APL patients who underwent allogeneic or autologous HSCT during second complete remission (CR2) and compared them with those in APL patients who did not receive HSCT.
A total of 302 adult patients with previously untreated de novo APL were registered in the Japan Adult Leukemia Study Group (JALSG) APL97 study between 1997 and 2002, and treated with ATRA and chemotherapy (Asou et al., 2007). Of 283 assessable patients with t(15;17) and/or PML-RARA, 267 (94.3%) achieved CR and 67 (23.7%) relapsed. Except for 2 patients unable to complete the follow-up survey, 65 relapsed patients received salvage treatment with ATRA alone (n=17), ATRA plus chemotherapy (n=33), chemotherapy alone (n=6), tamibarotene alone (n=7), allogeneic HSCT (n=1), unknown (n=1), and 58 of these patients (89%) achieved CR2. Of the patients who achieved CR2, 27 received HSCT (21 allogeneic and 6 autologous) during CR2, 30 did not receive HSCT, and 1 was not assessable. All 6 patients who underwent autologous HSCT were confirmed of having achieved molecular CR. We compared the survival rates of patients with CR2 achievement who received HSCT (HSCT group) or did not receive HSCT (no-HSCT group). Survival was calculated from the date of CR2 to death or last visit. Probabilities of survival were estimated using the Kaplan-Meier method and compared by the log-rank test.
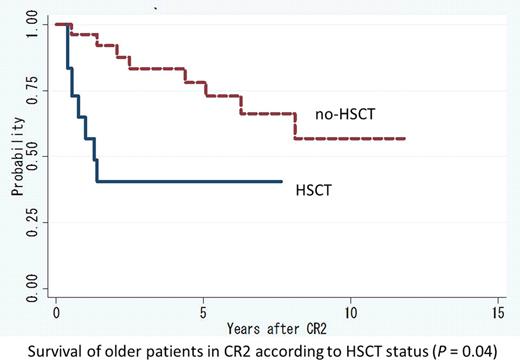
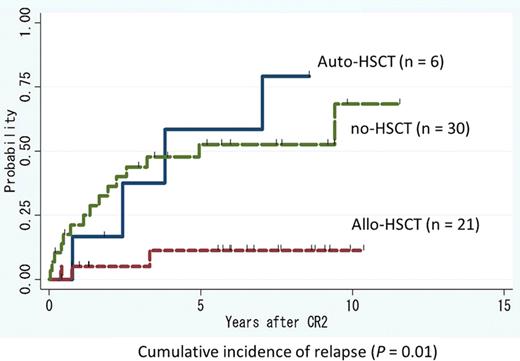
Median age at first relapse was significantly lower in the HSCT group (36 years; range: 22 – 60) than in the no-HSCT group (53 years; range: 16 – 72) (P = 0.006). Median first CR duration was 21.6 months (range: 5.5 – 80.8) in the HSCT group and 17.6 months (range: 5.9 – 90.5) in the no-HSCT group (P = 0.75). Among patients in the no-HSCT group, 6 underwent allogeneic (n = 4) or autologous (n = 2) HSCT during CR3 or more. Five-year survival rate in the HSCT and no-HSCT groups were 70.4% and 77.4%, respectively (P = 0.86). Six patients died within 5 years of CR2 in the HSCT group. The causes of death included complications of allogeneic HSCT (n = 4) and relapse after HSCT (n = 2; 1 autologous, 1 allogeneic). In contrast, 7 patients in the no-HSCT group died of complications of allogeneic HSCT after second relapse or later (n = 4), disease progression (n = 2), and ischemic heart disease (n = 1). Among younger patients (aged ≤39 years), there was no statistically significant difference in 5-year survival between the HSCT group (100.0%) and the no-HSCT group (82.5%) (P = 0.10). In contrast, among older patients (aged ≥40 years), 5-year survival rate was significantly higher in the no-HSCT group (78.0%) than in the HSCT group (40.5%) (P = 0.04) (using the time-dependent covariates). Within the HSCT group, 5-year survival rate was significantly better in younger patients (n = 15) (100.0%) than in older patients (n = 12) (50.0%) (P = 0.006). Among the 27 patients in the HSCT group, there was no significant difference in 5-year survival between patients who underwent allogeneic HSCT (76.2%) and those who underwent autologous HSCT (83.3%) (P = 0.69). However, the 5-year cumulative incidence of relapse was significantly higher in patients who underwent autologous HSCT (58.3%) than those who underwent allogeneic HSCT (11.3%) (P = 0.01). After second relapse, 3 of 4 patients who underwent autologous HSCT at CR2 achieved CR3 through treatment with arsenic trioxide (ATO), tamibarotene or high-dose cytarabine.
This study indicates that allogeneic HSCT is recommended in younger APL patients (aged ≤39 years) during CR2, while autologous HSCT is accompanied by frequent relapses. Our retrospective study also revealed that outcomes were significantly better in elderly patients who did not receive HSCT than those who did during CR2. This may result from a high HSCT-related mortality rate among elderly patients, and suggests a need for appropriate salvage treatment after relapse. We can now use ATO for salvage therapy routinely, allowing for further improvement of relapsing APL outcomes, even for patients not eligible for HSCT.
Takeshita:Takeda: Research Funding; Novaltis: Research Funding. Naoe:Kyowa-Hakko Kirin.: Research Funding; Dainipponn-Sumitomo Pharma.: Research Funding; Chugai Pharma.: Research Funding; Novartis Pharma.: Honoraria, Speakers Bureau; Zenyaku-Kogyo: Research Funding; Otsuka Pharma.: Research Funding.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal