Abstract
Abstract 283
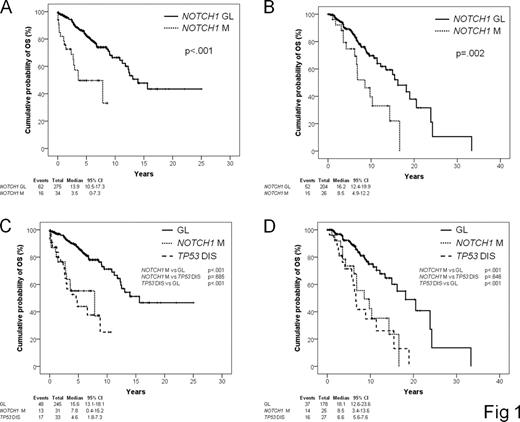
The clinical course of chronic lymphocytic leukemia (CLL) ranges from very indolent, with a nearly normal life expectancy, to rapidly progressive leading to death and occasionally undergoing transformation to Richter syndrome (RS). TP53 disruption identifies a fraction of high risk CLL destined to experience a very short survival. High risk CLL, however, cannot be fully recapitulated by TP53 disruption and other lesions of cancer genes may be implicated in this aggressive phenotype. Analysis of the CLL coding genome has recently disclosed that the NOTCH1 proto-oncogene is recurrently mutated at CLL presentation. Here we assessed the prognostic role of NOTCH1 mutations in CLL. Two series of previously untreated CLL were utilized as training (n=309, median follow-up 6 years) and validation (n=230, median follow-up 7 years) cohorts. NOTCH1 mutations were analyzed by DNA Sanger sequencing in blind with respect to clinical data. In the training series, NOTCH1 mutations occurred in 34/309 (11.0%) patients, being mostly represented (26/34, 76.5%) by a recurrent two bp frameshift deletion (c.7544_7545delCT). The remaining NOTCH1 mutations (8/34, 23.5%) were frameshift deletions other than c.7544_7545delCT (n=7) and frameshift insertions (n=1). All mutations were predicted to disrupt the NOTCH1 PEST domain. CLL with NOTCH1 mutations preferentially carried unmutated IGHV genes (76.5%, p<.001). Other characteristics at presentation associated with NOTCH1 mutations were advanced Rai stage (26.5%, p=.006) and trisomy 12 (44.1%, p<.001). By univariate analysis, NOTCH1 mutations associated with an increase in the hazard of death (HR: 3.77; 95% CI: 2.14–6.66) and a significant overall survival OS shortening (p<.001) (Fig. 1A). Multivariate analysis selected NOTCH1 mutations as an independent risk factor of OS (HR: 4.22; 95% CI: 2.15–8.28; p<.001), after adjusting for age (p<.001), Rai stage (p=.005), IGHV mutation status (p=.465), 11q22-q23 deletion (p=.128), trisomy 12 (p=.183) and TP53 disruption (p<.001). The poor prognosis conferred by NOTCH1 mutations was attributable, at least in part, to a shorter time to progression requiring treatment (p<.001), and a higher cumulative probability of RS development (p=.026). Although NOTCH1 mutated patients were devoid of TP53 disruption in 31/34 (91.2%) cases, the OS predicted by NOTCH1 mutations was similar to that of TP53 mutated/deleted CLL (Fig. 1C). Analysis of the validation series confirmed: i) the prevalence of NOTCH1 mutations at CLL presentation (26/230, 11.3%); ii) the spectrum of NOTCH1 mutations at CLL presentation (c.7544_7545delCT: 21/26, 80.7%; other mutations: 5/26, 19.3%) iii) the adverse prognostic impact of NOTCH1 mutations in CLL both by univariate analysis (Fig. 1B) and by multivariate analysis (HR: 2.08; 95% CI: 1.10–3.93; p=.023); iv) the preferential mutually exclusive distribution of NOTCH1 mutations and TP53 disruption (25/26, 96.2%); v) that OS of NOTCH1 mutated CLL is similarly poor as that of TP53 disrupted CLL (Fig. 1D). The current study on 539 CLL documents that NOTCH1 mutations: i) represent one of the most frequent cancer gene mutations known to be involved at CLL presentation; ii) identify a subgroup of patients showing poor OS similar to that of TP53 disrupted cases; iii) exert a prognostic role independent of widely accepted clinical and genetic risk factors; iv) predict OS in series from different institutions, as documented by the training-validation approach chosen for the design of this study.
Disclosures:
No relevant conflicts of interest to declare.
Author notes
*
Asterisk with author names denotes non-ASH members.
© 2011 by The American Society of Hematology
2011

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal