Abstract
Abstract 4423
Treatment of chronic myeloid leukemia with imatinib leads to disease remission in a majority of patient, but in some patients (pts) controlling the disease remains a challenge. One of the proposed prognostic factors for identifying this subset of pts is the treatment response in the first months of therapy.
We conducted a study to evaluate the importance of the early complete cytogenetic response (CCyR) and the factors associated with its achievement.
This is a retrospective study in a cohort of pts with chronic-phase chronic myeloid leukemia (CP-CML) enrolled in 14 Hematology centers in South Brazil. All pts received imatinib 400mg as first or second-line therapy. Early-imatinib treatment was considered when imatinib started before 12 months (mo) from diagnosis. Patient evaluation and response criteria followed the ELN recommendations. The ACE-27 (Adult Comorbidity Evaluation-27) is a 27 item comorbidity index for patients with cancer and assign weights from 1 to 3 based on the dysfunction grade of each condition (mild, moderate and severe, respectively). An ACE-27 score was applied to each patient. Imatinib suspensions were considered if superior to 20 days at any point during therapy. EFS was measured from the start of imatinib to the date of any of the following events while on therapy: death from any cause, loss of complete hematologic response, loss of complete cytogenetic response, discontinuation of therapy for toxicity or lack of efficacy, or progression to accelerated phase or blastic phase.
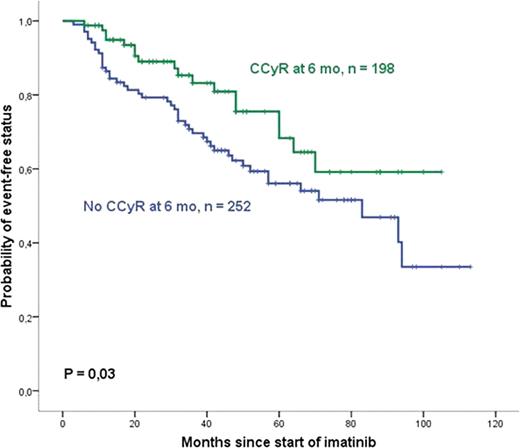
We analyzed data from 450 pts with CP-CML diagnosed since 1990. The median age at diagnosis was 48 yr (4 – 85) and 55% were male. The median time from diagnosis to imatinib was 7 mo (0 – 178) and 71% pts were early-imatinib treated. Prior therapy with interferon was used in 60% pts. The median of follow-up was 47 mo. With 6 months of imatinib therapy, 198 pts (44%) achieved CCyR. In this group, the four year cumulative incidence of events was 33 (17%) and the EFS was 75,5%. 252 (56%) were not in CCyR at 6 months of therapy. In this group, a greater proportion of cumulative of events was observed: 86 (34%), and the EFS was 62,3%. This difference was significant (P=0,03; Figure 1). In this group of pts, 63% achieved CCyR after 6 months any time during follow up and the median time for CCyR in these pts was 17 months. The chance of achieving major molecular response (MMR) during follow up was 79% for the pts with CCyR at 6 months compared to 53% for the group with no CCyR at 6 months (P<0,001). Some factors were associated with reduced chance of CCyR at 6 months. In a multivariate analysis, the pts with late-onset imatinib treatment (more than 12 mo from diagnosis) had a CCyR rate of 31%, in contrast, the pts who started imatinib before 12 mo had a rate of 50% (P=0,02). The pts with good adherence to treatment had greater CCyR rate than those with poor adherence (interruption greater than 20 days), 51,4% and 35%, respectively (P=0,04). Comorbidity measured by ACE-27 score also influenced the CCyR rates at 6 months: 54% of score 0 (no comorbidity) patients achieved CCyR, compared to 30% of pts with score 1 (mild comorbidity), 33% of pts with score 2 (moderate) and 47% of pts with score 3 (severe) (P=0,009). The greater CCyR rate in the severe comorbidity group probably lacks significance due to the reduced number of pts in this group (22).
A great proportion of pts achieve CCyR after 6 months of imatinib therapy, nevertheless, the pts who achieve CCyR by 6 months of therapy have greater proportion of major molecular response and event-free survival. Imatinib therapy should be started as soon as possible and additional efforts must be taken to avoid nonadherence. Finally, special attention should be given to pts with comorbidities as their results tend to be worse.
No relevant conflicts of interest to declare.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal