Abstract
Abstract 1373
The aim of this work was to establish a robust and simple method for the measurement of drug sensitivity in myeloma cells under conditions mimicking aspects of the bone marrow microenvironment. In particular we wanted to measure drug sensitivity in myeloma cells cultivated in the presence of stromal cells.
The tumor microenvironment can profoundly affect tumor cell survival as well as alter antitumor drug activity, and it is generally believed that growth and survival of myeloma cells is critically dependent on the bone marrow microenvironment. Bone marrow stromal cells (BMSC) have been shown to protect myeloma cells from common cytostatic or cytotoxic drugs in vitro. Common in vitro assays used for high-throughput drug screening cannot easily discriminate between stromal and tumor cell responses in co-cultures. Although a few recent studies have overcome this problem (Ramasamy K. et al., 157(5):564–79,2012, McMillin D. et al., 16(4):483–9, 2010), the application of stable transfection for labeling of cells limits the practical application of these co-culture studies to cell lines, excluding primary myeloma cells that inherently may be hard to transduce even by retroviral vectors.
Here, we analyzed survival of myeloma cells co-cultured with BMSC using an automated fluorescence microscope, ScanR. ScanR is a microscope based screening station. By staining the cell nuclei with DRAQ5, we were able to discriminate between BMSC and myeloma cells, based on their staining intensity and nuclear shape. Using the apoptotic marker YO-PRO-1, the effects of drug treatment on the viability of the myeloma cells in the presence of stromal cells could be measured. The main advantages of this method are the non-necessity of cell manipulation before co-culture and the low number of myeloma cells (5000 primary cells) that are needed per measurement, which makes the method ideal for experiments with primary myeloma cells. In fact, the analysis was easier and more robust when using slowly growing cells, i.e. by using primary myeloma cells compared to more rapidly proliferating myeloma cell lines. This method should be well-suited for high throughput analysis, as the cells are stained in situ with no washing, centrifugation, or fixation steps before analysis.
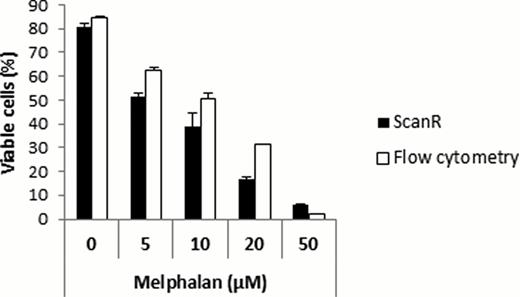
The method was compared to a conventional method for detecting cell viability; flow cytometry where annexin V labeling was used to detect apoptotic cells. As shown in figure 1, the dose-response curves obtained for ANBL-6 cells treated with different doses of melphalan were similar and showed the same trends for both methods. However, the effects of melphalan treatment were more evident analyzed by the ScanR system than by flow cytometry (EC50 YO-PRO-1 = 11μM versus EC50Annexin V= 15μM).
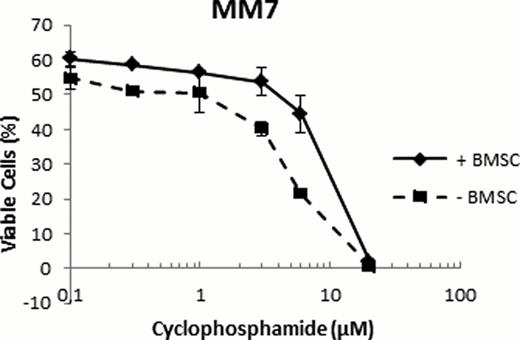
The stromal cell population applied in this study was able to support IL-6 dependent myeloma cell lines without addition of IL-6. This as IL-6 dependent INA-6 cells cultivated in the presence of BMSC survived in the absence of added IL-6. This study shows the importance of stromal cell support for primary myeloma cell survival in vitro, as half of the cell samples had a marked increase in their viability when cultured in the presence of BMSC. Stromal cell-induced protection against common myeloma drugs was also observed with this method. For instance, experiments with primary myeloma cells from patient MM7, showed that in the presence of BMSC, the EC50 for the common myeloma drug cyclophosphamide was increased from 5 μM to approximately 10 μM (figure 2).
No relevant conflicts of interest to declare.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal