Abstract
Abstract 3773
Age at diagnosis of CML varies by race in the United States with median occurring around ages 54 and 63 among Black and White patients, respectively. The treatment paradigm shifted when Imatinib was approved in 2001 for treatment of CML. More recently, second generation tyrosine kinase inhibitors (TKI) have also been used for treatment of CML. Differences in outcomes by race have been previously reported prior to the TKI treatment period. We aimed to assess whether the earlier age at diagnosis resulted in differential trends in age-adjusted incidence rates and survival outcomes by race in the post-Imatinib treatment period.
Data from the Surveillance, Epidemiology, and End Results (SEER) 18 Registries were extracted for diagnoses between 2002 and 2009 based on the assumption that cases diagnosed after 2002 would be treated with TKI's. CML was defined according to the International Classification of Diseases for Oncology 3rd edition code 9863 (CML-NOS) and 9875 (CML-Philadelphia Chromosome Positive). Cases diagnosed by autopsy or death certificate only were excluded. Incidence rates are expressed per 100,000 person-years and age-adjusted to the 2000 US Standard Population. Black/White incidence rate ratios (IRRBW) are shown with corresponding 95% confidence intervals (CI). Kaplan-Meier estimates of CML-specific survival (CPS) and overall survival (OS) were estimated at 5-years post-diagnosis with the event being time to CML-specific death or any death, respectively. Stratified Cox proportional hazards models were constructed to assess the impact of age and race on the risk of death expressed as a hazard ratio (HR).
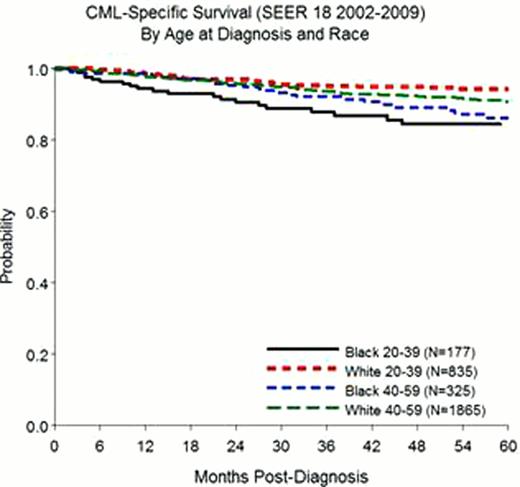
Since 2002, 6,632 patients diagnosed with CML were reported to the SEER 18 registries including 5,829 White patients (87.9%) and 803 Black patients (12.1%) with 57% being male. The age-adjusted incidence rate for Blacks was 1.18 (95% CI, 1.10–1.27) per 100,000 and 1.12 (95% CI, 1.09–1.27) per 100,000 for Whites. The corresponding IRRBW was 1.06 (95% CI, 0.98– 1.14). When considering 20-year age-groups, Blacks had higher incidence rates in the 20–39 and 40–59 age groups; IRRBW of 1.26 (95% CI, 1.06–1.49; p=0.0073) and 1.23 (95% CI, 1.09–1.39; p=0.0007), respectively. No statistically significant differences in IRRBW were seen within the 0–19, 60–79 and 80+ age-groupings although Whites have higher non-significant incidence rates in the latter 2 age-groups. Differences in IRRBW prompted an assessment of survival to determine if the excess incidence observed in the younger age groups corresponded with a worse survival. CPS at 5-years was 85.5% (95% CI, 84.3–86.6). In univariate analysis, age was an important predictor of outcome (p<0.0001) with patients diagnosed after age 80 having the worse outcomes (OS: 58.3%), followed by patients diagnosed between 60 and 79 years (OS 84.7%), 0–19 years (OS: 87.1%), 40–59 years (OS: 90.2%), and 20–39 years (OS: 92.6%). When considering all age-groups, race was not a significant predictor of death (HR 0.91; 95% CI, 0.72–1.15). However, in a stratified analysis with 20-year age groups, Blacks had an increased risk of death as compared to Whites (Figure 1) in the 20–39 age group (HR: 2.94; 95% CI, 1.72–5.26; p<0.0001) and the 40–59 age group (HR: 1.67; 95% CI, 1.22–2.27; p=0.0069) while no differences were seen within the 0–19, 60–79 and 80+ age groups. Conclusions from OS models were similar to that of the CPS models.
Through this analysis of population-based cancer registry data collected in the US between 2002 and 2009, we show that Blacks have a younger age at diagnosis with higher incidence rates observed in the 20–39 and 40–59 age-groups as compared to Whites. Both CPS and OS outcomes differed by race and age. Similar to the differences observed with the incidence rates, survival was worse in Blacks diagnosed within the 20–39 and 40–59 age-groups as compared to Whites. Although outcomes have globally improved in patients with CML since the advent of tyrosine kinase inhibitors, the persistence of incidence heterogeneity and poorer survival among Blacks warrants further attention. Access to care may be a possible reason for the differences observed but further studies are warranted to rule out biological differences which may be causing an earlier age at onset and poorer survival.
No relevant conflicts of interest to declare.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal