Abstract
Abstract 4518
Absolute lymphocyte count (ALC) recovery after autologous stem cell transplantation has been identified as an independent prognostic factor for survival in several hematological malignancies, including multiple myeloma (MM), as well as in some solid tumours. Preclinical studies suggest a role of the host immune system in tumor control and tumor immunosurveillance. In human studies, T cells were shown to have suppressive effects on polyclonal immunoglobulin production in MM patients. These reports suggest an important role of the host immune system in the treatment of cancer. Thus, we set out to investigate if ALC at different time points before and after autologous stem cell transplantation (ASCT) is a predictor of survival in AL amyloidosis patients treated at Princess Margaret Hospital.
Seventy-eight consecutive patients with documented symptomatic AL amyloidosis who underwent ASCT were identified from the Princess Margaret Hospital sysproteinemia database. This study was approved by the Ethics Committee of the Princess Margaret Hospital. The primary endpoint of the study was to assess the impact on overall survival (OS) based on ALC at day +15 and +30 post-ASCT in newly diagnosed AL amyloidosis patients who underwent a single ASCT. The Cox proportional hazard model was used to perform univariate analyses of possible prognostic variables for overall survival, after confirming the proportionality of each variable using time-dependent covariates.
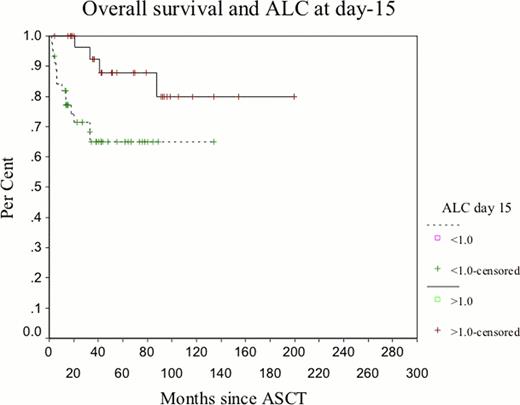
A total of 78 patients with AL amyloidosis underwent ASCT from 01/2004 to 03/2010. Clinical characteristics are shown in Table 1 . Patients received high-dose melphalan at 200 mg/m2 (24.4%), 140 mg/m2 (56.4%) or 100mg/m2 (19.2%) given intravenously on day -1 as per our risk-adapted approach, and stem cells were infused on day 0. At the time of this analysis, 60 patients were still alive and 29 (37%) had experience hematologic progression. Median progression-free survival (PFS) was 26.5 months (23–45) for all patients. The choice of ALC ≥1á0 × 109/L as the cut-off point was supported by the data because it yielded the greatest differential in survival, based on the chi-square χ2 = 100á4, P < 0á0001) analyzed at different cut-off points between the 25% and 75% quartiles from the log-rank test. An ALC of ≥ ≥1á0 × 109/L at day +15 and +30 post-ASCT significantly correlated with a better OS (mean OS of 170 versus 91 months and mean OS of 161 versus median of 33.4 months, respectively) (p=0.017 and p=0.0012) and PFS (median of 34.3 versus 16.4 months) (p=0.027). (Fig 1a-b). Early death and 1-year mortality were seen in 50% and 66.6% of cases with an ALC of <1á0 × 109/L at day +15 and +30 versus 0% and 12.5% in those patients with ALC ≥1á0 × 109/L (p=0.016 and 0.025, respectively). Multivariate analysis identified an ALC at day +15 and +30 as independent prognostic factors for OS while age > 60, B2-microglobulin > 460 μmol/L, LDH > 350 IU/L, CRP > 20mg/L, albumin < 35g/L, and creatinine>200 μmol/L were not associated with survival.
In conclusion, ALC is a surrogate marker of the host immune system that correlates with better survival in AL amyloidosis patients undergoing a single ASCT. Immune reconstitution studies post-ASCT have demonstrated delayed recovery of T and B cells. However, normal numbers and function of NK cells have been documented as early as 2 weeks and these cells have been speculated as leading to a better outcome. Previously, it was reported that the number of infused NK cells along with the stem cells correlate with ALC day +15 recovery. The role of vaccination strategies and cellular therapies in AL amyloidosis should be investigated based on these findings.
Clinical and Laboratory characteristics of patients with AL Amyloidosis undergoing ASCT
| Clinical Characteristics . | N . | Median . | Range . | % . |
|---|---|---|---|---|
| Age (years) | 78 | 57 | 33–74 | |
| Gender | ||||
| Male | 53 | 68% | ||
| Female | 25 | 32% | ||
| IgG Kappa | 5 | 6.5% | ||
| IgG Lambda | 20 | 26% | ||
| IgA Lambda | 4 | 5.1% | ||
| IgM Kappa | 2 | 2.7% | ||
| IgM Lambda | 2 | 2.7% | ||
| Free Kappa | 10 | 13% | ||
| Free Lambda | 34 | 44% | ||
| Renal Involvement | 56 | 71.8% | ||
| Cardiac Involvement | 37 | 47.4% | ||
| 3 or more organs involved by AL | 16 | 20.5% | ||
| GI involvement | 8 | 10.3% | ||
| Hemoglobin (g/L) | 78 | 138 | 104–182 | |
| Creatinine (μmol/L) | 78 | 129 | 37–700 | |
| Absolute Lymphocyte count | 78 | |||
| Day0 | 1.9 | 0.2–4.4 | ||
| Day+15 | 1.03 | 0.3–5.9 | ||
| Day+30 | 1.8 | 0–5.5 | ||
| Albumin g/L | 78 | 35.1 | 25–58 | |
| B2-microglobulin (μmol/L) | 78 | 348 | 70–2900 | |
| 24 Hr Proteinuria (g/d) | 78 | 5.6 | 0–24.9 | |
| ***BMPC (%) | 78 | 8 | 1–30 |
| Clinical Characteristics . | N . | Median . | Range . | % . |
|---|---|---|---|---|
| Age (years) | 78 | 57 | 33–74 | |
| Gender | ||||
| Male | 53 | 68% | ||
| Female | 25 | 32% | ||
| IgG Kappa | 5 | 6.5% | ||
| IgG Lambda | 20 | 26% | ||
| IgA Lambda | 4 | 5.1% | ||
| IgM Kappa | 2 | 2.7% | ||
| IgM Lambda | 2 | 2.7% | ||
| Free Kappa | 10 | 13% | ||
| Free Lambda | 34 | 44% | ||
| Renal Involvement | 56 | 71.8% | ||
| Cardiac Involvement | 37 | 47.4% | ||
| 3 or more organs involved by AL | 16 | 20.5% | ||
| GI involvement | 8 | 10.3% | ||
| Hemoglobin (g/L) | 78 | 138 | 104–182 | |
| Creatinine (μmol/L) | 78 | 129 | 37–700 | |
| Absolute Lymphocyte count | 78 | |||
| Day0 | 1.9 | 0.2–4.4 | ||
| Day+15 | 1.03 | 0.3–5.9 | ||
| Day+30 | 1.8 | 0–5.5 | ||
| Albumin g/L | 78 | 35.1 | 25–58 | |
| B2-microglobulin (μmol/L) | 78 | 348 | 70–2900 | |
| 24 Hr Proteinuria (g/d) | 78 | 5.6 | 0–24.9 | |
| ***BMPC (%) | 78 | 8 | 1–30 |
Reece:Otsuka: Honoraria, Research Funding; Janssen: Consultancy, Honoraria, Research Funding; Celgene: Consultancy, Honoraria, Research Funding; Bristol Myers Squibb: Honoraria, Research Funding; Novartis: Honoraria, Research Funding; Millinneum Pharmaceuticals: Research Funding; Merck: Consultancy, Honoraria, Research Funding. Chen:Roche: Honoraria; Johnson & Johnson, Lundbeck, Celgene: Consultancy; Johnson & Johnson, Celgene, GlaxoSmithKline: Research Funding. Tiedemann:Janssen: Honoraria; Celgene: Honoraria. Kukreti:Roche: Honoraria.
Author notes
Asterisk with author names denotes non-ASH members.


This feature is available to Subscribers Only
Sign In or Create an Account Close Modal