Abstract
Abstract 5031
Myeloma inevitably becomes refractory to available agents primarily due to accumulation of genetic and epigenetic mutations in the aberrant clone. The s-phase dependent DNA methylation inhibitor azacitidine may help overcome epigenetically mediated resistance in myeloma cells that cycle despite treatment and reactivate alternate growth control programs in cells with genetic apoptosis resistance. Here we present interim results of an ongoing phase I/II study that explores the safety, antimyeloma activity, and epigenetic effects of continuous azacitidine at predicted non-cytotoxic doses in combination with Rd in RRMM
Entry criteria included measurable disease, relapsed or refractory state, and glomerular filtration rate ≥ 60ml/min (Cockroft-Gault). Lenalidomide 25mg d1–21 every 28d and dexamethasone 40mg weekly were fixed. In phase I (3×3 design) azacitidine was escalated from 30 to 40mg/m2 SC weekly (DL 1 and 2) to 30, 40, and maximally 50mg/m2 SC twice a week (DL 3–5). Dose limiting toxicity (DLT) was assessed during cycle 1 and response every 28 days according to IMWG uniform response criteria (partial response, PR, or better) and adapted EBMT criteria (minor response, MR). Plasma activity of cytidine deaminase (CDA) was measured in dose levels 3–5, using HPLC to quantify deamination of added cytidine (Wilcoxon exact test). Phase II enrolls 10 additional patients at the highest tolerated dose and includes global gene expression and DNA methylation arrays before and after cycle 1 in CD138+, CD34+, and remaining bone marrow cells.
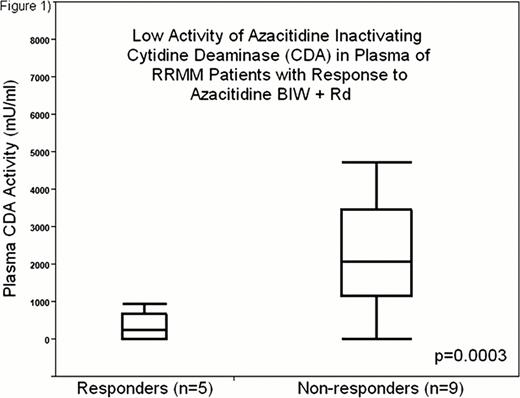
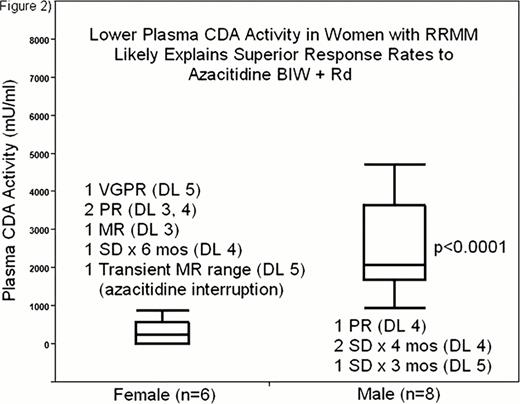
Twenty patients with relapsed (n=1) or refractory (n=19) MM were enrolled after they had received a median of 4 prior regimens (range 1–10). Their disease was refractory to lenalidomide (n=17), bortezomib and/or carfilzomib (n=16), or both lenalidomide and proteasome inhibitors (n=14). 18 patients had received alkylators, 8 high dose chemotherapy, and 6 anthracyclines. One DLT was observed in 1 out of 6 patients treated at DL 4; neutropenic fever without other evidence for infection. No DLT occurred in 4 patients treated so far at DL 5. Grade 3 and 4 toxicities were seen in 11 patients, neutropenia in 9 (lowest ANC 0. 24K/mm3), thrombocytopenia in 4 (lowest 16K/mm3, transfusions used in 1 to avoid treatment delay), neutropenic fever in 2 (1 after cycle 1), and diarrhea, depression, deep venous thrombosis, and a possibly related pleural effusion with hypoxemia in 1 patient each. DL 1 and 2 (n=6) yielded only one MR, but as azacitidine was increased to twice a week (DL 3–5, n=14) the clinical benefit response rate (CBRR, ≥MR) and response rate (RR, ≥PR) increased to 35. 7% and 28. 6%, respectively with 1 very good partial response (VGPR), 3 PR and 1 MR. The patient with MR had secondary plasma cell leukemia (sPCL) after 7 prior regimens and had to be taken off trial due to cytopenias but was continued on reduced azacitidine (20mg/m2), lenalidomide (5–10mg), and prednisone (50mg TIW) reaching PR off trial and achieving a 10 month time to progression (TTP). Of all other responding patients, only the one treated with weekly azacitidine progressed while receiving azacitidine (TTP 3 months), one patient with sPCL progressed during azacitidine interruption for neutropenic fever, another patient was taken off trial due to pleural effusion with hypoxemia, 2 are still on study, currently for 8 and 3 months. The median time to response was 1 cycle (range 1–7). Responders in DL 3–5 had similar treatment histories as the entire cohort, refractory to last Rx: 80%, median previous regimens: 4 (range 1–7), refractory to lenalidomide: 60%, to proteasome inhibitors: 80%, and to both: 60%; but their plasma CDA activity was lower than in non-responders (Figure 1). Women treated at DL 3–5 (n=6) responded better than men (n=8), likely related to lower plasma CDA activity (Figure 2).
Azacitidine was generally well tolerated up to the target maximal dose of 50mg/m2SC twice a week (BIW) in combination with Rd given to 4 out of 6 patients required for establishing the phase 2 dose. Encouraging response rates in refractory MM correlated with low plasma activity of the azacitidine inactivating enzyme CDA found predominantly in women. Results support development of azacitidine in RRMM and argue for testing combination with the competitive inhibitor of CDA, tetrahydrouridine. Until then, dose escalation in non-responding but tolerating patients will be explored.
Reu:Celgene: Research Funding. Off Label Use: Azacitidine in multiple myeloma. Kindwall-Keller:Celgene: Speakers Bureau; TEVA: Speakers Bureau. Duong:Celgene: Research Funding.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal