Abstract
Abstract  547
547
In early stage HL abbreviated chemotherapy (ACT) followed by involved field radiotherapy (IFRT) is the current standard of care but some patients (pts) are probably cured by ACT alone. PET imaging has the potential to identify pts with an excellent prognosis after ACT and so provide the opportunity to avoid radiotherapy and reduce late treatment toxicity in these individuals. We present results of the RAPID trial evaluating PET response directed therapy in pts with previously untreated stages I and II HL, no B symptoms or mediastinal bulk. All pts taking part in RAPID received 3 cycles ABVD followed by a PET scan at one of fifteen quality controlled/assured PET Scan Centres across the UK. Acquired images were transmitted electronically to the Core Lab in London for central review. If the PET scan was reported ‘negative’ (score 1 or 2 on a 5 point scale), pts were randomised between IFRT and no further treatment (NFT). Those with a ‘positive’ PET scan (score 3, 4 or 5) had a 4th cycle ABVD and IFRT. This non-inferiority trial required 400 PET negative pts to be randomised for exclusion of a ≥7% difference in 3-year progression-free survival (PFS) from 95% in the IFRT arm, with 90% power and 5% significance level.
602 pts (321 male, 281 female; median age 34 years) with newly diagnosed, stages IA/IIA HL were registered into the RAPID trial between October 2003 and August 2010. Following 3 cycles ABVD, 571 pts had a PET scan of which 426 (74.6%) were classified as PET negative (score 1, n=301; score 2, n=125). 420 PET negative pts were randomised to receive IFRT (n=209) or NFT (n=211); 6 PET negative pts were not randomised (pt choice, n=3, clinician choice, n=2; error, n=1). 22 of 209 pts randomised to receive IFRT did not receive this treatment because 16 pts (PET score 1, n=14; PET score 2, n=2) declined after they became aware of the randomisation decision, 5 had died and 1 developed pneumocystis jiroveci pneumonia.
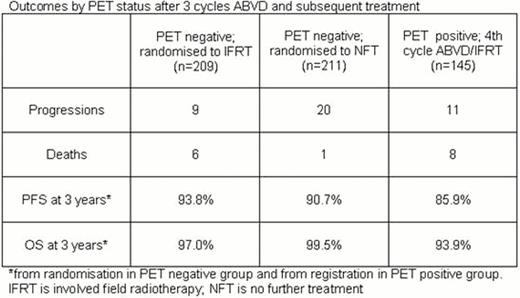
After a median follow-up of 45.7 months from randomisation, 384 of 420 (91.4%) PET negative pts are alive and progression-free, 29 (6.9%) are alive and have progressed and 7 (1.7%) have died giving a combined 3-year PFS of 92.2% and overall survival (OS) of 98.3% in the randomised population. In the IFRT arm of the randomised PET negative population, 194 pts are alive and progression-free, 9 have progressed, and 6 have died (pneumonitis, n=2; HL, n=1; cardiovascular disease, n=1; intracerebal haemorrhage, n=1; angioimmunoblastic T cell lymphoma, n=1). In the NFT arm 190 pts are alive and progression-free, 20 have progressed, and 1 has died (bronchopneumonia, n=1). 3-year PFS is 93.8% IFRT versus 90.7% NFT (risk difference −2.9%, 95% CI −10.7 to 1.4%; the lower limit marginally exceeds the maximum allowable difference of −7%) and 3-year OS is 97.0% IFRT versus 99.5% NFT. For the 145 PET positive pts who received a 4thcycle ABVD and IFRT, 126 are alive and progression-free, 11 progressed, and 8 died to give a 3-year PFS of 85.9% and OS of 93.9% from registration. These results are summarized in the Table.
These results show that in early stage HL, pts with a negative PET after 3 cycles of ABVD have an excellent prognosis without any further treatment. The 3-year PFS is slightly higher in the PET negative pts receiving IFRT (93.8% vs 90.7%), where a non-inferiority margin of 7% was deemed acceptable. We conclude that in stages IA/IIA HL, RT is unnecessary in the 75% of pts who become PET negative after 3 cycles ABVD. Such a response adapted approach based on centrally reviewed PET imaging reduces treatment time and costs, improves tolerability and most importantly removes the burden of early and late toxicity of radiotherapy from the PET negative population.
No relevant conflicts of interest to declare.
Author notes
Asterisk with author names denotes non-ASH members.

This icon denotes a clinically relevant abstract

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal