Abstract
Abstract 645
Acquired aplastic anemia (AA) is a non-clonal hematopoietic disorder caused by the immune system attack on hematopoietic stem cells (HSCs) and has been thought to be rarely associated with intrinsic HSC defects. However, clonal hematopoiesis by HSCs with genetic alterations may not be uncommon in AA since clonal granulocyte populations showing the paroxysmal nocturnal hemoglobinuria (PNH) phenotype or chromosome 6p uniparental disomy (6pUPD) are detected in approximately 50% and 13% of AA patients, respectively (Katagiri T, et al, Blood, 2011). Abnormal HSCs other than those with the PIG-A mutation or 6pUPD may also contribute to hematopoiesis in AA patients due to immune escape or preferential activation of HSCs with genetic alterations in response to immune pressure in the bone marrow (BM).
To identify novel genetic abnormalities in patients with AA, we carried out whole exome sequencing of peripheral blood leukocytes from 96 AA patients using a next generation sequencer. Among 40 abnormalities found in more than one patient, we selected abnormalities in genes potentially related to HSC regulation and examined the relevance of the genetic alterations to BM failure.
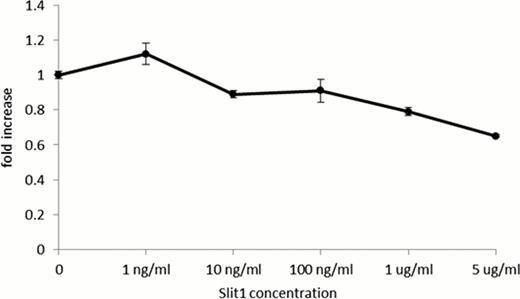
Missense mutations of SLIT1, a family of secreted glycoproteins that bind members of the Robo receptor family and regulate axon guidance, were detected in 15 of 96 patients and confirmed by Sanger sequencing in 3 patients who had been in remission for 1–3 years after immunosuppressive therapy at the time of blood sampling. Since previous studies showed that the Slit-Robo pathway negatively regulates proliferation of murine HSCs as well as a human cancer cell line in an autocrine manner (Prasad A et al., J Biol Chem; 283: 26624. 2008) and the break point of chromosome 10 in a patient with immune-mediated AA possessing t(1:10) was located within the SLIT1 gene, we studied SLIT1 and Robo receptor expression in BM mononuclear cells (BMMCs) obtained from healthy volunteers and patients with AA, and in myeloid leukemia cell lines. Although SLIT1 mRNA was scarce in un-stimulated BMMCs from healthy volunteers, SLIT1 gene expression increased 2.5–3.5 fold when incubated in conditioned medium from PHA or LPS stimulated peripheral blood mononuclear cells. The steady state expression level of SLIT1 by BMMCs from AA patients was 6 fold higher than from healthy individuals. SLIT1 mRNA was also detected in several myeloid leukemia cell lines including K562 and MV4-11, and its expression increased 2 fold by culturing the cells for 48 hours with the human stromal cell line HS-5. Examination of Robo receptor expression using antibodies specific to Robo1-4 showed that BM CD34+ cells from healthy individuals and K562 cells expressed Robo1. Recombinant Slit1 inhibited proliferation of K562 in a dose-dependent manner at concentrations of 1 and 5 mg/ml (Figure 1).
The presence of clonal leukocytes with SLIT1 mutations in patients with immune-mediated AA suggests that HSCs with these mutations have survival or proliferation advantages over HSCs without SLIT1 mutations. Slit1 secreted from HSCs or hematopoietic progenitor cells in response to inflammatory cytokines may inhibit their own activation by binding to Robo1, but mutant Slit1 may fail to inhibit activation, leading to preferential commitment of the mutant HSCs to hematopoiesis. The Slit-Robo pathway may therefore serve as a novel therapeutic target for immune-mediated BM failure.
No relevant conflicts of interest to declare.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal