Abstract
Abstract 848
DNA methylation is an epigenetic modification in vertebrate genomes critical for regulation of gene expression. DNA methylation is catalyzed by a family of DNA methyltransferase enzymes, Dnmt1, Dnmt3a, and Dnmt3b. Dnmt1 is primarily a maintenance methyltransferase, targeting hemimethylated DNA to reestablish methylation marks after DNA replication. Dnmt3a and Dnmt3b are de novo methyltransferases that are essential for normal embryonic development. In humans, somatic mutations in DNTM3A have been identified in ∼20% of human acute myeloid leukemia (AML) and ∼10% of myelodysplastic syndrome (MDS) patients, but the mechanisms through which these mutations contribute to pathogenesis is not well understood. Congenital mutations in DNMT3B can cause ICF (immunodeficiency, centromeric instability, and facial anomalies) syndrome. These patients exhibit chromosomal instability due to heterochromatin decondensation and demethylation of satellite DNA.
Our group has recently reported that Dnmt3a is essential for HSC differentiation (Challen Nature Genetics, 2011). Conditional knockout of Dnmt3a (Dnmt3a-KO) resulted in HSCs that could not sustain peripheral blood generation after serial transplantation, but phenotypically defined HSCs accumulated in the bone marrow.
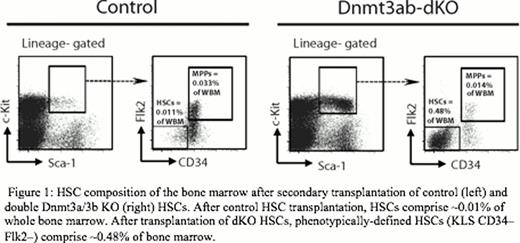
Dnmt3b is also highly expressed in HSCs, but its contribution to gene regulation in hematopoiesis is unclear. Here, we examine the role of Dnmt3b, alone and in combination with Dnmt3a KO, in the regulation of hematopoiesis. We performed conditional ablation of Dnmt3b, as well as Dnmt3a and Dnmt3b simultaneously using the Mx1-cre system. Unlike the Dnmt3a-KO HSCs, loss of Dnmt3b had a minimal impact on blood production. Even after several rounds of transplantation, 3b-KO HSCs performed similarly to WT controls. However, the Dnmt3ab-dKO (double knock-out) peripheral blood contribution was quickly and severely diminished, accompanied by a dramatic accumulation of Dnmt3ab-dKO HSCs in the bone marrow (Figure 1). The dKO phenotype paralleled that of the 3a-KO HSC, but was more extreme.
To examine the impact of loss of Dnmt3a and -3b on DNA methylation in HSCs, we performed Whole Genome Bisulfite Sequencing (WGBS) on Dnmt3a-KO, Dnmt3ab- dKO and control HSCs. As we previously found with more limited DNA methylation analysis, loss of Dnmt3a led to both increases and decreases of DNA methylation at distinct genomic regions (Challen, Nature Genetics, 2011). However, loss of both Dnmt3a and -3b primarily resulted in loss of DNA methylation that was much more extensive than that seen in the 3a-KO. In addition, RNAseq of the mutant HSCs revealed increased expression of repetitive elements, inappropriate splicing, and truncation of 3ÕUTRs.
To gain insight into the accumulation of Dnmt3ab-dKO HSCs in the bone marrow, we performed a time course analysis of the proliferation and apoptosis status of the HSCs. Every four weeks after transplantation of HSCs, we sacrificed a cohort of 3 control and 3 dKO mice, counted donor derived HSCs in the bone marrow, and analyzed their Ki67 and Annexin V expression. Up to 12 weeks post-transplant, no significant differences are seen in the expression of Ki67 or Annexin V.
These data show that while Dnmt3b alone has minimal impact on DNA methylation in HSCs, Dnmt3a and -3b act synergistically to effect gene expression changes that permit HSC differentiation. In the absence of both of these de novo DNA methyltransferases, there is an immediate and extreme shift toward self-renewal of dKO HSCs. The Ki67 and Annexin V expression patterns suggest that a lack of de novo DNA methylation does not affect the proliferation or apoptosis of HSCs, but instead that the accumulation of HSCs and lack of peripheral blood contribution is primarily due to an imbalance between self-renewal and differentiation. By understanding the mechanisms through which Dnmt3a and -3b exert these effects, we should identify genes that are critical for normal hematopoietic differentiation. These genes may serve as targets for therapeutic intervention in malignancies caused by defective DNA methyltransferases.
HSC composition of the bone marrow after secondary transplantation of control (left) and double Dnmt3a/3b KO (right) HSCs. After control HSC transplantation, HSCs comprise ∼0.01% of whole bone marrow. After transplantation of dKO HSCs, phenotypically-defined HSCs (KLS CD34–Flk2–) comprise ∼0.48% of bone marrow.
HSC composition of the bone marrow after secondary transplantation of control (left) and double Dnmt3a/3b KO (right) HSCs. After control HSC transplantation, HSCs comprise ∼0.01% of whole bone marrow. After transplantation of dKO HSCs, phenotypically-defined HSCs (KLS CD34–Flk2–) comprise ∼0.48% of bone marrow.
No relevant conflicts of interest to declare.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal