Key Points
Dengue infection triggers functional inflammasome assembly in platelets.
Platelets may contribute to increased vascular permeability in dengue virus infection by synthesis and release of IL-1β.
Abstract
Dengue is the most frequent hemorrhagic viral disease and re-emergent infection in the world. Although thrombocytopenia is characteristically observed in mild and severe forms of dengue, the role of platelet activation in dengue pathogenesis has not been fully elucidated. We hypothesize that platelets have major roles in inflammatory amplification and increased vascular permeability during severe forms of dengue. Here we investigate interleukin (IL)-1β synthesis, processing, and secretion in platelets during dengue virus (DV) infection and potential contribution of these events to endothelial permeability during infection. We observed increased expression of IL-1β in platelets and platelet-derived microparticles from patients with dengue or after platelet exposure to DV in vitro. We demonstrated that DV infection leads to assembly of nucleotide-binding domain leucine rich repeat containing protein (NLRP3) inflammasomes, activation of caspase-1, and caspase-1–dependent IL-1β secretion. Our findings also indicate that platelet-derived IL-1β is chiefly released in microparticles through mechanisms dependent on mitochondrial reactive oxygen species–triggered NLRP3 inflammasomes. Inflammasome activation and platelet shedding of IL-1β–rich microparticles correlated with signs of increased vascular permeability. Moreover, microparticles from DV-stimulated platelets induced enhanced permeability in vitro in an IL-1–dependent manner. Our findings provide new evidence that platelets contribute to increased vascular permeability in DV infection by inflammasome-dependent release of IL-1β.
Introduction
Dengue is a tropical infectious disease caused by 1 of 4 dengue virus serotypes (DV-1 to DV-4). According to the World Health Organization (WHO), >2.5 billion people are at risk for contracting dengue, and >50 million individuals are annually infected.1,2 A progressive global expansion of dengue, including a higher frequency of severe dengue, has been recently reported, with an incidence >3 times the previously estimated by WHO.3 Dengue induces a spectrum of clinical manifestations that range from a self-limited fever to severe dengue, which is associated with life-threatening increases in vascular permeability, hypovolemia, hypotension, and shock.2,4 Thrombocytopenia is also commonly observed in mild and severe dengue syndromes and correlates with clinical outcome.2,5,6 Although thrombocytopenia is a hallmark of dengue, the role of platelet activation in the pathogenesis of dengue has not been elucidated.
Platelets are highly specialized and essential effector cells in hemostasis. Besides the hemostatic activities of platelets, it is increasingly recognized that platelets are also major inflammatory cells with key roles in innate and adaptive immune responses.7,8 Platelets express, store, and/or synthesize and rapidly release a variety of factors, including cytokines that regulate interactions with endothelium and may alter endothelial functions in pathological conditions. Nevertheless the roles of platelets in inflammatory amplification and altered vascular barrier integrity in dengue have not been addressed.
Interleukin-1β (IL-1β) is an important proinflammatory cytokine increased during DV infection.5,9-11 This cytokine has been linked to increased endothelial permeability,5,11 thrombosis, and dysregulated hemostasis in dengue.9 IL-1β is synthesized as a larger precursor protein that is cleaved into the active cytokine by inflammasomes, intracellular multimolecular complexes that control caspase-1 activity in the innate immune system. The best-characterized inflammasome contains the nucleotide-binding domain leucine rich repeat containing protein (NLRP3), an adaptor molecule known as apoptosis-associated speck-like protein containing a caspase recruitment domain (ASC), and activated caspase-1.12 The NALP3–caspase-1 inflammasome responds to activation by bacterial and viral components,13 ATP, asbestos, monosodium urate crystals,14 and production of reactive oxygen species (ROS) by mitochondria.15 Increased IL-1β has been shown in patients with dengue,5 and in vitro DV infection of macrophages causes inflammasome activation.16
The ability of activated platelets to splice constitutive intron-containing IL-1β pre-RNA to produce mature IL-1β mRNA and synthesize IL-1β has been previously demonstrated.17-19 Nevertheless, the mechanisms involved in IL-1β processing and secretion by platelets remains elusive. It has been previously shown that lipopolysaccharide (LPS)-stimulated platelets release IL-1β packaged into microparticles (MPs).20 MPs are small (0.1-1.0 µm) membrane-bound bodies that are released from cells during activation and cell death.21 We have previously shown that platelets from dengue patients have characteristics indicating increased activation and apoptosis,22 but whether these activated platelets synthesize IL-1β and shed IL-1β–containing MPs is unknown. Here we demonstrate increased expression of IL-1β in platelets and platelet-derived MPs from patients with dengue or after platelet exposure to DV in vitro. In exploring the mechanisms involved in IL-1β secretion from platelets, we describe for the first time the presence of inflammasome components in platelets and inflammasome activity during DV infection. Specifically, our data strongly suggest that assembly of the NRLP3–caspase-1 inflammasomes mediates IL-1β secretion in MPs during DV infection. In addition, receptor-interacting protein (RIP) kinases and generation of ROS by mitochondria are required for caspase-1 inflammasome activation. Our results also implicate platelet synthesis of IL-1β, caspase-1 activation, and shedding of IL-1β–rich MPs as key effectors in vascular permeability during dengue illness.
Patients and methods
Human subjects
Peripheral vein blood samples were obtained from 36 serologically/molecularly confirmed DV-infected patients from the Instituto de Pesquisas Clínicas Evandro Chagas–Fundação Oswaldo Cruz, Rio de Janeiro, Brazil, whose characteristics are presented in Table 1. The average day of sample collection after onset of illness was 4.5 ± 1.7. Peripheral vein blood was also collected from 22 aged-matched healthy subjects. The study protocol was approved by the Institutional Review Board (Instituto de Pesquisas Clínicas Evandro Chagas #016/2010), and the experiments were performed in compliance with this protocol. Written informed consent was obtained from all volunteers prior to any study-related procedure in accordance with the Declaration of Helsinki.
Characteristics of DV-infected patients
| Characteristic . | Control (n = 22) . | Dengue (n = 36) . |
|---|---|---|
| Age, years | 30 (27-34) | 38 (27-45) |
| Gender, male | 9 (41%) | 20 (55.55%) |
| Platelet count, ×1000/mm3 | — | 113.5 (81.2-162.5) |
| Hematocrit, % | — | 43.5 (40.7-45.3) |
| Albumin, g/dL | — | 3.7 (3.5-4.0) |
| TGO/AST, IU/L | — | 68.5 (34.7-114.3) |
| TGP/ALT, IU/L | — | 72 (49-105.5) |
| Hemorrhagic manifestations* | — | 14 (38.88%) |
| Venous hydration | — | 9 (25%) |
| Secondary Infection | — | 23 (64%) |
| Mild dengue | — | 22 (61.11%) |
| Mild dengue with warning signs† | — | 12 (33.33%) |
| Severe dengue‡ | — | 2 (5.55%) |
| PCR positive | 21 | |
| DV-1 | 12 (57%) | |
| DV-2 | 1 (5%) | |
| DV-4 | 8 (38%) |
| Characteristic . | Control (n = 22) . | Dengue (n = 36) . |
|---|---|---|
| Age, years | 30 (27-34) | 38 (27-45) |
| Gender, male | 9 (41%) | 20 (55.55%) |
| Platelet count, ×1000/mm3 | — | 113.5 (81.2-162.5) |
| Hematocrit, % | — | 43.5 (40.7-45.3) |
| Albumin, g/dL | — | 3.7 (3.5-4.0) |
| TGO/AST, IU/L | — | 68.5 (34.7-114.3) |
| TGP/ALT, IU/L | — | 72 (49-105.5) |
| Hemorrhagic manifestations* | — | 14 (38.88%) |
| Venous hydration | — | 9 (25%) |
| Secondary Infection | — | 23 (64%) |
| Mild dengue | — | 22 (61.11%) |
| Mild dengue with warning signs† | — | 12 (33.33%) |
| Severe dengue‡ | — | 2 (5.55%) |
| PCR positive | 21 | |
| DV-1 | 12 (57%) | |
| DV-2 | 1 (5%) | |
| DV-4 | 8 (38%) |
Data are expressed as median (interquartile range) or number (%).
ALT, alanine aminotransferase; AST, aspartate aminotransferase; TGO, glutamic-oxalacetic transaminase; TGP, glutamic-pyruvic transaminase.
Gingival, vaginal, and/or gastrointestinal bleeding, petechiae, and purpura.
Abdominal pain or tenderness, persistent vomiting, clinical fluid accumulation, mucosal bleed, and/or increased hematocrit concurrent with rapid decrease in platelet count; according to WHO criteria.2
Severe plasma leakage, fluid accumulation with ascites (evidenced by ultrasonography), and/or severe bleeding (vaginal bleed and/or gastrointestinal bleed); according to WHO criteria.2
The cohort was comprised of patients with mild to severe dengue. Specifically, 33.3% of the patients presented warning signs and 5.5% presented with severe dengue according to WHO guidelines2 (Table 1). Levels of immunoglobulin M (IgM) and IgG specific to DV E protein were measured using a standard capture enzyme-linked immunosorbent assay kit according to the manufacturer’s instructions (E-Den01M and E-Den01G; PanBio). Primary and secondary infections were distinguished using the IgM/IgG antibody ratio as previously described,23-25 Sixty-four percent of patients were found to have secondary DV infection.
Platelet isolation
Platelets were isolated using the methods of Hamburger and McEver.26 Briefly, the peripheral blood samples were drawn into acid-citrate-dextrose and centrifuged at 200g for 20 minutes to obtain platelet-rich plasma. Platelet-rich plasma was recentrifuged at 500g for 20 min in the presence of 100 nM Prostaglandin E1 (PGE1) (Cayman Chemicals). The supernatant was discarded, and the platelet pellet was resuspended in 2.5 mL of phosphate-buffered saline containing 2 mM EDTA, 0.5% human albumin serum, and 100 nM PGE1 and incubated with 10 µL of anti-CD45 tetrameric antibody complexes for 10 minutes and with 20 µL of dextran-coated magnetic beads for an additional 15 minutes before purification in a magnet (Human CD45 depletion kit; StemCell, Easy Sep Technology). Recovered platelets were resuspended in 25 mL of piperazine-N,N′-bis(2-ethanesulfonic acid)/saline/glucose (5 mM piperazine-N,N′-bis[2-ethanesulfonic acid], 145 mM NaCl, 4 mM KCl, 50 mM Na2HPO4, 1 mM MgCl2-6 H2O, and 5.5 mM glucose) containing 100 nM of PGE1. The platelet suspension was recentrifuged at 500g for 20 minutes, the supernatant was discarded, and the pellet was resuspended in medium 199 (M199; Lonza Biologics). The purity of the platelet preparations (>99% CD41+) was confirmed by flow cytometry.
Flow cytometric analyses
Freshly isolated platelets (106-107) were resuspended in 500 µL of modified Tyrode’s (137 mM NaCl, 2.68 mM KCI, 5 mM N-2-hydroxyethylpiperazine-N′-2-ethanesulfonic acid, 1 mM MgCl2, 11.9 mM NaHCO3, 0.42 mM NaH2PO4, 4.7 mM glucose; pH 7.4). A minimum of 10 000 events per gate was acquired using a FACScalibur flow cytometer (BD Bioscience). The following indices were assessed: CD41 surface expression was determined by incubating (37°C for 30 minutes) platelets with a phycoerythrin- or fluorescein isothiocyanate–conjugated antibodies that target CD41 (eBioscience) (0.5 µg/mL); mitochondrial-derived ROS (ROSm) was detected using the cationic probe MitoSOX Red (Molecular Probes) (2.5 µM at 37°C for 10 minutes); and activation of caspase-1 was assessed by incubating platelets with the fluorescent probe green Fluorescent Labeled Inhibitor of Caspase-1 (FLICA), which irreversibly binds to activated caspase-1, according to the manufacturer’s instructions (Immunochemistry Technologies). For intracellular IL-1β determination, CD41-stained platelets were fixed with paraformaldehyde 4% for 20 minutes, washed once, and permeabilized with Triton 0.1% for 10 minutes. Platelets were then incubated for 30 minutes with anti–IL-1β antibody (5 µg/mL; Santa Cruz Biotechnology), followed by an additional 30-minute incubation with secondary Alexa Fluor 546–conjugated anti-rabbit IgG. Isotype-matched antibodies were used to control nonspecific binding of antibodies. Platelets were distinguished by specific binding of anti-CD41 and characteristic forward and side scattering. MPs size was determined using 0.989-µm latex beads. Additional details regarding MPs detection and isolation are presented in supplemental Materials on the Blood Web site.
Western blotting
Western blotting was performed as described in supplemental Methods. The primary antibodies used for this work were rabbit anti-human IL-1β (Santa Cruz Biotechnology) and mouse anti-human β-actin (Sigma-Aldrich).
Confocal fluorescence microscopy
Immunofluorescence for NLRP3 and ASC was performed as described in supplemental Methods. Preparations were analyzed on a FV10i-O (Olympus) confocal scanning microscope. Olympus Fluoview-FV1000 5.0 image acquisition software was used for recording.
In vitro platelet stimulation
DV serotype 2 strain 16881 was propagated in C6/36 Aedes albopictus mosquito cells as indicated in supplemental Methods. Platelets from healthy, uninfected donors were incubated (6 hours at 37°C in a 5% CO2 atmosphere) with DV-2 at a multiplicity of infection of 1 plaque forming unit per platelet. To characterize the pathways involved in platelet inflammasome activation, we preincubated platelets with the caspase-1 inhibitor YVAD-fmk (MBL International) (10 µM), or the RIP-1 inhibitor necrostatin-1 (Nec-1; Calbiochem) (50 µg/mL) for 30 minutes or with the mitochondrial targeted antioxidant mitoTEMPO (Enzo Life Sciences) (500 µM) for 1 hour before DV exposure.
Cytokine measurement
The content of IL-1β and regulated on activation normal T expressed and secreted (RANTES) in supernatants from DV-exposed platelets was measured by Luminex using a Multiplex cytokine immunoassay according to the manufacturer's instructions (Bio-Plex Human Cytokine Assay; Bio-Rad).
Transwell assay of endothelial permeability
Permeability changes in endothelial cells were determined by measuring the passage of fluorescein isothiocyanate–conjugated albumin across human dermal microvascular endothelial cell (HMEC-1) monolayers after incubation with MPs recovered from DV or mock exposed platelets. To characterize the role played by IL-1β, we incubated HMEC-1 monolayers with the IL-1 receptor antagonist (IL-1Ra) (10 µg/mL). Details are given in supplemental Materials.
Statistical analysis
All statistical analyses were performed using GraphPad Prism 5.0 software. The Kolmogorov-Smirnov test was used to determine whether samples followed a normal distribution. A nonparametric Mann-Whitney test was used to determine whether differences were present between 2 experimental groups. A paired 2-tailed t test was used to compare stimulated and nonstimulated platelets from the same healthy donor. Correlations were assessed using the Pearson test.
Results
Platelets from dengue-infected patients express IL-1β
Activated platelets can mediate inflammatory and immune responses using a variety of mechanisms, including release of stored cytokines and newly synthesis of others.8,27 The ability of platelets to splice pre-mRNA and synthesize IL-1β by translating the mature mRNA transcript has been well demonstrated in vitro.17,18 Most recently, synthesis of IL-1β by platelets in vivo has been reported in mice with experimental cerebral malaria.19 To assess whether platelets synthesize IL-1β during dengue illness, platelets from healthy volunteers or DV-infected patients were labeled with an antibody that recognizes both full-length (pro) and cleaved (active) IL-1β. The percentage of IL-1β–expressing platelets was significantly higher (P < .01) in samples from patients with dengue (53.1 ± 20.4%) compared with samples from healthy volunteers (7.5 ± 5.7%) (Figure 1A).
IL-1β synthesis in DV-infected platelets and platelets from healthy volunteers. (A) The percentage of IL-1β–expressing platelets was assessed in platelets that were isolated from healthy subjects (control) or patients with dengue (dengue). The boxes indicate the median and interquartile ranges and the whiskers indicate 5 to 95 percentile. (B-C) Washed platelets from healthy volunteers were exposed to mock, infective DV-2 (DV) or heat-inactivated DV-2 (heat DV) as described in Patients and methods. Panel B shows the percentage of IL-1β–expressing platelets, and panel C shows the concentration of IL-1β in supernatants of platelets incubated in each condition. The bars represent mean ± standard error of the mean (SEM) of 8 experiments performed using samples from independent healthy donors. *P < .05 and **P < .01 compared with healthy volunteers or mock exposed platelets. (Insets) Representative density plots of (A) IL-1β–expressing platelets from 1 healthy volunteer and 1 dengue patient or (B) platelets from 1 healthy volunteer that were exposed to mock, DV, or heat DV. The values in each region of the quadrants indicate the cell frequencies in the respective quadrant.
IL-1β synthesis in DV-infected platelets and platelets from healthy volunteers. (A) The percentage of IL-1β–expressing platelets was assessed in platelets that were isolated from healthy subjects (control) or patients with dengue (dengue). The boxes indicate the median and interquartile ranges and the whiskers indicate 5 to 95 percentile. (B-C) Washed platelets from healthy volunteers were exposed to mock, infective DV-2 (DV) or heat-inactivated DV-2 (heat DV) as described in Patients and methods. Panel B shows the percentage of IL-1β–expressing platelets, and panel C shows the concentration of IL-1β in supernatants of platelets incubated in each condition. The bars represent mean ± standard error of the mean (SEM) of 8 experiments performed using samples from independent healthy donors. *P < .05 and **P < .01 compared with healthy volunteers or mock exposed platelets. (Insets) Representative density plots of (A) IL-1β–expressing platelets from 1 healthy volunteer and 1 dengue patient or (B) platelets from 1 healthy volunteer that were exposed to mock, DV, or heat DV. The values in each region of the quadrants indicate the cell frequencies in the respective quadrant.
DV induces synthesis and secretion of IL-1β in platelets
Next we examined whether DV-2 directly induces IL-1β synthesis in platelets from healthy volunteers yielding an expression pattern similar to that of platelets from dengue patients. Platelets from healthy donors were incubated for 6 hours with infectious DV-2, heat-inactivated DV-2, or mock. Exposure of platelets to DV significantly increased (P < .05) the percent of IL-1β–positive platelets (infectious DV-2, 26.0 ± 8.6% vs 7.4 ± 3.2% or 4.2 ± 2.8% for heat-inactivated DV-2 and mock, respectively) (Figure 1B). The amounts of secreted IL-1β were also increased in the supernatants of DV-exposed platelets compared with those exposed to mock or to heat-inactivated DV (Figure 1C). Platelet exposure to purified DV similarly increased IL-1β synthesis. Virus purification was obtained using a Centricon YM-100 Centrifugal Filter (Merck Millipore), and platelets were exposed separately to the filtrate or to the retentate (purified virus). Exposure to purified DV-2 significantly increased (P < .05) the percentage of IL-1β–positive platelets compared with the virus filtrate or to mock sample processed in parallel (purified DV-2, 31.2 ± 12.5% vs 5.9 ± 3.2 or 2.9 ± 1.5% for virus filtrate and mock retentate, respectively; data not shown). These results indicate that human platelets synthesize, accumulate, and secrete IL-1β on DV infection.
Dengue induces shedding of IL-1β–rich MPs in platelets
We observed higher quantities of platelet-derived MPs in samples from dengue patients compared with control participants (Figure 2A). As it was previously described that IL-1β can be released in MPs,20,28 we examined platelet-derived MPs for IL-1β. IL-1β was detected in an average 25.3 ± 11.2% of MPs in samples from dengue patients and 3.5 ± 1.9% in controls (Figure 2B). In addition, exposure of platelets from healthy volunteers to DV-2, but not to heat-inactivated DV-2 or mock, significantly (P < .01) increased the shedding of MPs (Figure 2C). The amounts of IL-1β–containing MPs were also increased when platelets were exposed to DV-2 compared with heat-inactivated DV-2 or mock (16.4 ± 5.0 vs 3.5 ± 1.4 or 3.2 ± 2.3, respectively, P < .01) (Figure 2D). Similarly, platelet exposure to purified DV increased the number of IL-1β–rich MPs (purified DV-2, 21.9 ± 6.9 vs 3.9 ± 2.4 or 1.4 ± 0.6 for virus filtrate and mock retentate, respectively, P < .05; data not shown).
Dengue induces shedding of IL-1β–rich microparticles in platelets. (A) The proportion of MPs per 100 platelets was assessed in samples from healthy subjects (control) or dengue-infected patients (dengue). (B) The percentage of IL-1β–positive MPs in samples obtained from control participants or dengue patients. The boxes indicate the median and interquartile ranges and the whiskers indicate 5 to 95 percentile. (C-D) Washed platelets from healthy volunteers were exposed to mock, infective DV-2 (DV), or heat-inactivated DV-2 (heat DV) as described in Patients and methods. (C) The levels of platelet-derived MPs and (D) the percentage of IL-1β–containing MPs for each condition are shown. The bars represent mean ± SEM of 8 experiments performed using samples from independent healthy donors. *P < .05 and **P < .01 compared with healthy volunteers or mock exposed platelets. (Insets) Representative density plots showing (B) IL-1β–expressing MPs from 1 healthy volunteer and 1 dengue patient or (D) platelets from 1 healthy volunteer that were exposed to mock, DV, or heat DV. The values in each region of the quadrants indicate the cell frequencies in the respective quadrant.
Dengue induces shedding of IL-1β–rich microparticles in platelets. (A) The proportion of MPs per 100 platelets was assessed in samples from healthy subjects (control) or dengue-infected patients (dengue). (B) The percentage of IL-1β–positive MPs in samples obtained from control participants or dengue patients. The boxes indicate the median and interquartile ranges and the whiskers indicate 5 to 95 percentile. (C-D) Washed platelets from healthy volunteers were exposed to mock, infective DV-2 (DV), or heat-inactivated DV-2 (heat DV) as described in Patients and methods. (C) The levels of platelet-derived MPs and (D) the percentage of IL-1β–containing MPs for each condition are shown. The bars represent mean ± SEM of 8 experiments performed using samples from independent healthy donors. *P < .05 and **P < .01 compared with healthy volunteers or mock exposed platelets. (Insets) Representative density plots showing (B) IL-1β–expressing MPs from 1 healthy volunteer and 1 dengue patient or (D) platelets from 1 healthy volunteer that were exposed to mock, DV, or heat DV. The values in each region of the quadrants indicate the cell frequencies in the respective quadrant.
Activation of NLRP3 inflammasome in platelets from patients with dengue
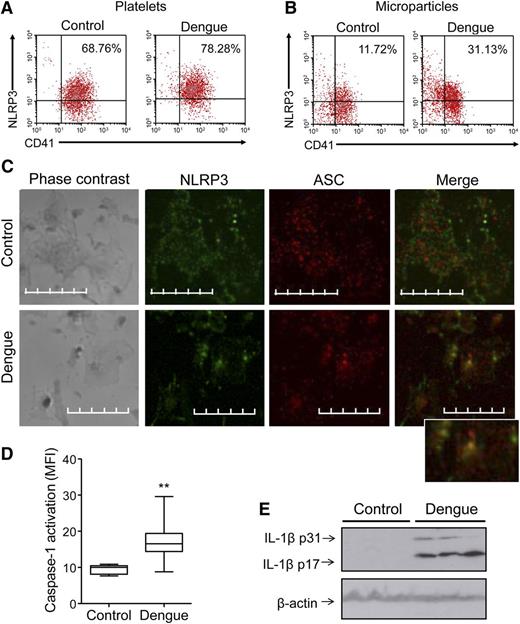
Inflammasomes are intracellular multimolecular complexes that control caspase-1 activity in the innate immune system and are critical for IL-1β processing and secretion. NLRP3, along with the adaptor protein ASC, mediates caspase-1 activation via assembly of the inflammasome in response to various infectious and tissue damage stimuli.12 Inflammasome components and assembly have not been previously shown in platelets. Our preliminary RNA sequencing analyses indicate that megakaryocytes and platelets have components of the inflammasome (data not shown). Flow cytometric analysis of intracellular NLRP3 demonstrated that this key component protein is present in platelets from both patients and controls (Figure 3A). Furthermore, levels of NLRP3 were enriched in MPs from DV-infected patients compared with those from control participants (Figure 3B). To assess activation of the NLRP3 inflammasome in more depth, we examined the assembly of the NLRP3-inflammasome complex using confocal microscopy. Platelets isolated from dengue-infected patients or healthy volunteers were stained with antibodies against NLRP3 and ASC. We observed cytoplasmic colocalization of NLRP3 and ASC in platelets from dengue patients but not in platelets from healthy volunteers, in which both proteins were present but did not colocalize (Figure 3C). Consistent with these data, the intensity of caspase-1 activation was increased (P < .01) in platelets from dengue patients (17.11 ± 4.74 mean fluorescence intensity [MFI]) compared with controls (9.53 ± 1.28 MFI) (Figure 3D). Moreover, higher amounts of cleaved IL-1β were observed in platelets from DV-infected patients compared with platelets from healthy volunteers by western blot (Figure 3E). Together, these results indicate the presence of the NLRP3 inflammasome components in platelets and assembly of the NLPR3 inflammasome with caspase-1 activation on DV infection.
Activation of the NLRP3 inflammasome in platelets from DV-infected patients. (A-B) Representative density plots showing the expression of NLRP3 in (A) platelets and (B) MPs from healthy subjects (control) or patients with dengue (dengue). The values in each region of the quadrants indicate the cell frequencies in the respective quadrant. (C) Platelets obtained from healthy subjects and patients with dengue were stained with anti-NLRP3 (green) and anti-ASC (red) and examined by confocal microscopy using an Olympus FV10i-O with a 240× lens. The images were processed using FLUOVIEW FV1000 MPE software version 5.0 and further analyzed using Adobe Photoshop CS Version 8.0. Bars represent 10 µm. (D) Caspase-1 activation in platelets isolated from control participants and dengue patients was assessed by the MFI of the fluorescent probe FLICA. The boxes indicate the median and interquartile ranges and the whiskers indicate 5 to 95 percentile. **P < .01 compared with healthy volunteers. (E) Western blot analysis of pro (p31) and cleaved (p17) IL-1β and of β-actin in platelets that were isolated from 3 healthy volunteers and 3 dengue patients.
Activation of the NLRP3 inflammasome in platelets from DV-infected patients. (A-B) Representative density plots showing the expression of NLRP3 in (A) platelets and (B) MPs from healthy subjects (control) or patients with dengue (dengue). The values in each region of the quadrants indicate the cell frequencies in the respective quadrant. (C) Platelets obtained from healthy subjects and patients with dengue were stained with anti-NLRP3 (green) and anti-ASC (red) and examined by confocal microscopy using an Olympus FV10i-O with a 240× lens. The images were processed using FLUOVIEW FV1000 MPE software version 5.0 and further analyzed using Adobe Photoshop CS Version 8.0. Bars represent 10 µm. (D) Caspase-1 activation in platelets isolated from control participants and dengue patients was assessed by the MFI of the fluorescent probe FLICA. The boxes indicate the median and interquartile ranges and the whiskers indicate 5 to 95 percentile. **P < .01 compared with healthy volunteers. (E) Western blot analysis of pro (p31) and cleaved (p17) IL-1β and of β-actin in platelets that were isolated from 3 healthy volunteers and 3 dengue patients.
Platelet shedding of IL-1β–containing MPs depends on caspase-1 activation
Exposure of platelets from healthy volunteers to DV-2 significantly increased (P < .05) the intensity of caspase-1 activation (infectious DV-2, 47.4 ± 10.6 vs 26.1 ± 9.8 or 25.8 ± 9.7 MFI for heat-inactivated DV-2 and mock, respectively) (Figure 4A), consistent with the pattern of increased activity in platelets from patients with dengue (Figure 3D). To clarify the role played by caspase-1 in platelet shedding of IL-1β–rich MPs, we treated platelets with the caspase-1 inhibitor YVAD prior to DV exposure. As expected, pretreatment of platelets with YVAD did not affect platelet synthesis of IL-1β (Figure 4B) but significantly reduced (P < .05) IL-1β secretion by DV-exposed platelets (Figure 4C). Interestingly, platelet exposure to DV-2 in the presence of YVAD partially impaired the shedding of MPs (data not shown) and completely impaired (P < .05) DV-induced shedding of IL-1β–containing MPs (Figure 4D), indicating that DV-triggered IL-1β packaging and release into MPs depend on caspase-1 inflammasome activation.
Platelet shedding of IL-1β–rich MPs depends on caspase-1 activation. (A) Platelets from healthy subjects were exposed to mock, infective DV-2 (DV), or heat-inactivated DV-2 (heat DV), and caspase-1 activation was evaluated through the MFI of the fluorescent probe FLICA. (B-D) Platelets were exposed to mock, DV, or heat DV in the presence or absence of the caspase-1 inhibitor YVAD. B depicts the percentage of IL-1β–expressing platelets, and panel C shows the concentration of IL-1β in the supernatant of platelets incubated in each condition. Panel D shows the percentage of IL-1β–containing MPs for each condition. The bars represent the mean ± SEM of 6 to 8 independent experiments using samples from different healthy donors. *P < .05 and **P < .01 compared with mock exposed platelets that received the same treatment (dimethylsulfoxide [DMSO] or YVAD). #P < .05 between DV-2–treated platelets that were incubated with YVAD or DMSO.
Platelet shedding of IL-1β–rich MPs depends on caspase-1 activation. (A) Platelets from healthy subjects were exposed to mock, infective DV-2 (DV), or heat-inactivated DV-2 (heat DV), and caspase-1 activation was evaluated through the MFI of the fluorescent probe FLICA. (B-D) Platelets were exposed to mock, DV, or heat DV in the presence or absence of the caspase-1 inhibitor YVAD. B depicts the percentage of IL-1β–expressing platelets, and panel C shows the concentration of IL-1β in the supernatant of platelets incubated in each condition. Panel D shows the percentage of IL-1β–containing MPs for each condition. The bars represent the mean ± SEM of 6 to 8 independent experiments using samples from different healthy donors. *P < .05 and **P < .01 compared with mock exposed platelets that received the same treatment (dimethylsulfoxide [DMSO] or YVAD). #P < .05 between DV-2–treated platelets that were incubated with YVAD or DMSO.
Mitochondrial oxidant signaling induces inflammasome activation in DV-exposed platelets
Excess production of ROS by mitochondria activates the NLRP3-inflammasome assembly in monocytic cells.15 We recently reported increased generation of mitochondrial-derived ROS in platelets from patients with dengue.22 To investigate the role played by mitochondrial-derived ROS in platelet inflammasome activation, platelets were treated with the mitochondrial targeted antioxidant mitoTEMPO prior to being exposed to DV-2. As previously demonstrated,22 generation of mitochondrial ROS was significantly higher in platelets exposed to DV-2 compared with mock or to heat inactivated DV-2. As expected, exposure of platelets to DV-2 in the presence of mitoTEMPO significantly (P < .05) reduced mitochondria-generated ROS (Figure 5A). Increase in caspase-1 activation and IL-1β secretion elicited by DV-2 were similarly impaired (P < .05) by treatment with mitoTEMPO (Figure 5B-C), indicating that DV-triggered platelet inflammasome activation depends on mitochondrial signaling.
DV-elicited inflammasome activation depends on mitochondrial-derived ROS and RIP kinases. Platelets from healthy subjects were exposed to mock, infective DV-2 (DV), or heat-inactivated DV-2 (heat DV) in the presence or absence of the mitochondrial targeted antioxidant mitoTEMPO or the RIP1 inhibitor necrostatin-1 (Nec-1). Panels A and B show the fold changes for (A) MitoSox Red fluorescence and (B) caspase-1 activation related to mock values. (C-D) The levels of (C) IL-1β and (D) RANTES were determined in the supernatants of platelets incubated in each condition. The bars represent the mean ± SEM of 6 independent experiments using samples from different healthy donors. *P < .05 and **P < .01 compared with mock exposed platelets that were treated with the same drug. #P < .05 between DV-2–exposed platelets that were incubated with mitoTEMPO or Nec-1 and platelets that were incubated with DMSO. ND, not detected; ns, nonsignificant.
DV-elicited inflammasome activation depends on mitochondrial-derived ROS and RIP kinases. Platelets from healthy subjects were exposed to mock, infective DV-2 (DV), or heat-inactivated DV-2 (heat DV) in the presence or absence of the mitochondrial targeted antioxidant mitoTEMPO or the RIP1 inhibitor necrostatin-1 (Nec-1). Panels A and B show the fold changes for (A) MitoSox Red fluorescence and (B) caspase-1 activation related to mock values. (C-D) The levels of (C) IL-1β and (D) RANTES were determined in the supernatants of platelets incubated in each condition. The bars represent the mean ± SEM of 6 independent experiments using samples from different healthy donors. *P < .05 and **P < .01 compared with mock exposed platelets that were treated with the same drug. #P < .05 between DV-2–exposed platelets that were incubated with mitoTEMPO or Nec-1 and platelets that were incubated with DMSO. ND, not detected; ns, nonsignificant.
To determine whether antioxidant treatment impaired inflammasome activity in a selective fashion, we evaluated the effects of mitoTEMPO on DV-triggered release of the chemokine RANTES/chemokine ligand 5, which is preformed and stored in platelet granules.8 As shown in Figure 5D, treatment with mitoTEMPO did not inhibit the secretion of RANTES in platelets exposed to DV-2, suggesting specificity of the ROS signaling to the NLRP3 inflammasome.
Essential role for RIP proteins in DV-induced inflammasome activation in platelets
RIP-1/RIP-3 kinases have been shown to activate NLRP3-dependent caspase-1 activity by promoting mitochondrial ROS production.29 Considering the critical role for mitochondrial ROS in DV-induced activation of the inflammasome in platelets, we tested whether the RIP-1/RIP-3 inhibitor Nec-1 influences the generation of mitochondrial-derived ROS in platelets exposed to DV. As shown in Figure 5A, exposure of platelets to DV-2 in the presence of Nec-1 prevented mitochondrial generation of ROS. Consistent with inhibition of the inflammasome by mitoTEMPO and ROS being an effector in RIP signaling, activation of caspase-1 and secretion of IL-1β elicited by DV-2 were also impaired (P < .05) by treatment of platelets with Nec-1 (Figure 5B-C). Similar to mitoTEMPO, treatment with Nec-1 did not affect the release of preformed RANTES by DV-stimulated platelets (Figure 5D).
Platelet IL-1β synthesis and processing is associated to vascular permeability in dengue
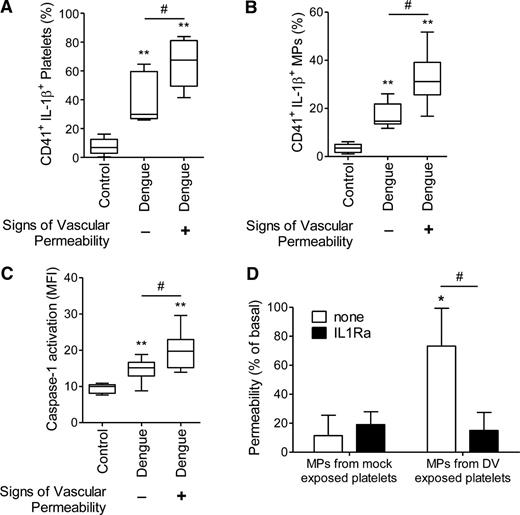
Increased vascular permeability in dengue patients was evidenced by ≥1 of the following signs: increase in hematocrit >20%, hypoalbuminemia, postural hypotension, ascites, and/or oliguria.2 According to the presence or absence of these signs, 45% of patients were classified as positive and the remaining 55% as negative for signs of increased vascular permeability. Characteristics of dengue-infected patients positive for signs of increased vascular permeability are presented in supplemental Table 1. The percentage of IL-1β–positive platelets and IL-1β–rich platelet-derived MPs was significantly higher (P < .05) in patients who had signs of increased vascular permeability (65.4 ± 16.4% and 32.4 ± 11.4% for platelets and platelet-derived MPs, respectively) compared with patients who did not have evidence for altered vascular barrier function (38.9 ± 16.2% and 17.3 ± 5.2% for platelets and platelet-derived MPs, respectively) (Figure 6A-B). Similar results were observed for caspase-1 activation (20.1 ± 5.2 vs 14.8 ± 2.8 for patients who were positive or negative for signs of increased vascular permeability, respectively, P < .05) (Figure 6C).
Vascular permeability is associated to platelet shedding of IL-1β–containing MPs. A-C show the percentage of (A) IL-1β–expressing platelets and (B) IL-1β–containing platelet-derived MPs and (C) the MFI of FLICA indicating caspase-1 activation in platelets isolated from healthy volunteers (control) or from DV-infected patients (dengue) that were positive (+) or negative (–) for signs of vascular permeability. The boxes indicate the median and interquartile ranges and the whiskers indicate 5 to 95 percentile. **P < .01 compared with healthy volunteers. #P < .05 between patients that were positive and negative for signs of increased vascular permeability. (D) Passive leakage of fluorescein isothiocyanate–conjugated albumin across endothelial cell layers was assessed after cell incubation with MPs recovered from mock- or DV-exposed platelets in the presence or absence of soluble IL-1Ra. The bars represent the mean ± SEM of 4 experiments using platelets from independent healthy donors. *P < .05 compared with mock MP-exposed endothelial cells layers. #P < .05 between endothelial cells layers that were incubated with sIL-1Ra or vehicle.
Vascular permeability is associated to platelet shedding of IL-1β–containing MPs. A-C show the percentage of (A) IL-1β–expressing platelets and (B) IL-1β–containing platelet-derived MPs and (C) the MFI of FLICA indicating caspase-1 activation in platelets isolated from healthy volunteers (control) or from DV-infected patients (dengue) that were positive (+) or negative (–) for signs of vascular permeability. The boxes indicate the median and interquartile ranges and the whiskers indicate 5 to 95 percentile. **P < .01 compared with healthy volunteers. #P < .05 between patients that were positive and negative for signs of increased vascular permeability. (D) Passive leakage of fluorescein isothiocyanate–conjugated albumin across endothelial cell layers was assessed after cell incubation with MPs recovered from mock- or DV-exposed platelets in the presence or absence of soluble IL-1Ra. The bars represent the mean ± SEM of 4 experiments using platelets from independent healthy donors. *P < .05 compared with mock MP-exposed endothelial cells layers. #P < .05 between endothelial cells layers that were incubated with sIL-1Ra or vehicle.
Moreover, we found that hematocrit values in DV-infected patients positively correlated with the percentage of IL-1β–positive platelets, platelet-derived MPs, and IL-1β–containing platelet-derived MPs. Further, patient serum albumin levels negatively correlated with IL-1β–positive platelets and caspase-1 activation. There was no significant association between inflammasome activation and platelet counts (Table 2).
Correlations of individual components of platelet inflammasome with laboratorial data from DV-infected patients
| Variable . | Hematocrit* . | Albumin† . | Platelet count* . | |||
|---|---|---|---|---|---|---|
| Pearson r . | P . | Pearson r . | P . | Pearson r . | P . | |
| IL-1β–positive platelets | 0.5725 | .0409 | −0.5726 | .0408 | −0.4181 | .1762 |
| Platelet-derived MPs | 0.4865 | .0296 | −0.2845 | .2855 | −0.4164 | .0857 |
| IL-1β–rich MPs | 0.6003 | .0301 | −0.3499 | .2915 | −0.4069 | .1892 |
| Caspase-1 activation | 0.4421 | .0864 | −0.5096 | .0437 | −0.2224 | .3751 |
| Variable . | Hematocrit* . | Albumin† . | Platelet count* . | |||
|---|---|---|---|---|---|---|
| Pearson r . | P . | Pearson r . | P . | Pearson r . | P . | |
| IL-1β–positive platelets | 0.5725 | .0409 | −0.5726 | .0408 | −0.4181 | .1762 |
| Platelet-derived MPs | 0.4865 | .0296 | −0.2845 | .2855 | −0.4164 | .0857 |
| IL-1β–rich MPs | 0.6003 | .0301 | −0.3499 | .2915 | −0.4069 | .1892 |
| Caspase-1 activation | 0.4421 | .0864 | −0.5096 | .0437 | −0.2224 | .3751 |
Significant correlations are in bold.
Hematocrit and platelet count analyses were available for all 36 patients.
Plasma levels of albumin were available for only 29 patients.
IL-1β in MPs from DV-activated platelets induces increased endothelial permeability
To examine the effects of IL-1β–containing platelet-derived MPs on endothelial permeability, we treated cultured human microvascular endothelial cells with MPs recovered from DV-2– or mock-exposed platelets before measuring permeability in a transwell assay. MPs from DV-2–exposed platelets caused an increase (P < .05) in permeability of HMEC-1 cells that was blocked by IL-1Ra (Figure 6D). These results indicate that IL-1β–rich platelet-derived MPs may activate endothelium, increasing its permeability during dengue illness.
Discussion
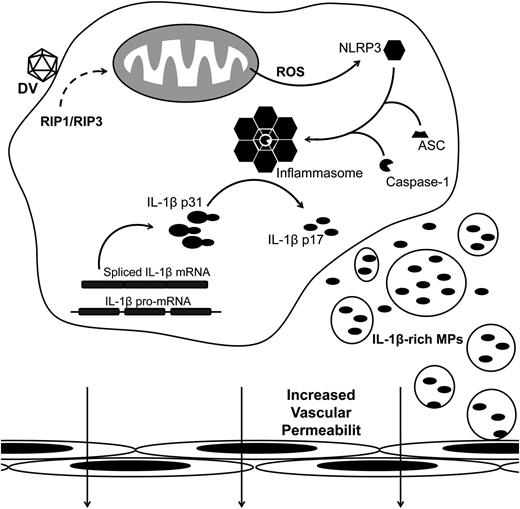
Thrombocytopenia and hemoconcentration are hallmarks of dengue illness. Although dengue pathogenesis is not fully elucidated, recent evidence supports a central role for proinflammatory cytokines in endothelial activation and plasma leakage during DV infection.5,9,30 Here we show that platelets from DV-infected patients contain both pro- and mature IL-1β (Figures 1A and 3E), which are also found in platelet-derived MPs in samples from infected patients (Figure 2B). Moreover, DV-activated platelets use caspase-1–dependent pathways to release IL-1β–bearing MPs, which also require RIP kinases and generation of ROS by mitochondria (Figures 3-5; summarized in Figure 7). Our data also indicate that IL-1β synthesis, activation of caspase-1, and release of IL-1β–rich MPs by platelets are associated with increased vascular permeability during dengue illness (Table 2; Figure 6).
Schematic representation for DV-induced platelet shedding of IL-1β–containing MPs. Platelets synthesize IL-1β after DV exposure. In parallel, RIP1/RIP3-mediated mitochondrial ROS generation activates NLRP3, which recruits ASC and caspase-1 to assemble the inflammasome complex. Active inflammasomes cleave the full length IL-1β (p31) into the mature cytokine (p17) and promote the shedding of IL-1β MPs. Platelet-derived IL-β–containing MPs contribute to the increase in endothelium permeability.
Schematic representation for DV-induced platelet shedding of IL-1β–containing MPs. Platelets synthesize IL-1β after DV exposure. In parallel, RIP1/RIP3-mediated mitochondrial ROS generation activates NLRP3, which recruits ASC and caspase-1 to assemble the inflammasome complex. Active inflammasomes cleave the full length IL-1β (p31) into the mature cytokine (p17) and promote the shedding of IL-1β MPs. Platelet-derived IL-β–containing MPs contribute to the increase in endothelium permeability.
We previously demonstrated that platelets are activated in patients with dengue.22 Dengue patients in the present study similarly had increased platelet P-selectin surface expression (data not shown). It is known that activated platelets mediate inflammatory and immune responses using a variety of mechanisms, including release of stored cytokines and synthesis of others, among them IL-1β.17,18,27 Our results indicate that DV induces platelets to synthesize IL-1β. A previous report from our group demonstrates that increased IL-1β in dengue patients correlates with thrombocytopenia.5 Conceivably, activated platelets may secrete high levels of IL-1β before being cleared or eliminated through apoptosis.22,31
The pathways involved in activation of post-transcriptional splicing and IL-1β synthesis by platelets were recently described,20,32 but the pathways that couple IL-1β synthesis with shedding of IL-1β–containing MPs remained unknown.20 We provide strong evidence for activation of NLRP3-inflammasome in platelets and its involvement in the shedding of IL-1β–containing MPs. It was recently reported that generation of ROS by mitochondria regulates the NLRP3 inflammasome response in monocytic cells.15 Furthermore, RIP1-RIP3 signaling has been shown to promote mitochondrial ROS production in response to viral infection or other proinflammatory stimuli.33,34 Consistent with these observations, our results indicate that RIP kinases activate caspase-1 inflammasomes in DV-exposed platelets by inducing mitochondria to generate ROS.
Increased caspase-1 gene expression was previously observed in DV-infected cultured cells.35 Caspase-1–dependent IL-18 cleavage was also previously reported.36 In addition, activation of the NLRP3-inflammasome was recently demonstrated in DV-infected macrophages in culture.16 Assembly of NLRP3 inflammasomes has also been described in other viral infections, including those by the DV family flavivirus.37,38 When hepatitis C virus infects hepatocytes, it induces the assembly of NLRP3–ASC–caspase-1 complex and, as a consequence, increases IL-1β secretion.37 Here, we observed evidence for ASC recruitment by NLRP3 leading to caspase-1 activation in platelets from DV-infected patients, providing new evidence for DV-induced inflammasome activation and its occurrence in this critical effector cell. Also, our studies indicate that activation of the caspase-1 inflammasome mediates shedding of IL-1β–containing MPs by platelets.
Shedding of MPs occurs as a facet the larger process of platelet activation, which is characterized by a multiplicity of subevents and regulatory mechanisms. MP release from activated platelets requires calpain to cleave moesin, a protein that links the underlying actin cytoskeleton with the plasma membrane.39-41 Calpain alone is not sufficient, however, suggesting a requirement for other proteases.40,42 Here we observed partial inhibition of release of platelet MPs (data not shown) and complete inhibition of release of IL-1β–containing MPs by blocking caspase-1 activation. Consistent with our data, it was previously shown that caspase-1 and caspase-3 are involved in moesin cleavage and MP release by agonist-stimulated platelets.42
MPs have been shown to modulate endothelial responses by delivering cytokines to endothelial cells. Brown and McIntyre demonstrated that IL-1β is found associated with MPs from LPS-stimulated platelets and that these MPs induced the expression of vascular cell adhesion molecule-1 on endothelial cells in an IL-1R–dependent manner.20 IL-1β–containing monocyte-derived MPs were also shown to activate human endothelial cells depending on IL-1R.43 The mechanisms by which IL-1β–containing MPs induce activation of IL-1R on target cells are not completely clear. It is known, however, that IL-1β is associated with the plasma membranes of activated platelets and that it can signal endothelial cells in a juxtacrine fashion without being released into solution. This signaling is blocked by IL-1RA.44 IL-1β associated with the membranes of platelet MP may signal in a similar fashion. Furthermore, Mause and coworkers45 have previously shown that platelet-derived MPs deposit RANTES on endothelial cells during transient interactions. It is also possible that a similar process enhances IL-1β delivery. We observed that MPs recovered from DV-exposed platelets triggered increased human endothelial cell permeability in a fashion dependent on IL-1β and blocked by the IL-1R antagonist. Consistent with these in vitro results, IL-1β synthesis, caspase-1 activation, and shedding of IL-1β–rich MPs were associated with enhanced vascular permeability in dengue-infected patients, suggesting that these events may contribute to the vasculopathy of dengue.
Although we provide evidence for IL-1β synthesis by platelets during dengue infection, we cannot exclude a contribution by other cell types. Mononuclear cells are important sources of IL-1β and IL-1β–containing MPs43 and have been shown to secrete IL-1β in response to DV infection in vitro.16 Nevertheless, platelets may be key sources of IL-1β in inflammation and infection8 and are major sources of circulating IL-1β in a mouse model of malaria.19 Although this study indicates that platelets may synthesize IL-1β in dengue infection, we cannot exclude platelet endocytosis of IL-1β or IL-1β synthesis by dengue-infected megakaryocytes. Nevertheless, our in vitro results confirm DV-triggered IL-1β synthesis by platelets. Whether this is due to outside-in activation of intracellular signaling pathways and/or internalization of DV into platelets46,47 requires further investigation. We also recognize that other factors including cytokines, antibodies, and coagulation factors may also contribute to platelet activation in dengue.48 It was previously shown that DV binding to platelets may be potentiated by the presence of DV-specific antibodies.49 Therefore, increased an DV-IgG immunocomplex in secondary infection might potentially amplify platelet IL-1β synthesis. It is also possible that increased LPS levels in patients with dengue50 contribute to activation of IL-1β synthesis and shedding of IL-1β–containing MPs in platelets.20
In summary, we report increased accumulation of IL-1β in platelets and platelet-derived MPs from patients with dengue and synthesis of IL-1β in platelets activated by DV in vitro. We also provide evidence for DV-triggered inflammasome activation in platelets and inflammasome-dependent shedding of IL-1β–containing MPs, which likely contributes to the development of increased vascular permeability and hemoconcentration during dengue illness.
The online version of this article contains a data supplement.
The publication costs of this article were defrayed in part by page charge payment. Therefore, and solely to indicate this fact, this article is hereby marked “advertisement” in accordance with 18 USC section 1734.
There is an Inside Blood commentary on this article in this issue.
Acknowledgments
The authors thank Gisele B. Lima for virus production; Alan B. Carneiro, Edson F. Assis, and Matheus A. Rajão for technical assistance; and the Programa de Desenvolvimento Tecnológico em Insumos para Saúde platform for multiplex analysis. The authors also thank Dr José Carlos Alves Filho for providing IL-1Ra.
This work was supported by grants from Conselho Nacional de Desenvolvimento Científico e Tecnológico, Fundação de Amparo a Pesquisa do Estado do Rio de Janeiro, Programa Estratégico de Apoio à Pesquisa em Saúde/Fiocruz, Instituto Nacional de Ciéncia e Tecnologia de Biologia Estrutural e Bioimagem, Programa de Apoio a Núcleos de Excelência Dengue, and National Institute of Science and Technology in Dengue, and the National Institutes of Health, National Heart, Lung, and Blood Institute grants (HL066277, HL091754, and HL044525 to A.S.W. and G.A.Z).
Authorship
Contribution: E.D.H. performed the majority of experiments, data analyses, and manuscript drafting and preparation; J.F.L. performed part of experiments and analyzed data; C.F. performed part of experiments, image analyses, and manuscript drafting and preparation; M.F.O. performed the experimental design and manuscript editing/review; M.T.B. performed the experimental design and manuscript editing/review; R.V.-d.-S. performed patient enrollment, clinical classification, and experimental analyses; A.T.D.P. performed experimental design and manuscript editing/review; A.S.W. performed experimental design and manuscript editing/review; G.A.Z. performed experimental design and manuscript editing/review; and F.A.B. and P.T.B. directed all aspects of the study, data analyses, and manuscript preparation and review.
Conflict-of-interest disclosure: The authors declare no competing financial interests.
Correspondence: Fernando A. Bozza, Instituto de Pesquisas Clinicas Evandro Chagas, Fundação Oswaldo Cruz, Avenida Brasil 4365, Rio de Janeiro, Brazil; e-mail: bozza.fernando@gmail.com; or Patricia T. Bozza, Laboratório de Imunofarmacologia, Instituto Oswaldo Cruz, Fundação Oswaldo Cruz, Avenida Brasil 4365, Rio de Janeiro, Brazil; e-mail: pbozza@ioc.fiocruz.br.
References
Author notes
F.A.B. and P.T.B. contributed equally to this work.



![Figure 4. Platelet shedding of IL-1β–rich MPs depends on caspase-1 activation. (A) Platelets from healthy subjects were exposed to mock, infective DV-2 (DV), or heat-inactivated DV-2 (heat DV), and caspase-1 activation was evaluated through the MFI of the fluorescent probe FLICA. (B-D) Platelets were exposed to mock, DV, or heat DV in the presence or absence of the caspase-1 inhibitor YVAD. B depicts the percentage of IL-1β–expressing platelets, and panel C shows the concentration of IL-1β in the supernatant of platelets incubated in each condition. Panel D shows the percentage of IL-1β–containing MPs for each condition. The bars represent the mean ± SEM of 6 to 8 independent experiments using samples from different healthy donors. *P < .05 and **P < .01 compared with mock exposed platelets that received the same treatment (dimethylsulfoxide [DMSO] or YVAD). #P < .05 between DV-2–treated platelets that were incubated with YVAD or DMSO.](https://ash.silverchair-cdn.com/ash/content_public/journal/blood/122/20/10.1182_blood-2013-05-504449/4/m_3405f4.jpeg?Expires=1765908074&Signature=g4nfEQ0K~Xoc-~h6ReNFoq1vFlTgLkeabLqoQsvHG8d8XL1q9Csp31nxFoY~udQHHkiKJk551UUIngdRLSWqu2BknmIfzLTKTKUlyDNserMuGRdUyuYNkh~9~-uyKXdnxXi2BYx76DvNLoWXsbAU8By4Kms4eV1tBibbuZPC~-plYuJMlZVe8H-GFBFHNVKm0tqR0Ol4muX-wVMB1HMRShBbTmmA5Gx2D-Udx7KL78538K-gDxmSz04fkMU~G7hGDCg~Rt7v77hjIHEblcWcqCQrvTLNPG9fF09KUjxEr6k1k1fWC51UdS6lrQ2~MHqtC6bGYtLLapjajI7~-2xKTA__&Key-Pair-Id=APKAIE5G5CRDK6RD3PGA)