Abstract
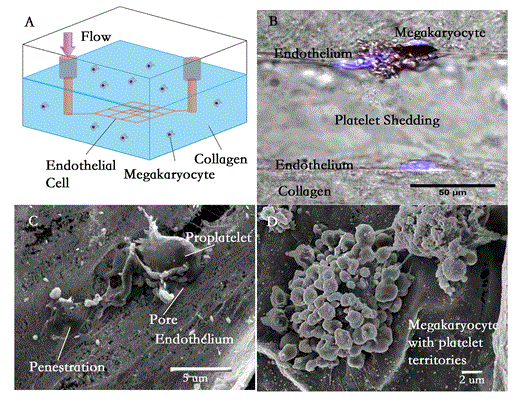
Platelets are anucleate blood cells, which, in addition to their primary function of preventing and arresting hemorrhage, play important roles in regulating vascular integrity, angiogenesis, inflammation, and the immune response. A healthy adult human produces approximately 150 billion platelets each day from marrow megakaryocytes. Megakaryocytes in the marrow arise from hematopoietic stem cells aligned alongside osteoblasts, migrate towards small blood vessels as they differentiate, and then shed platelets into the blood to eventually generate the entire circulating platelet pool. Despite the obvious necessity of this process for the maintenance of platelet number, little is known on the nature of the relationship between megakaryocytes and the marrow microvasculature. This is perhaps a consequence of the limited tools available to study this process. Existing in vitro models, which rely on culturing megakaryocytes on top of an endothelial cell monolayer, fail to recapitulate the 3D microenvironment including the vessel lumen, fluid flow and mass transport; animal models too are limited for the study of the individual cellular components in different combinations.
In summary, with this 3D megakaryocyte/microvessel co-culture system, we show that endothelial cells attract megakaryocytes to migrate towards microvessels through SDF-1/CXCR4 signaling. The megakaryocytes, in turn, induce fenestrations in the microvessels. The fenestrae act as “doors” for the megakaryocytes to enter the vessels, where they experience fluid shear stress to shed platelets from the proplatelet processes or megakaryocytes with well-defined platelet territories (we found evidence for both). These results demonstrate the usefulness of this system for the study of thrombopoiesis, and the possibility that it can be used to reconstitute the entire marrow microenvironment. Because it is possible to use entirely human components, this system can also be used to screen thrombopoietic drugs, to study disorders of platelet formation and structure, and can potentially be scaled up as a bioreactor to produce functional platelets for transfusion.
Sniadecki:Stasys Medical Corporation: Equity Ownership, Founder Other.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal