Abstract
Recent studies demonstrated that early molecular response was predictive for outcome in chronic phase chronic myeloid leukemia (CML) patients on imatinib (IM) (Hanfstein et al., Leukemia 2012, Marin et al., JCO 2012). It is thus essential to identify patients likely to achieve better long-term outcome if they are switched early to second-line therapy. In reference laboratories, BCR-ABL1 transcript levels are expressed as BCR-ABL/ABL ratios (%) according to the international scale (IS, Hughes et al., Blood 2006). Nevertheless, for high levels of BCR-ABL1, the copy number of total-ABL1 comprises a part of BCR-ABL1, which results in underestimation of the ratio at diagnosis. But the BCR-ABL1 levels for which this underestimation begins to be significant is currently unknown. Besides, since the cut-off value of 10% seems critical to predict outcome for patients treated with IM for 3 months, it stresses the necessity of a precise quantification at this level. In this study, we evaluated the ability of GUS, identified as another suitable control gene (CG) (Beillard et al., Leukemia 2003), to accurately define the relevant thresholds for known therapeutic decision levels (10% and major molecular response MMR: 0.1%) in a series of front line therapy patients on IM.
We collected 141 blood samples of CML patients between August 2000 and December 2012, including a first group of diagnosis samples (n= 83) and follow-up samples chosen according to their transcript value: the second group with a BCR-ABL1/ABL1 ratio between 6 and 14% (n=21) and the third group at the MMR threshold of 0.1% (n=37). The expression of BCR-ABL1, total-ABL1 and GUS were quantified as previously described (Gabert et al., Leukemia 2003). Results were expressed as percent ratios relative either to ABL1, with original values converted to IS, or to GUS, without transformation of the original values.
The mean BCR-ABL1/ABL1 ratio was 55.91% (range: 34.83-98.01%) at diagnosis, 9.66% (range: 6.72-13.53%) in the second group (6-14% values) and 0.093% (range: 0.05-0.18%) in the third group (MMR patients).
To determine the impact of GUS as CG on the MMR threshold, we compared in the third group the copy number ratios of BCR-ABL1 relative either to ABL1 (=0.093%) or GUS. The BCR-ABL1/GUS mean value was 0.036%. Thus, the translation of the MMR threshold from BCR-ABL1/ABL1 to BCR-ABL1/GUS would need the introduction of an additional conversion factor (Figure 1). This factor is defined as the ratio of (BCR-ABL1/ABL1)/(BCR-ABL1/GUS) =GUS/ABL1 for each measurement and its mean was valued at 2.774 (2.507-3.040, 95% confidence interval).
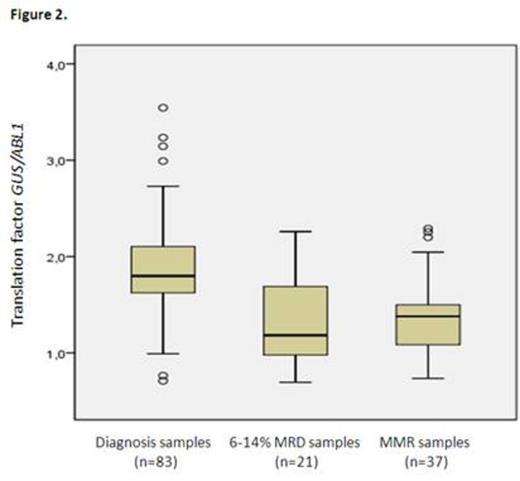
To assess whether this factor was constant within the different disease time, we next calculated it for the two other groups (figure 2). In diagnosis samples, the mean value of the factor was 3.772 (3.554-3.991, 95%CI) whereas it valued at 2.668 (2.263-3.072, 95%CI) in samples with a 6-14% BCR-ABL1/ABL1 ratio. Comparison of means showed a significant difference between the 3 groups (p<0.001, ANOVA test). This difference over time in the course of the disease was due to GUS overexpression in CML cells at diagnosis, GUS overexpression being superior to the overestimation of ABL1 at diagnosis due to the quantification test bias (Figure 3). This overexpression of GUS, previously reported (Beillard et al., Leukemia 2003), impacts the BCR-ABL1/GUS ratio at diagnosis and warrants the use of a unique translation factor for all disease levels. The number of copies of GUS did not differ significantly between the 6-14% samples and MMR samples (p=0.123, Student's T-Test), nor did the ABL1 copy number (p=0.06). This means that the quantification bias observed for diagnosis samples does not impact the values under 14%.
The use of GUS as CG for the MRD would require defining the factor allowing translation between BCR-ABL1/GUS and BCR-ABL1/ABL1 ratios, which implies a new standardization of the international scale. Nevertheless, this factor seems to depend on the transcripts levels, since the value of BCR-ABL1/GUS obtained at diagnosis is impacted by the higher GUS copy number in CML cells. As the copy number of ABL1 and GUS did not differ significantly between the groups of patients with 0.1% and 6-14% BCR-ABL1/ABL1 ratio, we concluded that there is no quantification bias at these critical therapeutic decision levels. As a consequence, ABL1 as GUS may be used efficiently as CG to assess early response to TKI.
Cony-Makhoul:BMS: Honoraria. Michallet:MSD: Consultancy, Honoraria, Research Funding; Astellas: Consultancy, Honoraria, Research Funding; Novartis: Consultancy, Honoraria, Research Funding; Pfizer: Consultancy, Honoraria, Research Funding; Bristol-Myers Squibb: Consultancy, Honoraria, Research Funding; Genzyme: Consultancy, Honoraria, Research Funding. Nicolini:Novartis, Ariad, Teva, BMS and Pfizer: Honoraria from Novartis, Ariad, Teva, BMS and Pfizer. Grants from Novartis. Other.
Author notes
Asterisk with author names denotes non-ASH members.



This feature is available to Subscribers Only
Sign In or Create an Account Close Modal