Abstract
Anthracycline-containing regimens (CHOP and CHOP-like regimens) are still the cornerstone of elderly patients with diffuse large B-cell lymphoma (DLBCL), although the therapeutic response is acceptable, high incidence of co-morbidities and treatment-related adverse effects including cardiac and hematologic toxicities are major obstacle in clinical practice, and management of elderly DLBCL patients remains a great challenge. We employed a platinum-based regimen (oxaliplatin-gemcitabine) which is widely used to treat solid tumors plus anti-CD20 antibody rituximab(R-GemOx) as first-line treatment in elderly patients with DLBCL. We have performed an interim analysis of this ongoing Phase 2 study and evaluated safety and efficacy of this regimen.
This study was a prospective, multicenter, single-arm phase 2 clinical trial (NCT01670370). Eligible patients included those with previously untreated, CD20 positive DLBCL, ≥70 years of age or ≥60 years with ECOG performance status score ≥2. Therapeutic protocol of R-GemOx regimen was as follows: rituximab 375 mg/m2 i.v. on day 1; gemcitabine 1 g/m2 i.v. on day 2; oxaliplatin 100 mg/m2 i.v. on day 2, each cycle of treatment was administered every 14 to 21 days according to recovery of toxicities. Interim evaluation was conducted after 3 cycles of treatment, patients who responded to the treatment and did not have disease progression received up to six cycles of R-GemOx treatment.
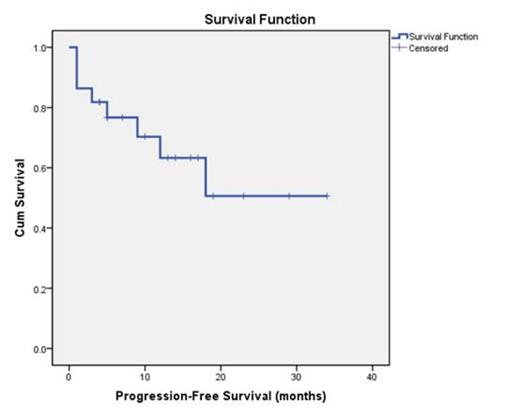
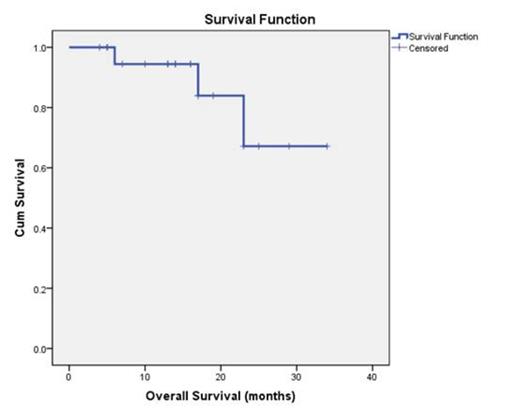
At the time of interim analysis, 25 patients were enrolled between July 2010 and March 2013 and 22 patients were eligible to evaluate. Patients were 50.0% (11/22) male with median age 74 (range: 61-85) yrs, four patients (18.2%) were <age 70. ECOG PS ≥2 was 16/22 and the international prognostic score (IPI) enrolled to study was low risk group (0-1 score) 4/22, low intermediate group (2 score) 7/22, high intermediate/ high risk group (3-5 score) 11/22. The median number of cycles of therapy was 5, three pts have discontinued study due to progression. Evaluation of response was according to response criteria for lymphoma: The overall response rate (ORR) among treated pts was 81.8%, with 63.6% of patients achieving CR/CRu and 18.2% achieving PR, one patient (4.5%) had SD and 3 patients (13.3%) had PD. With the median follow-up of 15 months (4-34), progression-free survival rate was 50.6% (Fig. 1), and overall survival rate was 67.2% (Fig. 2). In terms of toxicities, most treatment-related adverse events (AEs) were mild or moderate in severity. The most common grade ≥3 AEs were gastrointestinal complications including diarrhea and vomiting (18.2%) and bone marrow suppression (13.6%), other nonserious AEs observed included hypodynamia, mild paresthesia and hepatic injury. No clinical impairment of cardiac and nephrotoxicity were observed.
R-GemOx immunochemotherapy regimen is highly active and well-tolerated in previously untreated elderly patients with DLBCL, gastrointestinal complications and hematologic toxicities can occur and should be carefully monitored, the encouraging interim data suggest R-GemOx is a promising first-line regimen for this subgroup of patients, Longer follow-up and further evaluation are required to evaluate efficacy and safety of this regimen.
No relevant conflicts of interest to declare.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal