Abstract
Waldenstršms macroglobulinemia (WM) is a rare and incurable indolent Non-Hodgkin's lymphoplasmacytic lymphoma. Rarity and limited availability of authentic preclinical models has been a major constraint in drug development for WM. We recently reported the establishment and characterization of a novel human WM cell line, RPCI-WM1. Thereafter we evaluated the tumorigenic potential of this unique WM cell line in a xenograft mouse model along with its ability to produce human IgM in vivo. Moreover, we assessed its potential as a possible model for enhanced understanding of WM cell biology in vivo and a mechanism to conduct functional studies and preclinical anti-WM drug evaluation.
The primary goal of this experiment was to develop and validate an in vivo WM model that can be used to investigate WM cell biology as well as drug evaluation.
The RPCI-WM1 cell line (1x106 cells) was injected both subcutaneously and via tail vein into female SCID mice. Engraftment was validated via detection of human IgM (ELISA) and tumor growth (as well as response to therapy) was monitored by weekly assessment of IgM levels and external tumor formation measurements.
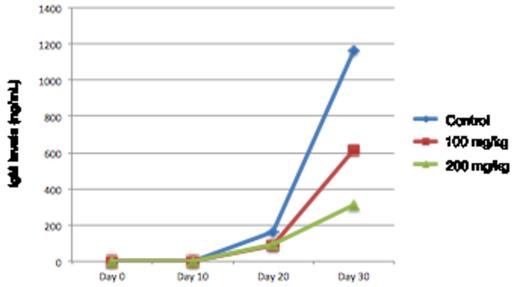
After subcutaneous injection (flank) into mice, the RPCI-WM1 cells developed localized tumors that secreted human IgM detectable in serum as early as 14 days. Progressive increase in IgM was measured in the serum and this corresponded with tumor growth in a time-dependent fashion. On average, tumor tissues grew to a median of 3 cm and IgM levels reached a median of 823 ng/mL by Day 30 post-implantation. We also assessed the practical utility of this xenograft mouse model for evaluation of therapeutics, using a compound X (propriety drug with demonstrable in vitro anti-WM effects). Eleven days post-implantation, the mice were randomized into 3 groups receiving either DMSO (n=6), 100 mg of compound X (n=7) or 200 mg/kg of compound X (n=7) every alternate day via intraperitoneal injection for 26 days. On Day 37, mice were sacrificed and tumor volume was measured. We observed a 48% reduction in tumor volume in the 100mg/kg cohort, with no additional anti-WM benefit noted in the 200mg/kg cohort. Human IgM secreted by the xenografted tumors was measured by ELISA and was significantly lower in mice treated with compound X versus the control group (Figure 1). We also conducted immunohistochemistry (IHC) analysis on tumor tissues obtained from 9 mice treated with compound X (both dose cohorts) or DMSO alone. IHC staining demonstrated changes in the known target proteins of compound X (such as BCL2, p53, Ki-67) between the treated and untreated mice.
Reduced IgM secretion in RPCI-WM1 xenografted mice treated with compound X.
Thus we demonstrate that the RPCI-WM1 cell line can be dependably used to induce tumor growth through xenotransplantation in SCID mice and this mimics the IgM secreting clinical disease model. This can then be used to serve as a platform for preclinical testing of novel anti-WM therapeutics and allow for subsequent correlative studies for pharmacodynamics purpose. Comprehensive data regarding this model its utility in evaluation of various biological questions such as those relevant to the tumor microenvironment as well therapeutic modeling will be presented.
We are grateful to the Leukemia and Lymphoma Society and the International Waldenstršm Macroglobulinemia Foundation for their continued support.
No relevant conflicts of interest to declare.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal