Abstract
Advanced-stage, relapsed indolent non follicular lymphomas (INFLs) have a relatively poor prognosis, with low complete response to conventional chemotherapy and short survival. Thus, there is a need for innovative treatment with high efficacy and a good safety profile. Lenalidomide (R®) is an immunomodulatory drug with a direct tumoricidal effect and action on T, NK and stromal cells that may enhance antibody-dependent cell mediated cytotoxicity as well as the development of specific anti-tumor immune responses. These immunologic effects may synergize with the action of rituximab (R). To test the efficacy of R® combined with R (R2), we have conducted a multi center, open label phase II clinical trial in patients (pts) with relapsed INFL.
Eligible pts must have indolent non follicular B-cell lymphoma relapsed after at least 2 but less than 4 prior lines of R-containing immuno-chemotherapy with measurable disease. Patients received oral R® 20 mg once daily on days 1-21. R is administered at a dose of 375 mg/m2 at day 14 of every course. Treatment is repeated every 28 days for up to 6 courses. The primary objectives of the study were to evaluate the antitumor activity of oral R® when given in combination with R and to assess the safety of R2 regimen evaluated by standard criteria (CTC-NCI 3.0). The secondary objectives were the evaluation of progression free survival (PFS) and overall survival (OS).
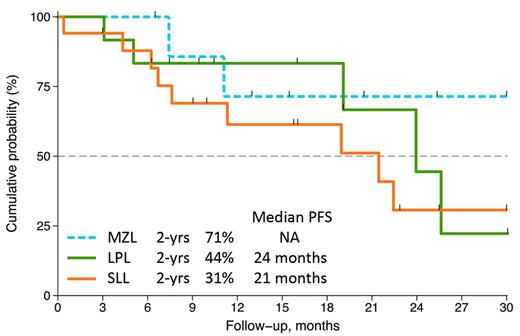
From July 2010 and March 2013, 44 pts entered the protocol. Six out of 44 pts were excluded from this analysis as 2 withdrew informed consent and 4 refused to start treatment immediately after signing the informed consent. Enrolled pts (38 cases) had: 18 small lymphocytic lymphoma (SLL), 12 lymphoplasmacytic lymphoma (LPL) and 8 marginal zone lymphoma (MZL). Median age was 68 years (51-75) and 58% were male. LDH value was increased in 21% of pts and β-2-microglobulin in 75%; 51% of pts had Hb<12 g/dL and 66% had bone marrow involved (median infiltrating 40%). Of the 38 pts, 7 achieved a complete remission and 14 a partial remission with an ORR of 55%. Seven pts had a stable disease and 5 a lymphoma progression. In general, the regimen R2 was relatively well-tolerated. Grade 3-4 hematological events were observed in 22 pts. The most common adverse events were neutropenia (58%), thrombocytopenia (11%), anemia (10%) and infection (10%). Grade 3-4 non hematological events were fever (5%), dyspnea (3%), allergic reaction to R (3%), renal failure (3%) and erythema (3%). Growth factors were administered in 58% of pts. The median dose intensity was 0.94 for R and 0.98 for R®. With a median follow-up of 19 months (range 1-43), overall 6 pts died, 5 for lymphoma progression and 1 for treatment related toxicity. The 2-years OS and the 2-years PFS were shown in Figure 1. Figure 2 shows the PFS by histology. The percentage of 2-years PFS (71%) for MZL appear impressive.
The chemo-free R2 scheme as treatment for relapse INFLs produces response in about 50% of pts and remission appear durable in pts with MZL. The toxicity profile of the combination is tolerable with manageable hematologic side effects. These results support the design of clinical trial with this chemo-free combination as first line treatment for INFLs, in particular for MZL.
No relevant conflicts of interest to declare.
Author notes
Asterisk with author names denotes non-ASH members.


This feature is available to Subscribers Only
Sign In or Create an Account Close Modal